Your new post is loading...

|
Scooped by
Beeyond
April 26, 2:54 PM
|
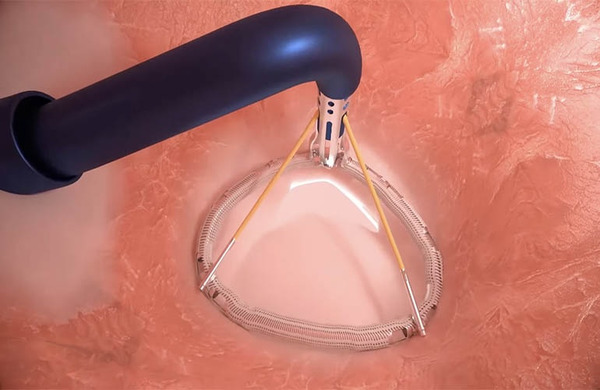
The initial commercial availability of the HighLife TMVR system across Europe provides access to a transfemoral mitral valve replacement option featuring a dual-component valve-in-ring design, capable of treating the broadest range of native mitral annulus sizes (30–53mm) of any CE mark–approved TMVR system.
Clinical experience with the HighLife TMVR system includes patients treated across multiple countries, with longer-term follow-up data demonstrating durable outcomes, including sustained reduction of mitral regurgitation to mild or less; durable annular sealing enabled by a sub-annular ring fixation design, with no reported peri- or post-procedural paravalvular leakage PVL closure.

|
Scooped by
Beeyond
April 26, 2:53 PM
|
A new multicenter study published in The Annals of Thoracic Surgery finds that reoperative surgical mitral valve replacement (rSMVR) is associated with significantly better long-term survival compared to transcatheter mitral valve-in-valve (mViV) procedures in patients with failing bioprosthetic mitral valves. While both approaches demonstrated similar safety and procedural success at 30 days, key differences emerged over time, particularly beyond the first year.

|
Scooped by
Beeyond
April 26, 2:52 PM
|
Presenting the latest results during a late-breaking trials session at PCR London Valves (16–18 November, London, UK), Vlasis Ninios (Interbalkan Medical Center, Thessaloniki, Greece) reported that, in a population of patients including many who would have been ineligible to receive other mitral valve technologies, implanters achieved a high rate of technical success with the procedure, resulting in symptomatic improvements for patients.
Left ventricular outflow tract (LVOT) obstruction is a common limitation of existing TMVR technologies leading to high rates of screen failure for patients screened for TMVR trials, Ninios highlighted in his presentation, ranking this alongside mitral annulus size and the presence of mitral annular calcification (MAC) as the exclusions that comprise the “Achilles’ heel” of existing TMVR technologies. AltaValve System’s atrial fixation TMVR device is designed to minimise the risk of LVOT obstruction and treat a broad population of mitral regurgitation (MR) patients as well as varied mitral annulus sizes.

|
Scooped by
Beeyond
April 26, 2:35 PM
|
Tricuspid regurgitation occurs when the tricuspid valve does not close properly, allowing blood to flow backward in the heart. The condition is common in older adults and can lead to worsening heart failure symptoms, repeated hospitalization and increased risk of death.
Historically, open-heart surgery was often the only treatment option for this condition, but many older patients were not candidates because of age or other medical problems. Today, transcatheter tricuspid valve replacement offers a less invasive alternative in which doctors replace the damaged valve using a catheter, or tube, threaded through a blood vessel.

|
Scooped by
Beeyond
April 8, 4:48 PM
|
Prior to this, ProStyle M® had undergone a two-year First-in-Man (FIM) study involving a total of 10 patients with a mean age of 71 years. The study results demonstrated excellent anchoring performance with no regurgitation observed in all cases. As the clinical study progresses, further evidence will be generated to verify the product's safety and efficacy.
KingstronBio takes innovation as its core driving force and has long been deeply engaged in the heart valve field. The company owns a number of patented technologies at home and abroad, including the Micro-Ex™ anti calcification treatment technology and AirBo™ dry storage technology for heart valves.

|
Scooped by
Beeyond
April 8, 4:47 PM
|
Auparavant, ProStyle M® avait fait l'objet d'une étude First-in-Man (FIM) de deux ans portant sur un total de 10 patients dont l'âge moyen était de 71 ans. Les résultats de l'étude ont démontré une excellente performance d'ancrage, aucune régurgitation n'ayant été observée dans tous les cas. Au fur et à mesure que l'étude clinique progresse, des preuves supplémentaires seront produites pour vérifier l'innocuité et l'efficacité du produit.
KingstronBio fait de l'innovation son principal moteur et est engagée depuis longtemps dans le domaine des valves cardiaques. L'entreprise possède un certain nombre de technologies brevetées dans le pays et à l'étranger, notamment la technologie de traitement anti-calcification Micro-Ex™ et la technologie de stockage à sec AirBo™ pour les valves cardiaques.

