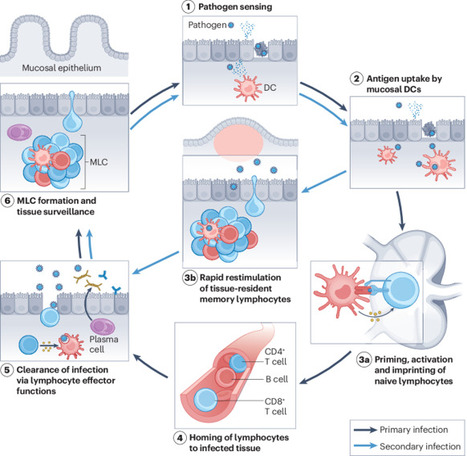
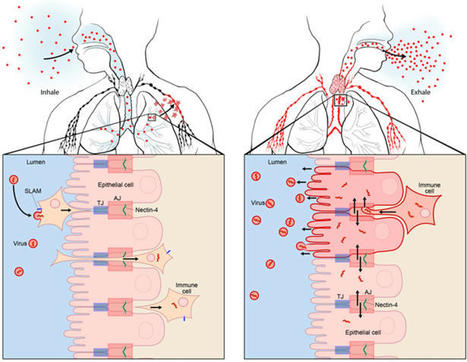
Measles VirusMeasles virus (MV) binds to cell surface receptors by way of its hemagglutinin (H) protein to initiate infection.From: xPharm: The Comprehensive Pharmacology Reference, 2007Related terms:Subacute Sclerosing PanencephalitisVaccine EfficacyCD46Monospecific AntibodyParamyxoviridaeImmunityWild TypeHuman Immunodeficiency VirusView all TopicsMeasles VirusR. Cattaneo, M. McChesney, in Encyclopedia of Virology (Third Edition), 2008Measles virus (MV), one of the most contagious viruses known, was recognized clinically by the rash and other signs from early historical times. Measles still causes more than 300 000 deaths each year, mostly due to secondary infections facilitated by MV-induced immunosuppression. Therefore the World Health Organization has targeted it for eradication, an endeavor facilitated by the availability of a live attenuated vaccine with an outstanding efficacy and safety record. MV is an enveloped negative-strand RNA virus, and the study of its biology has given detailed insights about how it utilizes host cell components to promote its replication and particle assembly. Moreover, the MV interactions with the cellular receptors and the mechanisms used to invade and inactivate the host immune system are now better understood. The knowledge gained from basic research is currently used to develop multivalent MV-based vaccines, and vectors for targeting and eliminating cancer cells.View chapterPurchase bookRead full chapterURL: https://www.sciencedirect.com/science/article/pii/B978012374410400443XDevelopmental Aspects of Pagetic OsteoclastsDeborah L. Galson, ... G. David Roodman, in Advances in Pathobiology and Management of Paget's Disease of Bone, 2016MVNP Downregulation of FoxO3 and Sirt1 also Increase IL-6MVNP also was demonstrated to increase NFκB activity by downregulating expression of Sirtuin 1 (Sirt1), a class III protein deacetylase that targets acetylated NFκB and negatively regulates its activity [31]. MVNP decreases Sirt1 by triggering increased phosphorylation of Forkhead-box class O3 (FoxO3), resulting in decreased FoxO3 protein stability and decreased transcription of its target gene Sirt1 in OCL precursors and NIH3T3 cells. Several protein kinases have been reported to downregulate FoxO3 stability through phosphorylation, including AKT, ERK1/2, IKKβ, and IKKε [50–52]. It’s not yet known which, if any, of these are triggered by MVNP to phosphorylate FoxO3. However, TBK1 overexpression in TRAP-TBK1 BMM was sufficient to decrease Sirt1 mRNA (unpublished data), suggesting that activated TBK1 may phosphorylate FoxO3. Wang et al. [31] showed that NIH3T3 cells stably transduced with MVNP (MVNP-NIH3T3) demonstrated higher IL-6 promoter luciferase reporter activity than NIH3T3 cells transduced with empty vector (EV-NIH3T3), and ectopic expression of Sirt1 significantly decreased both the basal and MVNP-stimulated IL-6 promoter activity. Further, resveratrol, a Sirt1 gene activator, suppressed the high level of IL-6 mRNA in MVNP-NIH3T3 cells. Significantly, resveratrol inhibited OCL differentiation of BMM from both wild-type and MVNP mice. Strikingly, at a resveratrol dose that had little effect on wild-type OCL differentiation, the enhanced MVNP OCL differentiation was suppressed to wild-type levels. Higher resveratrol doses then suppressed wild-type and MVNP OCL differentiation to similar levels. Hence, MVNP acts via two pathways to increase IL-6 expression (Fig. 4.2).View chapterPurchase bookRead full chapterURL: https://www.sciencedirect.com/science/article/pii/B978012805083500004XNeurovirologyJane E. Libbey, Robert S. Fujinami, in Handbook of Clinical Neurology, 2014Measles virusMeasles virus (Chapter 27) is a Morbillivirus, of the family Paramyxoviridae, and is an enveloped single-stranded negative-sense RNA virus (Nathanson and Gonzalez-Scarano, 2007). Measles virus is spread by the respiratory route (Nathanson and Gonzalez-Scarano, 2007). One neurologic complication of measles virus infection, thought to be related to dysregulation of the cellular immune responses, as it can occur in the absence of viral replication in the CNS, is postinfectious encephalomyelitis, which develops within weeks of infection (Johnson et al., 1983, 1984; Hirsch et al., 1984; Johnson, 1987). However, measles virus can infect and persist in neurons (Parra et al., 1999; Ramakrishna et al., 2002). In immunocompetent individuals a CD4 + T-cell population mediates elimination of measles virus from neurons, possibly through secretion of IFN-γ (Parra