Your new post is loading...

|
Scooped by
Beeyond
May 4, 3:01 AM
|
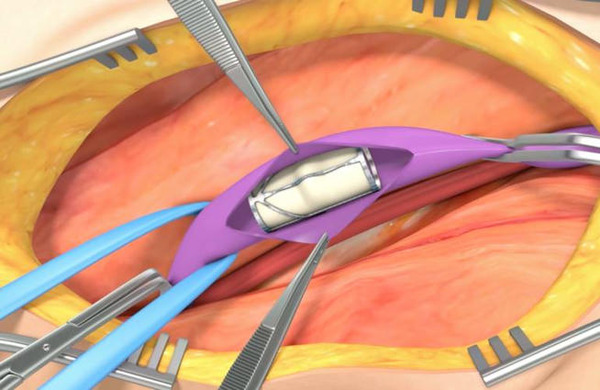
enVVeno Medical has received the investigational device exemption (IDE) approval from the US Food and Drug Administration (FDA) for a pivotal study involving a non-surgical replacement venous valve. The approval allows enVVeno Medical to begin the Transcatheter Venous Valve Endoprosthesis (TAVVE) pivotal study of its enVVe system for treating severe deep chronic venous insufficiency (CVI).

|
Scooped by
Beeyond
May 4, 3:00 AM
|
The FDA’s letter indicates that, upon completing its review, it determined that it is unable to approve the PMA for VenoValve in its current form. According to a news release, the FDA indicated that the favorable revised Venous Clinical Severity Score (rVCSS) data generated by the study to show clinical improvement, together with the improvements in pain scores and venous-specific quality of life indicators, were not sufficient on their own to determine the favorability of the risk-benefit profile for VenoValve.
EnVVeno Medical said that, without a specific hemodynamic measurement that correlates with patient improvement, the FDA raised concerns about bias. The agency also considered the possibility that clinical improvement occurred as a result of the patients being enrolled in a study.
Additionally, the company said the FDA focused on safety concerns attributed to the associated open surgical procedure. That required rehospitalizations, the company said. However, it would not expect to see similar safety events with a non-surgical replacement valve.

|
Scooped by
Beeyond
May 4, 2:53 AM
|
Patients with non-ST-elevation myocardial infarction (NSTEMI) or unstable angina undergoing a percutaneous coronary intervention (PCI) with a sirolimus-eluting balloon and rescue stenting fared as well as those who underwent revascularization with systematic use of drug-eluting stents at 1 year, according to a new substudy of the SELUTION DeNovo trial.

|
Scooped by
Beeyond
April 27, 6:22 AM
|
EuroPCR has long been a platform for innovation. In 2026, this commitment is strengthened with a newly expanded focus and for the first time, the Innovation Hub - a dedicated space with distinct zones. Designed to connect the full innovation ecosystem, it will create new opportunities to explore emerging technologies, give you a central place to share experiences and discuss how today’s ideas could become tomorrow’s clinical practice.

|
Scooped by
Beeyond
April 27, 3:42 AM
|
The anti–protein adsorption tests showed that the PCBDA/PDA–PEI coating resisted the non–specific adsorption of fibrinogen, which was a positive signal for enhanced anti–coagulation. Moreover, the platelet adhesion/activation tests confirmed its antithrombotic properties. The in vivo tissue response was also greatly suppressed due to the inhibited recognition of PCBDA/PDA–PEI–coated implants by macrophages. Interestingly, unlike traditional zwitterionic polymers, due to the introduction of adhesive catechol moieties, PCBDA supported the growth of endothelial cells, demonstrating its ability to selectively direct the fate of endothelial cells and smooth muscle cells. In vivo stent implantation results showed inhibited intimal hyperplasia due to the protection of PCBDA/PDA–PEI. Overall, due to the specific characteristics of the mussel–inspired PCBDA/PDA–PEI coating, anticoagulation, anti–inflammatory, and anti–proliferation properties were obtained; thus, it is expected that this coating can be used to inhibit restenosis and realize endothelialization after stent implantation.

|
Scooped by
Beeyond
April 27, 3:41 AM
|
Acute Coronary Syndrome (ACS) occurs when blood flow to the heart is suddenly reduced, putting the heart muscle at risk. ACS often leads to Non-ST-Elevation Myocardial Infarction (NSTEMI), which accounts for 70% of heart attacks.
Most of these patients undergo percutaneous coronary intervention (PCI), or an angioplasty, to restore blood flow. Currently, during PCI, a cardiologist uses a drug-eluting stent (DES). A DES is a tiny metal mesh tube that keeps the artery permanently open. Studies have shown that there is a 1–4% annual rate of complications due to the permanent presence of metal in the artery.
The SELUTION Drug Eluting Balloon (SEB) is a drug-coated balloon that delivers medication directly to the artery wall without leaving a stent behind. This "Leave-Nothing-Behind" strategy, therefore, potentially reduces the long-term complications related to stents. However, the use of SEB in patients with ACS has not been assessed.

