Chimeric antigen receptor (CAR) T cells and bispecific T cell engagers have become integral components in the treatment of relapsed/refractory multiple myeloma. We report a 63-year-old male who received ciltacabtagene autoleucel CAR T cells and the GPRC5D × CD3 bispecific talquetamab for early relapse of his multiple myeloma. Nine months after CAR T therapy, he developed a symptomatic leukemic peripheral T cell lymphoma with cutaneous and intestinal involvement. Longitudinal single-cell RNA and T cell receptor sequencing of peripheral blood and bone marrow revealed two hyperexpanded CAR-carrying T cell clones. These expanded clones exhibited an exhausted effector-memory T cell transcriptional signature, and the neoplasm itself was sensitive to dexamethasone treatment. The immunophenotypic and transcriptional alterations of these abnormal T cells resembled those of T-large granular lymphocytic leukemia. Spatial transcriptomes of skin lesions confirmed the aberrant CAR-expressing T cells. Whole-genome sequencing revealed three distinct integration sites, within the introns of ZGPAT, KPNA4 and polycomb-associated noncoding RNAs. Before and after CAR T whole-genome analyses implicated clonal outgrowth of a TET2-mutated precursor propelled by additional subclone-specific loss of heterozygosity and other secondary mechanisms. This case highlights the evolution of a CAR-carrying peripheral T cell lymphoma following CAR T cell and bispecific T cell engager therapy, offering critical insights into the clonal evolution from a predisposed hematopoietic precursor to a mature neoplasm. A longitudinal multiomics analysis of a patient with multiple myeloma who developed peripheral T cell lymphoma after treatment with anti-BCMA CAR T cells and a GPRC5D-directed bispecific antibody reveals that two mutated CAR+CD8+ T cell clones were probably drivers of the neoplasm.

|
Scooped by
BigField GEG Tech
onto Genetic Engineering Publications - GEG Tech top picks April 3, 2025 7:10 AM
|




 Your new post is loading...
Your new post is loading...



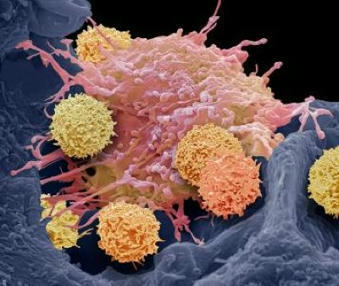


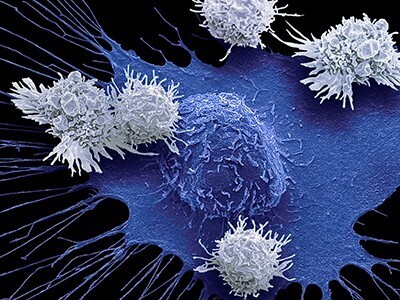












CAR-T cell therapy has revolutionized cancer treatment by engineering a patient’s immune cells to target and destroy malignant cells using a chimeric antigen receptor (CAR). However, emerging evidence suggests potential risks associated with this approach. A recent case study describes a 63-year-old patient with multiple myeloma who developed T-cell lymphoma nine months after receiving CAR-T cell therapy. Genetic analyses revealed that the lymphoma originated from the infused CAR-T cells, with pre-existing mutations in the patient's hematopoietic cells contributing to malignant transformation. This case highlights the need for rigorous genetic screening before CAR-T therapy and long-term monitoring to detect rare but serious complications. While CAR-T therapy remains a breakthrough in oncology, this finding underscores the importance of ongoing safety evaluations