et al., 1999; Schneider-Schaulies et al., 2003). Failure of the immune response to eliminate measles virus-infected cells completely from the CNS can result in viral persistence (Schneider-Schaulies et al., 1999).Measles virus is capable of persisting in neurons as a defective variant that spreads from neuron to neuron directly, without passage through the extracellular environment (Schneider-Schaulies et al., 1999, 2003; Lawrence et al., 2000; Nathanson and Gonzalez-Scarano, 2007). In this way the virus avoids detection and elimination by circulating high titers of measles virus-specific neutralizing antibody. The persistence of measles virus in the CNS can result in a progressive fatal encephalitis called SSPE developing in immunocompetent individuals several months to years after infection and recovery from acute measles virus infection (Schneider-Schaulies et al., 1999, 2003; Nathanson and Gonzalez-Scarano, 2007). As such, SSPE is primarily a disease of childhood and young adulthood (Gilden, 1983; Wolinsky, 1990).As described above, oligoclonal IgG antibody bands, with measles virus-restricted specificities, are characteristically found in the serum and CSF of patients with SSPE (Mehta et al., 1994; Burgoon et al., 2006; Tschen et al., 2006). These oligoclonal bands are a hyperimmune response to measles virus antigens, which, despite their neutralizing activity, are unable to control the viral infection (Mehta et al., 1994). This inability of measles virus-specific antibodies to control measles virus infection in SSPE patients may result from antibody-induced antigenic modulation by the virus (Fujinami and Oldstone, 1980, 1983; Fujinami et al., 1984). The expression of some measles virus antigens within and on the surface of measles virus-infected cells is altered upon binding of measles virus-specific antibodies such that the synthesis, assembly, and maturation of the virions are altered. In this way the measles virus-infected cells may avoid detection and lysis and measles virus may persist. Thus, in the case of measles virus, the antibody arm of the adaptive immune response to the virus may play a role in the initiation of viral persistence (Fujinami and Oldstone, 1980, 1983; Fujinami et al., 1984).View chapterPurchase bookRead full chapterURL: https://www.sciencedirect.com/science/article/pii/B9780444534880000109MeaslesWilliam J. Moss, in Tropical Dermatology (Second Edition), 2017Natural History, Pathogenesis, and PathologyMeasles virus is primarily transmitted by respiratory droplets over short distances and, less commonly, by small-particle aerosols that remain suspended in the air for long periods of time. The time from infection to clinical disease is approximately 10 days to the onset of fever and 14 days to the onset of rash. Persons with measles are infectious for several days before and after the onset of rash, when levels of measles virus in blood and body fluids are highest and when the symptoms of cough, coryza, and sneezing are most severe. The host immune response at sites of virus replication is responsible for the signs and symptoms of measles.Host immune responses to measles virus are essential for viral clearance, clinical recovery, and the establishment of long-term immunity.7 The protective efficacy of antibodies to measles virus is illustrated by the immunity conferred to infants from passively acquired maternal antibodies and the protection of exposed, susceptible individuals following administration of anti-measles-virus immunoglobulin. The duration of protective immunity following wild-type measles virus infection is generally lifelong.8 The immune responses to measles virus infection are associated with depressed responses to unrelated (non-measles-virus) antigens lasting for several weeks to months. This state of immune suppression enhances susceptibility to secondary bacterial and viral infections causing pneumonia and diarrhea, and is likely responsible for much of measles-associated morbidity and mortality.9 Vitamin A deficiency is a recognized risk factor for severe measles. The vitamin is essential for the maintenance of normal epithelial tissues throughout the body; measles virus itself infects and damages these tissues.View chapterPurchase bookRead full chapterURL: https://www.sciencedirect.com/science/article/pii/B9780323296342000146Measles Virus (Rubeola)Anne A. Gershon, in Mandell, Douglas, and Bennett's Principles and Practice of Infectious Diseases (Eighth Edition), 2015Growth of Measles Virus in Tissue CultureMeasles virus was first successfully isolated in the laboratory by Enders and