|
Scooped by
Beeyond
April 27, 3:40 AM
|
A new sub-study suggests that using a sirolimus-eluting balloon (SEB), a drug-coated balloon, can reduce the number of stents a patient may need, making it a safe and effective way to treat certain heart attacks or unstable chest pain. Researchers presented the late-breaking data today at the Society for Cardiovascular Angiography & Interventions (SCAI) 2026 Scientific Sessions & Canadian Association of Interventional Cardiology/Association Canadienne de cardiologie d'intervention (CAIC-ACCI) Summit in Montreal.

|
Scooped by
Beeyond
April 27, 3:40 AM
|
A novel sirolimus-eluting balloon (Selution; Cordis) compares favorably with DES in patients with ACS, according to a subanalysis of the SELUTION DeNovo trial. These findings support those of the main trial, presented at TCT 2025, which showed that use of the drug-coated balloon (DCB) plus provisional stenting as needed was noninferior at 1 year as an alternative to DES for the primary endpoint of target vessel failure, which included cardiac death, target-vessel MI, and clinically driven target-vessel revascularization.

|
Scooped by
Beeyond
April 26, 2:51 PM
|
“The reason for stent graft failure is neointernal hyperplasia that occurs mainly on the edge of the device and sometimes in the middle of the stent graft,” Harduin detailed. The Solaris DE covered stent is built with an impermeable electrospinning PTFE membrane to limit cellular migration, allied with the sirolimus coating to block neointimal hyperplasia and restenosis. “Sirolimus is a safe and effective cytostatic drug that has been used for a long time in interventional cardiology in coronary arteries to prevent neointimal hyperplasia, restenosis, and thrombosis,” said Harduin.
Animal studies of Solaris DE have, to date, shown the persistence of the sirolimus on the outer edge of the device, he said, with one study comparing it to a conventional stent graft finding no instances of neointimal hyperplasia at 60 days following Solaris DE implantation, compared to 4.9mm3 with the conventional device.

|
Scooped by
Beeyond
April 26, 2:50 PM
|
Concept Medical Inc., a global pioneer in innovative drug delivery technology, is proud to announce the successful enrollment of the first patient in the STARS DAPT (ST-Segment Elevation Myocardial InfArction treated with a Polymer-Free Sirolimus-based NanocarrieR Eluting Stent and a P2y12 inhibitor-based Single Antiplatelet Strategy after a Short Dual Antiplatelet Therapy versus conventional Dual AntiPlatelet Therapy) randomized controlled trial. This trial marks a significant milestone in evaluating the breakthrough technology of polymer free drug eluting stent (DES) for the treatment of STEMI patients.
The first four patients were enrolled by Professor Barbara Stähli and her team at the University Hospital of Zurich in Switzerland initiating the much awaited RCT which will assess the different strategies to manage ACS patients with antiplatelet regimen.
STARS DAPT is an investigator-initiated, prospective, multicentre, international, open-label, randomized controlled pilot study. The trial will enrol 350 all-comer subjects with STEMI undergoing primary PCI with the Abluminus np (Concept Medical). STARS DAPT aims to study the difference between a P2Y12 receptor inhibitor-based single antiplatelet therapy (SAPT) regimen after a short period of dual antiplatelet therapy (DAPT) versus conventional 6 or 12 months of DAPT. The co-primary endpoints at 12 months are major adverse cardiac and cerebrovascular events and major bleeding complications.

|
Scooped by
Beeyond
April 26, 2:41 PM
|
The BARISTA trial, led by Lieven Maene (Onze Lieve Vrouw, Aalst, Belgium), enrolled 200 patients with all types of iliac lesions. Significantly, 34% of the study population presented with TASC C and D lesions, representing more complex anatomical scenarios and underscoring the robustness of the clinical evaluation.
At one-year follow-up, the trial demonstrated positive clinical outcomes: - Freedom from target lesion revascularisation (fTLR): 96.4%
- Freedom from restenosis: 94.1%

|
Scooped by
Beeyond
April 26, 2:39 PM
|
Launched in Europe in 2016 and the USA two years later following successful results from the IMPERIAL randomised controlled trial (RCT),1 the Eluvia DES is designed to provide sustained drug release and efficient drug transfer with the aim of overcoming the challenge of restenosis in the superficial femoral artery (SFA).
Steiner’s clinical experience with the Eluvia DES dates back several years. At this time, she and colleagues conducted the BEST-SFA RCT, in which Eluvia was used as a comparator. “What we observed in this analysis, and in our own clinical experience, was that the stent performed very well, effectively inhibiting the need for reintervention.” Steiner recalls. Two-year results of this study were published in 2024 in JACC: Cardiovascular Interventions.2