|
Scooped by
Beeyond
April 6, 6:31 AM
|
In a clinical trial comparing repeat mitral valve replacement surgery with a transcatheter valve-in-valve approach for dysfunctional bioprosthetic mitral valves, results linked the less invasive technique with a lower rate for death or stroke after 1 year. SURViV, is the first prospective clinical trial to compare the two procedures head-to-head. “Transcatheter mitral valve-in-valve has emerged as a less-invasive therapy for select high-risk patients,” he said. “Yet until now no trial has directly compared these two strategies.” The trial enrolled 150 patients with mitral bioprosthetic valve dysfunction between February 2020 and November 2023 and randomized them to redo surgery or transcatheter mitral valve-in-valve. The procedures were done at seven referral centers in Brazil. The primary composite endpoint of all-cause death and disabling stroke at 1 year was 20.8% for the surgery group and 5.3% for those who underwent valve-in-valve repair (P = .005), Siqueira said.

|
Scooped by
Beeyond
March 30, 3:50 AM
|
Transcatheter mitral valve-in-valve (mVIV) has emerged as a minimally invasive alternative to redo mitral valve replacement surgery (rMVR) in patients who cannot undergo repeat open-heart surgery. The mVIV procedure involves using a long flexible tube called a catheter to place a new prosthetic valve inside the patient's poorly functioning one. Until now, however, the outcomes of mVIV and rMVR had not been compared in a randomized, controlled trial, Siqueira said.

|
Scooped by
Beeyond
February 16, 4:54 AM
|
The AMEND™ System delivers a closed, D-shaped, semi-rigid annuloplasty ring through catheter in a transseptal approach. It is specifically designed to mimic the anatomical remodeling and clinical results achieved by traditional surgical annuloplasty—the technique used in most open-heart mitral valve repairs. Unlike other transcatheter mitral repair technologies, the AMEND System is designed to preserve future treatment options if clinically required later.

|
Scooped by
Beeyond
December 29, 2025 7:58 AM
|
The Sapien M3 transcatheter mitral valve replacement (TMVR) system is indicated for the treatment of symptomatic moderate-to-severe or severe MR in patients who are deemed unsuitable for surgery or transcatheter edge-to-edge repair (TEER) therapy by a multidisciplinary heart team.

|
Scooped by
Beeyond
December 29, 2025 7:58 AM
|
Heart valve replacement is a common cardiac procedure, but many patients and families ask one key question: what is the average age for heart valve replacement?
In most cases, heart valve replacement is performed between 60 and 75 years of age, although the exact age varies depending on the valve involved, the underlying heart condition, and the patient’s overall health.

|
Scooped by
Beeyond
December 6, 2025 7:40 AM
|
Artificial intelligence combines ultrasound and X-ray images to give physicians a real-time 3D view of the repair device during transcatheter heart procedures.

|
Scooped by
Beeyond
December 6, 2025 7:39 AM
|
The device, which can be deployed for transcatheter edge-to-edge repair (TEER) in the mitral or tricuspid valves, has similar design features to existing mitral TEER devices including MitraClip (Abbott) and Pascal (Edwards Lifesciences), and includes a mechanical closure system and central spacer. The device has received approval from China’s National Medical Products Administration (NMPA) and CE mark to date.

|
Scooped by
Beeyond
December 6, 2025 7:38 AM
|
The study, which began enrolling in 2015, charts early use of the Intrepid device featuring a transapical delivery system, though later generations of the device have switched to using a transfemoral approach. Procedures were conducted at 21 sites spanning Europe, Australia and the USA and involved 95 patients with symptomatic severe MR who were at high risk for surgery.

|
Scooped by
Beeyond
December 6, 2025 7:38 AM
|
Left ventricular outflow tract (LVOT) obstruction is a common limitation of existing TMVR technologies leading to high rates of screen failure for patients screened for TMVR trials, Ninios highlighted in his presentation, ranking this alongside mitral annulus size and the presence of mitral annular calcification (MAC) as the exclusions that comprise the “Achilles’ heel” of existing TMVR technologies. AltaValve System’s atrial fixation TMVR device is designed to minimise the risk of LVOT obstruction and treat a broad population of mitral regurgitation (MR) patients as well as varied mitral annulus sizes.

|
Scooped by
Beeyond
November 30, 2025 4:14 PM
|
Over the past five years, we observed a 7.24% mortality rate at our tertiary care facility following prosthetic heart valve implantation across all age groups. The data suggest that mortality may be more common among females and older individuals; however, these differences did not reach statistical significance.

|
Scooped by
Beeyond
November 24, 2025 5:53 AM
|
Minimally invasive cardiac procedures such as transcatheter aortic valve replacement (TAVR), catheter-based mitral valve repair, and percutaneous atrial septal defect closure fail at times to attract the same attention given to traditional open-heart surgeries, particularly high-profile procedures such as a coronary artery bypass graft. Studies like this one from the March 2021 issue of the Journal of Thoracic Disease discuss the debate over outcomes, quality, and safety that has followed these procedures.