Peebles in 1954.18 The virus was initially propagated in primary human renal cells but later was cultivated in cultured simian kidney cells. Wild-type measles virus is rather difficult to propagate in vitro because it is slow growing, and only a limited number of types of cell cultures are permissive for the virus.16 Typically, cytopathic effects produced by measles virus in tissue cultures consist of stellate cells with increased refractility and, especially on passage, multinucleated syncytial giant cells containing intranuclear inclusions. In the absence of cytopathic effects, virus replication can also be detected by hemadsorption of rhesus monkey erythrocytes. Presumptive isolates of measles virus are identified by typing with monoclonal antibodies by using immunofluorescence or plaque reduction tests.3,19 Reverse-transcriptase polymerase chain reaction (RT-PCR) assays for measles virus are also available (see later).View chapterPurchase bookRead full chapterURL: https://www.sciencedirect.com/science/article/pii/B9781455748013001624MeaslesAlpay Azap, Filiz Pehlivanoglu, in Emerging Infectious Diseases, 20142 What is the Causative Agent? (Taxonomy and Description of the Agent)Measles virus (MV) is a member of the genus Morbillivirus of the family Paramyxoviridea.4 It is an enveloped, non-segmented, single-stranded, negative-sense RNA virus. Measles virions are seen as pleomorphic spheres with a diameter of 100–250 nm on electron microscopy. Measles virus encodes at least eight structural proteins, which have letter names: F, C, H, L, M, N, P, and V. Of these proteins, H (hemagglutinin) has a role in the attachment of the virus to host cells, and F (fusion) is involved in the spread of the virus from one cell to another.4Measles virus is closely related to the viruses causing diseases in animals such as canine and phocine distemper and rinderpest viruses. It is stated that MV adapted to humans when humans first began to domesticate animals in Mesopotamia around 3000 BC.1 Today, wild MV is pathogenic only for primates.View chapterPurchase bookRead full chapterURL: https://www.sciencedirect.com/science/article/pii/B9780124169753000261Infections that cause secondary immune deficiencyVincent Robert Bonagura, David Walter Rosenthal, in Stiehm's Immune Deficiencies (Second Edition), 2020Measles virus: temporary immunosuppressionMeasles virus (MV) continues to cause child morbidity and mortality worldwide, despite the availability and use of an effective live attenuated measles vaccine.10–14 Part of the reason why control of MV continues to be elusive is that it is highly contagious for susceptible individuals and there are difficulties with vaccine delivery. MV infection begins in the respiratory tract, spreads systemically in lymphoid, epithelial and endothelial cells, and ultimately infects multiple organs,15 causing a characteristic fever, rash, and conjunctivitis 10–14 days after respiratory infection (Fig. 49.3A and B). High fever, rhinorrhea and conjunctivitis typically precede the rash and the rash migrates from the head and neck to the hands and feet over 3–4 days. Many of these manifestations are caused by the immune response made to MV, and commonly this response clears MV in infected tissues and prevents re-infection for life (Fig. 49.3C and D). However, MV infection can cause several weeks of immune suppression after resolution in select individuals. This is the primary cause of measles-associated deaths: MV-induced secondary infection.16 Although vaccination against measles is very high in the United States, 92.7% of children aged 19–35 months were vaccinated in 2017,17 there are pockets of unvaccinated people who are susceptible to local outbreaks of measles. Since “herd immunity” is primarily effective when the vaccination rate is around 96%,18 vaccination levels below this level leave children and adults at risk for primary MV infection and secondary microbial infections.Measles was the first virus clearly identified to cause increased susceptibility to other microbial secondary infections. Most often, measles-associated deaths are caused by severe, overwhelming pneumonia and diarrhea.16 Suppression of delayed hypersensitivity has been identified in tuberculin-sensitized individuals many weeks after complete resolution of MV infection (Fig. 49.3C).19 Furthermore, several weeks after successful MV recovery, increased susceptibility to other infections has been reported, and T cell function and in vitro proliferation of T cells in response to mitogens has been shown to be markedly decreased (Fig. 49.4A and B).1,20,21 