|
Scooped by
Beeyond
April 26, 2:38 PM
|
The 12-month outcomes were presented in a late-breaking session at the Society of Interventional Radiology (SIR) annual scientific meeting (11–15 April, Toronto, USA). A study intended to expand real-world experience, the prospective, multicentre study evaluated the IN.PACT AV DCB across 17 US clinical sites, with independent core laboratory and clinical events committee oversight.
The post‑approval study was designed as a confirmatory extension of the pivotal randomised controlled trial of the device—further strengthening the evidence base by evaluating performance in a real‑world AV access population with high comorbidity burden, Medtronic states in a press release.

|
Scooped by
Beeyond
April 26, 2:36 PM
|
“Robocath represents a highly strategic addition to Stereotaxis, amplifying and accelerating our strategy as the leading robotic platform for the broad spectrum of endovascular procedures,” said David Fischel, Stereotaxis chairman and CEO. “By combining our complementary robotic mechanisms, we are creating a uniquely capable platform that expands our reach across interventional medicine. This transaction accelerates our strategy, enhances our technological leadership, provides attractive commercial synergies, opens new avenues for growth, and supports other strategic opportunities being pursued by Stereotaxis.”

|
Scooped by
Beeyond
April 26, 2:34 PM
|
Three trials presented at the American College of Cardiology’s Annual Scientific Session (ACC.26) examined the role of cardiac imaging in clinical decision-making. Two studies used intravascular ultrasound (IVUS) to guide percutaneous coronary intervention (PCI), and the other used coronary computed tomography angiography (CCTA) to measure whether a new drug could slow plaque progression. None of the trials, however, showed significantly improved outcomes in the specific patient populations studied.

|
Scooped by
Beeyond
April 26, 2:32 PM
|
Fondazione Ricerca e Innovazione Cardiovascolare ETS (Fondazione RIC), an independent non-profit cardiovascular research foundation and Educación en Procedimientos de Intervencionismo en Cardiología (Fundación EPIC), the Spanish interventional cardiology education and research foundation, announced the launch of the clinical trial—PICCOLETO IV—with the first patient enrolment at Auxologico Research Hospital (Milan, Italy).
PICCOLETO IV is an international multicentre, investigator-driven, open-label, randomised (1:1), clinical investigation designed to observe and evaluate the rate of ischaemic and bleeding adverse events of SAPT after PCI with latest-generation DCBs in an elderly or high-bleeding risk population with stable or unstable coronary syndromes.

|
Scooped by
Beeyond
April 26, 2:29 PM
|
In a 2026 Charing Cross (CX) Symposium (21–23 April, London, UK) session that combined podium-first presentations with debate, subsequent audience polling revealed an even split over the routine use of sirolimus in chronic limb-threatening ischaemia (CLTI) patients; 47% of attendees voted in favour of the routine use of sirolimus DCBs in the femoropopliteal segment—at least in CLTI patients— to just 38% against, with 15% of attendees undecided. This result was reflected by the panel who broadly agreed that the question should no longer be “if” sirolimus should be used, but when and where.

|
Scooped by
Beeyond
April 26, 2:28 PM
|
Results from the extended cohort were being presented for the first time, following the first reveal of the 186-patient primary cohort of AV fistula patients at the 2026 Society of Interventional Radiology (SIR) annual scientific meeting (11–15 April, Toronto, Canada). The group included a broader pool of patients, including both AV fistulas and grafts. Baseline characteristics were typical of the end-stage kidney population, though, “of note, there were more female Black patients in the AV graft group in the extended cohort due to one site enrolling the majority of the patients”.
The mandated primary endpoint—serious infection rate at 12 months—performance goal of 30% was met. Among the 99 patients in the extended group, Misra reported the infection rate was higher in those with AV grafts and “favourable compared to prior literature”.

|
Scooped by
Beeyond
April 24, 11:42 AM
|
La Graduate School Cardiovascular Sciences élargit son périmètre en intégrant l’hématologie et devient la Graduate School Cardiovascular & Blood Sciences (CVBS), dédiée aux sciences cardiovasculaires et au sang. Par cette évolution, l’Université Paris Cité renforce son ambition en santé globale et propose un pôle d’excellence unique en Europe, fondé sur une approche décloisonnée du système circulatoire, considéré non plus comme une simple mécanique, mais comme un écosystème dynamique où le cœur, les vaisseaux et le sang interagissent constamment.

