Neuromodulation technologies are crucial for investigating neuronal connectivity and brain function. Magnetic neuromodulation offers wireless and remote deep brain stimulations that are lacking in optogenetic- and wired-electrode-based tools. However, due to the limited understanding of working principles and poorly designed magnetic operating systems, earlier magnetic approaches have yet to be utilized. Furthermore, despite its importance in neuroscience research, cell-type-specific magnetic neuromodulation has remained elusive. Here we present a nanomaterials-based magnetogenetic toolbox, in conjunction with Cre-loxP technology, to selectively activate genetically encoded Piezo1 ion channels in targeted neuronal populations via torque generated by the nanomagnetic actuators in vitro and in vivo. We demonstrate this cell-type-targeting magnetic approach for remote and spatiotemporal precise control of deep brain neural activity in multiple behavioural models, such as bidirectional feeding control, long-term neuromodulation for weight control in obese mice and wireless modulation of social behaviours in multiple mice in the same physical space. Our study demonstrates the potential of cell-type-specific magnetogenetics as an effective and reliable research tool for life sciences, especially in wireless, long-term and freely behaving animals. Minimally invasive cellular-level target-specific neuromodulation is needed to decipher brain function and neural circuitry. Here nano-magnetogenetics using magnetic force actuating nanoparticles has been reported, enabling wireless and remote stimulation of targeted deep brain neurons in freely behaving animals.






 Your new post is loading...
Your new post is loading...





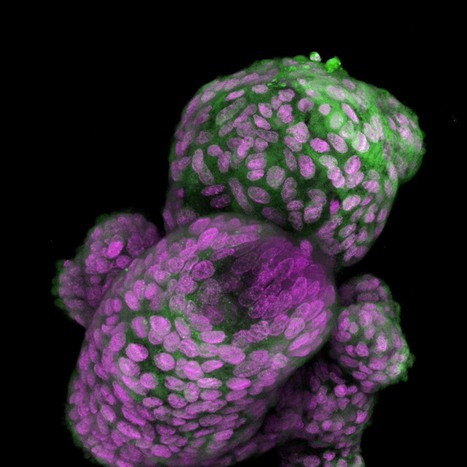

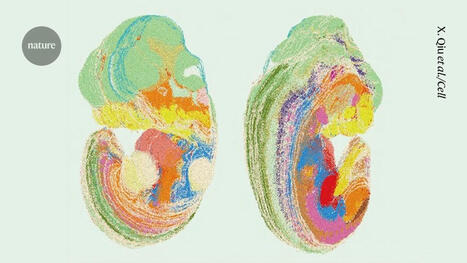

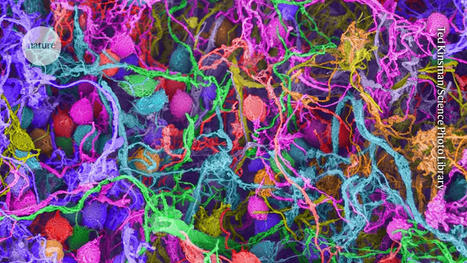







Brain metastases from breast cancer develop in patients with metastatic breast cancer. Current treatment options for brain metastases include surgery, radiation, chemotherapy and targeted therapies, but these have limited success and may worsen neurological function. Because 80% of women with brain metastases from breast cancer die within a year of diagnosis, Cittelly and her team want to find a way to target cancer cells after they have spread to the brain.
Working with cells in the lab, they have identified the interleukin 13 receptor alpha 2 (IL13Ra2) as a likely target for treatment. This is a protein that is found at increased levels in cancer cells that metastasize to other locations in the body, particularly the brain and lungs. In addition, the protein has shown vulnerability to treatment with CAR T cells in clinical trials of brain tumors. The researchers' next step is to initiate a collaboration with CAR T cell experts to better understand how CAR T cell therapy might target IL13Ra2.
Ultimately, Cittelly hopes to see clinical trials for patients with breast cancer that has metastasized to the brain, as they are currently excluded from clinical trials.