Immunosuppression occurs during a period of intense immune activation that occurs during the onset of the MV rash and anti-MV immune responses (Fig. 49.3C and D). Lymphopenia, skewing of Th2-like chemokine polarized responses, and suppression of lymphocyte proliferation have also been documented (Fig. 49.3D). MV infection causes decreases in T and B cells in the blood during the MV rash period.22–25 Altered trafficking and increased apoptosis of MV-infected and uninfected lymphocytes contribute to the development of lymphopenia.22,26–30 While lymphocyte numbers rapidly return to normal in the blood after the rash resolves, immunologic abnormalities persist.21,22,31,32 Immune suppression, Th2 cytokine polarization of CD4+ T cells, and Treg induction have been associated with indirect immunosuppression caused by MV infection.33,34 MV infection is also associated with suppression of IL-12 expression, lymphocyte CD30 expression, and IL-4, IL-10, and IL-13 expression after rash resolution.35–37 Reduction of IL-12 production reduces T cell expression of type I cytokines, particularly IFN-γ10,32 (Fig. 49.3D). It is possible that MV interacts with the complement regulatory molecule CD46 in polarizing Th2-like cytokine production, causing activation of signaling cascades that modify cell function, although this interaction is not firmly established.38,39 The MV-CD46 interaction may alter innate immunity by selectively downregulating receptor expression.40–46 This would increase susceptibility to complement-mediated lysis of MV-infected cells, and decrease antigen presenting cell production of IL-1247,48 and crosslinking of CD46 on T cells, leading to the induction of regulatory CD4+ T cells and enhanced IL-10 levels.49 These interactions would induce Th2-like polarization that would favor B cell maturation, provide lifelong MV antibody memory, and protect against MV re-infection. This polarization, however, would also depress APC activation and Th1-like responses to new pathogens.MV suppresses PBMC proliferation to mitogens after MV resolution, and this continues for several weeks (Fig. 49.4B).20,31 IL-2 supplementation can improve, but not fully restore, this responsiveness. This suggests that defective IL-2 expression is in part responsible for this proliferative defect.50 Cell cycle arrest in G1 after in vitro infection with MV is a recognized cause of hyporesponsiveness to mitogens.42,51–53 MV RNA can persist in PBMCs for months after MV resolution54,55 and may reduce mitogen proliferation, although this has not been established. The receptor used by wild-type MV to infect cells, CD150, is a dual function co-receptor for lymphocyte activation, and enhances IFN-γ expression.56–58 However, MV binding to CD150 can also downregulate receptor expression.59,60 T cell signaling through the MV glycoprotein complex of H and F1-F2 in the membranes of virions or MV-infected cells61–65 may also contribute to immunosuppression. This inhibitory signal prevents T cell S-phase entry for several days, and is independent of cell death, membrane fusion, soluble inhibitor production, or T cell infection.52,61,62,65–67 Thus, there is a delay in cell cycle progression and an accumulation of T cells in the G0/G1 phase.52,66,67 The mechanism by which H/F1-F2 suppresses mitogen-induced proliferation is unknown, but it is associated with MV-induced interference of T cell activation of phosphoinositide 3-kinase (PI3K) in T cells, or IL-2 receptor ligation.68 IL-2 added to MV-treated cells activates signal transducer and activator of transcription 3 (STAT3) but fails to activate Akt kinase, which is required for cell cycle progression.69 The modulatory effects of MV with glycoprotein complexes, and the downstream consequences of this interaction, have recently been summarized.10,68–70 While the relevance of these processes to the in vivo suppression of T cell lymphoproliferation remains to be identified. The combination of the established mechanisms leading to post-MV infection immunosuppression, and those that remain to be elucidated, cause, in select individuals, severe and on occasion, fatal secondary infection with other microbes.A key epidemiologic factor in measles-related deaths is vitamin A deficiency. In the developing world, the World Health Organization recommends vitamin A supplementation. Studies have demonstrated improved outcomes and suggest an effect on the mucosal barrier and also on improved T cell function though by an undefined mechanism.Read full chapterView PDFRead full chapterURL: https://www.sciencedirect.com/science/article/pii/B9780128167687000491MeaslesWilliam