|
Scooped by
Beeyond
April 23, 9:28 AM
|
LRP5 interacts with the purinergic ADP receptor P2Y12 on platelets. Upon ADP binding, Gi-coupled P2Y12 activates the PI3K/AKT pathway and inhibits adenylyl cyclase, leading to reduced intracellular cAMP levels. This results in VASP dephosphorylation, increased cytosolic Ca2+ release, and platelet activation with fibrinogen binding to integrin αIIbβ3, promoting aggregation and thrombus formation. When LRP5 is inhibited, P2Y12 signaling is attenuated, leading to higher cAMP levels, increased VASP phosphorylation, and suppression of Ca2+ mobilization. Consequently, platelet activation and aggregation are reduced, thereby decreasing thrombus formation without significantly increasing bleeding risk.

|
Scooped by
Beeyond
April 23, 8:59 AM
|
A l'ouverture du groupe des assises du médicament consacré au dispositif médical, j'osais évoquer la possibilité que la prochaine affaire Mediator puisse impliquer un dispositif médical (DM). Je ne m'attarderai pas sur les fermes dénégations qui m'avaient alors été opposées tant par les représentants des industriels que par ceux de l'Afssaps. Le marquage CE allié à la matério-vigilance étaient là pour protéger nos patients… nous pouvions être tranquilles…

|
Scooped by
Beeyond
April 22, 3:30 AM
|
The zwitterionic PCBDA formed a hydration layer once it contacted blood, preventing the non–specific adsorption of serum proteins, which further blocked subsequent coagulation and inflammation. The anti–protein adsorption tests showed that the PCBDA/PDA–PEI coating resisted the non–specific adsorption of fibrinogen, which was a positive signal for enhanced anti–coagulation. Moreover, the platelet adhesion/activation tests confirmed its antithrombotic properties. The in vivo tissue response was also greatly suppressed due to the inhibited recognition of PCBDA/PDA–PEI–coated implants by macrophages. Interestingly, unlike traditional zwitterionic polymers, due to the introduction of adhesive catechol moieties, PCBDA supported the growth of endothelial cells, demonstrating its ability to selectively direct the fate of endothelial cells and smooth muscle cells.

|
Scooped by
Beeyond
April 20, 10:06 AM
|
Key, 12-month findings demonstrated that most treated AV fistulas were brachiocephalic (51.6%), followed by brachiobasilic (20.8%) and radiocephalic (20.8%); the most common lesion locations were the cephalic arch (25.8%) and venous outflow (25.2%), and lesions were de novo (59.5%) or non-stented restenotic (40.5%).
Target lesion primary patency at 12 months was 70.2% and access circuit primary patency at 12 months was 52.6%. Target lesion and access circuit patency rates were aligned with the 12-month outcomes observed for the IN.PACT AV DCB in the pivotal randomised trial (65.3%, 55.1%, respectively) and better than percutaneous transluminal angioplasty (PTA) rates (46.3%, 35%, respectively).
Fewer than one reintervention per patient was required through 12 months, which is below the average of 1.5 reintervention procedures per year. The serious infection rate at 12 months was 13.4%, below literature reports ranging from 19–23%

|
Scooped by
Beeyond
April 18, 5:24 AM
|
Drug-coated balloon (DCB) angioplasty provides an alternative to drug-eluting stents and plain old balloon angioplasty for the treatment of coronary or peripheral artery disease.
The absence of a metallic scaffold with DCB angioplasty compared with stenting might confer a biomechanical and physiological benefit and avoids the implantation of additional stent layers in patients with in-stent restenosis.
A class effect cannot be assumed for DCBs; device manufacturers are challenged with finding the best combination of antiproliferative agent and excipient to achieve optimal clinical and angiographic outcomes.

|
Scooped by
Beeyond
April 18, 4:20 AM
|
Coronary artery anomalies (CAAs) are a group of congenital conditions characterized by abnormal origin or course of any of the 3 main epicardial coronary arteries. Although CAAs have been identified as a common underlying condition in young athletes with sudden cardiac death, the widespread use of invasive and noninvasive coronary imaging has led to increased recognition of CAAs among adults. CAAS are often discovered as an incidental finding during the diagnostic workup for ischemic heart disease. The clinical correlates and prognostic implication of CAAs remain poorly understood in this context, and guideline-recommended therapeutic choices are supported by a low level of scientific evidence. Several studies have examined whether assessment of CAA-related myocardial ischemia can improve risk stratification in these patients, suggesting that multimodality imaging and functional tests may be key in the management of CAAs. The aim of this review is to outline definitions, classification, and epidemiology of the most relevant CAAs, highlighting recent advances and the potential impact of multimodality evaluation, and to discuss current therapeutic opportunities.
|

Curated by Beeyond
BEEYOND is a consulting company in the field of disruptive innovation, accompanying established companies on out-of-the-core growth strategy, from creation of new concepts to product launch. Reach us at: contact@beeyond.fr.
|




 Your new post is loading...
Your new post is loading...