|
Scooped by
Beeyond
November 17, 2025 9:07 AM
|
Results of a pivotal clinical trial evaluating the safety and efficacy of a fully percutaneous transseptal mitral valve replacement (TMVR) procedure—Sapien M3 (Edwards Lifesciences)—in patients with symptomatic, moderate-to-severe mitral regurgitation (MR) who are not eligible for surgery or mitral-transcatheter edge-to-edge repair (M-TEER) procedures, demonstrated effective MR reduction with low rates of complications and mortality.

|
Scooped by
Beeyond
November 3, 2025 4:22 AM
|
Results from a pivotal clinical trial to evaluate the safety and efficacy of a fully percutaneous transseptal mitral valve replacement (TMVR) procedure in patients with symptomatic, moderate-to-severe mitral regurgitation (MR) who are not candidates for conventional surgery or transcatheter edge-to-edge repair (TEER) procedures, demonstrated effective MR reduction with low rates of complications and mortality.

|
Scooped by
Beeyond
November 3, 2025 4:21 AM
|
In two forms of severe mitral valve disease with high rates of mortality and limited treatment options, multicenter trials with novel transcatheter devices produced what experts described as unprecedented symptom control and rates of survival. The two trials — one directed at severe mitral annular calcifications (MACs) and the other at inoperable severe mitral regurgitation — were conducted without a comparator arm because no meaningful benefit could be expected from alternatives, according to the principal investigators of each trial.

|
Scooped by
Beeyond
November 3, 2025 4:19 AM
|
A moment ten years ago struck me hard and has stayed with me ever since. A nineteen-year-old girl had been diagnosed with rheumatic mitral valve disease. Her family was poor, and a friend brought me her scans. I knew I could save her valve and protect her future. No lifelong anticoagulants. No cold ticking in her chest, like a metronome counting down her days. No fear that a stumble on the stairs might turn into a medical emergency. I told her: make the journey at your own expense, and I will perform the surgery for free. But before I could schedule it, another hospital stepped in. They promised to cover all her hospitalization costs, and then replaced her valve with a mechanical one. She survived. But now, she will live the rest of her life with a foreign device inside her, bearing all the risks and sacrifices that come with it.

|
Scooped by
Beeyond
November 1, 2025 12:37 PM
|
Protembis was founded in 2013 and is headquartered in Aachen, Germany. The company develops innovative technologies to reduce the risk of new brain injury during cardiology procedures particularly in connection with transcatheter aortic valve replacement (TAVR).

|
Scooped by
Beeyond
September 14, 2025 12:21 AM
|
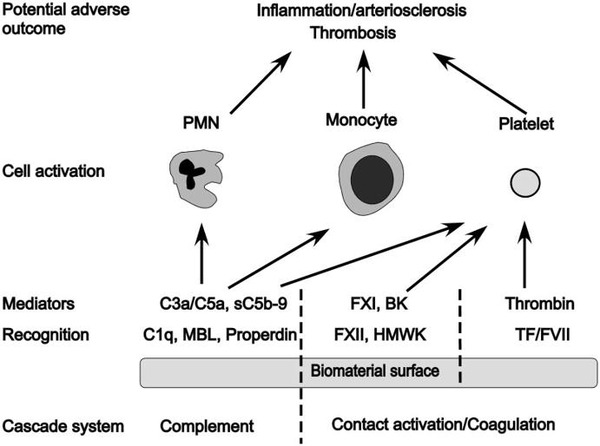
Overall, considerably more success has been achieved in reducing the thrombogenicity of bio-artificial surfaces than in controlling complement activation: For example, surfaces coated with different forms of heparin or PEG are associated with low or negligible activation of coagulation and subsequent platelet loss. Thus, there are numerous surfaces that have low thrombogenicity available that still bear substantial complement-activating capacity. Some of these materials will no doubt be a

|
Scooped by
Beeyond
September 13, 2025 11:52 PM
|
A new-look polymer heart valve is associated with encouraging one-year outcomes in patients undergoing surgical mitral valve replacement (SMVR), according to new data presented at New York Valves 2025 and published in the Journal of the American College of Cardiology.[1]
The Tria mitral valve from Utah-based Foldax is built using LifePolymer, a proprietary material that does not include any animal tissue. Both the frame of the valve and its leaflets are robotically generated to match the patient’s native mitral valve.

|
Scooped by
Beeyond
September 13, 2025 11:51 PM
|
Prosthetic heart valves are classified as Class III medical devices by the FDA; therefore, they require the highest level of regulatory examination before approval for the market by the competent authorities (e.g., the FDA in the US, notified bodies in the EU). For this purpose, the biggest challenge for PHVs is the successful demonstration of long-term durability in vivo: 10–15 years (or more) can be a reasonable timeframe for approval. Moreover, any new prosthetic device has to exhibit better performance compared to the existing ones and, in the specific case of PHVs, the mechanism of failure has to be precisely addressed. All these issues are particularly challenging!
|

Curated by Beeyond
BEEYOND is a consulting company in the field of disruptive innovation, accompanying established companies on out-of-the-core growth strategy, from creation of new concepts to product launch. Reach us at: contact@beeyond.fr.
|



 Your new post is loading...
Your new post is loading...