J. Moss, ... W. Harry Feinstone, in Vaccines for Biodefense and Emerging and Neglected Diseases, 2009Measles VirusMeasles virus is a spherical, nonsegmented, single-stranded, negative-sense RNA virus and a member of the Morbillivirus genus in the family of Paramyxoviridae. Other members of the Morbillivirus genus are rinderpest virus and canine distemper virus. Although RNA viruses have high mutation rates, measles virus is an antigenically monotypic virus, meaning that the surface proteins responsible for inducing protective immunity have retained their antigenic structure. The public health significance is that measles vaccines developed decades ago from a single measles virus strain remain protective worldwide. Measles virus is killed by ultraviolet light and heat. Attenuated measles vaccine viruses retain these characteristics, necessitating a cold chain for transportation and storage.The measles virus RNA genome consists of approximately 16,000 nucleotides and is enclosed in a lipid-containing envelope derived from the host cell. The genome encodes eight proteins, two of which (V and C) are nonstructural proteins and are transcribed from the phosphoprotein (P) gene. Of the six structural proteins, P, large protein (L), and nucleoprotein (N) form the nucleocapsid housing the viral RNA. The hemagglutinin protein (H), fusion protein (F), and matrix protein (M), together with lipids from the host cell membrane, form the viral envelope.The H protein interacts with F to mediate fusion of the viral envelope with the host cell membrane (Malvoisin and Wild, 1993). The primary function of the H protein is to bind to the host cellular receptors for measles virus. The two identified receptors are CD46 and CD150 (SLAM). CD46 is a complement regulatory molecule expressed on all nucleated cells in humans. SLAM, an acronym for signaling lymphocyte activation molecule, is expressed on activated T and B lymphocytes and antigen-presenting cells. The binding sites on H for these receptors overlap and strains of measles virus differ in the efficiency with which each is used. Wild-type measles virus binds to cells primarily through the cellular receptor SLAM whereas most vaccine strains bind to CD46; however, most measles virus strains can use both CD46 and SLAM as receptors during acute infection (Schneider et al., 2002). Additional, as yet unidentified receptors for measles virus exist on human endothelial and epithelial cells (Andres et al., 2003).Other measles virus proteins are involved in viral replication. The P protein regulates transcription, replication, and the efficiency with which the N assembles into nucleocapsids (Spehner et al., 1997). The M protein links ribonucleoproteins with envelope proteins during virion assembly. The functions of V and C proteins have not been clearly defined, but both appear to contribute to the virulence of measles virus by regulating transcription and sensitivity to the antiviral effects of interferon (IFN) α/β (Valsamakis et al., 1998; Patterson et al., 2000).Variability within the genome is sufficient to allow for molecular epidemiologic investigation. Genetic characterization of wild-type measles viruses is based on sequence analysis of the genes encoding the N and H proteins. One of the most variable regions of the measles virus genome is the 450-nucleotide sequence at the carboxy-terminal of the N protein, with up to 12% variability between wild-type viruses. The World Health Organization (WHO) recognizes 8 clades of measles virus (designated A through H) and 23 genotypes (World Health Organization, 2006). New genotypes likely will be identified with enhanced surveillance and molecular characterization. As measles control efforts intensify, molecular surveillance of circulating measles virus strains can be used to document interruption of measles virus transmission and to identify the source and transmission pathways of measles virus outbreaks (Rota and Bellini, 2003). Molecular epidemiologic tools also would be important in documenting deliberate bioterrorist introductions of wild-type or genetically modified measles virus strains.Read full chapterView PDFRead full chapterURL: https://www.sciencedirect.com/science/article/pii/B9780123694089000305Viruses as Infectious Agents: Human and Animal VirusesRoberto Cattaneo, Michael McChesney, in Encyclopedia of Virology (Fourth Edition), 2021Life CycleMeV begins its circuit through selected organs of the human body within alveolar macrophages and dendritic cells (DC), which express the primary receptor SLAM. These cells first transfer the infection