A phase I-II study of niacin in patients with newly diagnosed glioblastoma: safety and interim phase II analysis
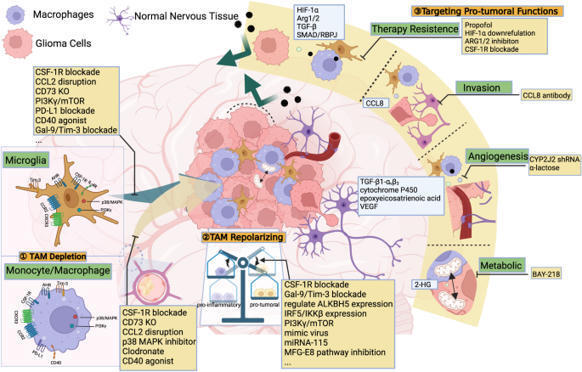
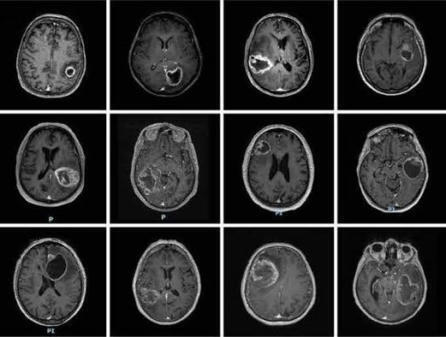
Survival of patients with glioblastoma (GB) treated with standard of care (SOC) surgery, radiotherapy, and temozolomide is 15 months with progression free survival at 6 months (PFS-6 M) of 53.9%. In vivo studies showed increased survival in mice with GB treated with niacin. This is a first in human Phase I-II study aiming to evaluate safety and efficacy of controlled-release niacin (NiacinCRT ™) added to SOC.
 Your new post is loading...
Your new post is loading...