Filamentous pathogens such as the oomycete Phytophthora infestans infect plants by developing specialized structures termed haustoria inside the host cells. Haustoria are thought to enable secretion of effector proteins into the plant cells. Haustorium biogenesis is therefore critical for pathogen accommodation in the host tissue. Haustoria are enveloped by a specialized host-derived membrane, the extrahaustorial membrane (EHM), which is distinct from the plant plasma membrane. The mechanisms underlying the biogenesis of the EHM are unknown. Remarkably, several plasma membrane localised proteins are excluded from the EHM but the remorin REM1.3 accumulates around P. infestans haustoria. Here, we used overexpression, co-localization with reporter proteins, and super-resolution microscopy in cells infected by P. infestans to reveal discrete EHM domains labelled by REM1.3 and P. infestans effector AVRblb2. Moreover, SYT1 synaptotagmin, another previously identified perihaustorial protein, localized to subdomains which are mainly not labelled by REM1.3 and AVRblb2. Functional characterization of REM1.3 revealed that it is a susceptibility factor that promotes infection by P. infestans. This activity, and REM1.3 recruitment to the EHM, require REM1.3 membrane binding domain. Our results implicate REM1.3 membrane micro-domains in plant susceptibility to an oomycete pathogen.
Via
Kamoun Lab @ TSL
 Your new post is loading...
Your new post is loading...
 Your new post is loading...
Your new post is loading...
























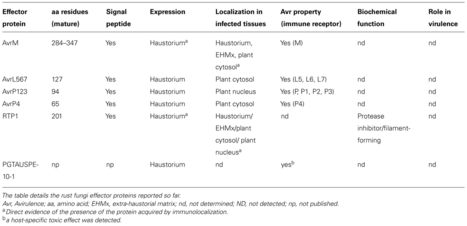









Motivation: Single Nucleotide Polymorphism (SNP) discovery is an important preliminary for understanding genetic variation. With current sequencing methods we can sample genomes comprehensively. SNPs are found by aligning sequence reads against longer assembled references. De Bruijn graphs are efficient data structures that can deal with the vast amount of data from modern technologies. Recent work has shown that the topology of these graphs captures enough information to allow the detection and characterisation of genetic variants, offering an alternative to alignment-based methods. Such methods rely on depth-first walks of the graph to identify closing bifurcations. These methods are conservative or generate many false-positive results, particularly when traversing highly inter-connected (complex) regions of the graph or in regions of very high coverage.
Results: We devised an algorithm that calls SNPs in converted De Bruijn graphs by enumerating 2k + 2 cycles. We evaluated the accuracy of predicted SNPs by comparison with SNP lists from alignment based methods. We tested accuracy of the SNP calling using sequence data from sixteen ecotypes of Arabidopsis thaliana and found that accuracy was high. We found that SNP calling was even across the genome and genomic feature types. Using sequence based attributes of the graph to train a decision tree allowed us to increase accuracy of SNP calls further. Together these results indicate that our algorithm is capable of finding SNPs accurately in complex sub-graphs and potentially comprehensively from whole genome graphs.