through the epithelial barrier, and then spread it into lymphoid tissues (Fig. 4, left panel). SLAM was originally identified on activated B and T lymphocytes, but it is also expressed constitutively on immature thymocytes, memory T cells, and certain B cells. Subsets of other cell types, including monocytes and DC, also express SLAM. This cellular distribution overlaps with the susceptibility of different cell types to wild-type MeV infection. Another strong argument for the central role of SLAM in MeV tropism is the fact that three morbilliviruses (MeV, canine distemper virus, and rinderpest virus) enter cells via SLAM (human, canine, or bovine, respectively). Experiments in macaques revealed that the earliest target cells after intratracheal MeV inoculation are DCs and alveolar macrophages.More recently nectin-4, also called poliovirus receptor-like-4 (PVRL4), was shown to serve as the receptor for MeV spread in the upper airway epithelium (Fig. 4, right panel). This adherens junction protein interacts with H with five times higher affinity than SLAM. Nectin-4 sustains basolateral entry of MeV, and of all animal morbilliviruses examined, into upper airway epithelial cells, including those of the trachea.Contrary to the infections with other respiratory viruses, MeV enters the airway epithelium “en masse”: several days after host-to-host transmission, highly infected immune cells synchronously deliver large amounts of virus to the upper airways, for secondary amplification. Since the upper airways are the anatomical location most useful to support particle aerosolization, this two-phase mechanism of host invasion may account for the extremely contagious nature of MeV infection.The live attenuated MeV vaccine strain, Edmonston, can also use the regulator of complement activation membrane cofactor protein (MCP; CD46) as a receptor. The primary function of CD46 is to bind and promote inactivation of the C3b and C4b complement products, a process protecting human cells from lysis by autologous complement, a function that requires ubiquitous expression. Only tissue-culture adapted MeV interact with CD46, and indiscriminate cell entry through this protein correlates with MeV attenuation. A chimeric MeV expressing a vaccine strain H protein, which binds to CD46, is attenuated in the nonhuman primate, and productive infection is still confined to SLAM-expressing cells in vivo.View chapterPurchase bookRead full chapterURL: https://www.sciencedirect.com/science/article/pii/B9780128096338215069Rubeola VirusYvonne A. Maldonado, Avinash K. Shetty, in Principles and Practice of Pediatric Infectious Diseases (Fifth Edition), 2018Description of the PathogenMeasles virus (MV) is an enveloped, nonsegmented, negative-strand RNA virus belonging to the Morbillivirus genus in the Paramyxoviridae family. MV is closely related to rinderpest virus, a cattle pathogen that was eradicated in 2011.6 MV genome consists of 15,894 nucleotides and encodes eight proteins.7 The surface envelope glycoproteins and the hemagglutinin (H) and fusion (F) proteins are the main targets for development of neutralizing antibodies.8 The matrix (M) protein is important in virus assembly. The internal proteins, nucleoprotein (NP), polymerase phosphoprotein (P), and large protein (L), form the nucleocapsid. Two nonstructural proteins (C and V) regulate the cellular response to infection.7The receptor binding H protein interacts with F protein and mediates virus attachment and host cell entry. The F protein enhances cell-to-cell spread of the virus. Neutralizing antibodies are primarily directed against the H protein and confer lifelong immunity.8 There are three identified cellular entry receptors for MV. Human signaling lymphocyte activation molecule (i.e., hSLAM or CD150) is a common immune cell receptor for all MV strains, whereas membrane cofactor protein (i.e., MCP or CD46) is a receptor for vaccine strains.9 A third receptor, poliovirus receptor–like 4 (i.e., PVRL4 or Nectin 4) is expressed on the adherens junctions of epithelial cells.10Wild-type MV infects only primates. Although MV is a monotypic virus, minor genetic and antigenic variation occurs in some virus strains.11 These antigenic variations have not compromised long-lasting immunity. MV is inactivated by lipid solvents, heat, and light.View chapterPurchase bookRead full chapterURL: https://www.sciencedirect.com/science/article/pii/B9780323401814002279
|
|
Suggested by
LIGHTING
onto Mucosal Immunity March 26, 2022 5:34 AM
|
No comment yet.
Sign up to comment




 Your new post is loading...
Your new post is loading...