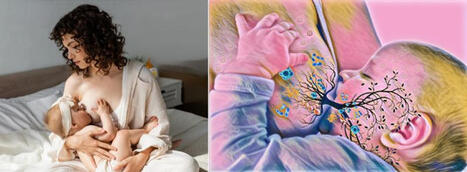
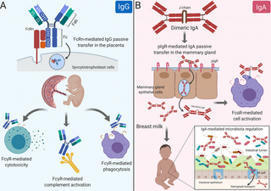
Citation: Langel SN, Otero CE, Martinez DR, Permar SR (2020) Maternal gatekeepers: How maternal antibody Fc characteristics influence passive transfer and infant protection. PLoS Pathog 16(3): e1008303. https://doi.org/10.1371/journal.ppat.1008303 Editor: Matthew J. Evans, Mount Sinai School of Medicine, UNITED STATES Published: March 26, 2020 Copyright: © 2020 Langel et al. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. Funding: SNL is supported by an NIH National Institute of Allergy and Infectious Diseases (NIAID: https://www.niaid.nih.gov) Ruth L. Kirschstein National Research Service Award T32 AI007392. CEO is supported by an NIH NIAID Ruth L. Kirschstein National Research Service Award T32 CA009111. DRM is supported by an NIH NIAID Ruth L. Kirschstein National Research Service Award T32 AI007151 and a Burroughs Wellcome Postdoctoral Enrichment Award (www.bwfund.org). SRP is supported by NIH NIAID R01 AI122909, P01 AI129859, and P01 AI117915. The funders had no role in study design, data collection, analysis, decision to publish, or preparation of the manuscript. The content is solely the view of the authors and does not necessarily represent the official views of the National Institutes of Health. Competing interests: I have read the journal's policy and have the following conflicts: SRP serves as a consultant for Pfizer, Sanofi, Moderna, and Merck vaccines and has a sponsored program on preclinical cytomegalovirus vaccine development with Merck and Moderna. All other authors declare no competing interests. Introduction Maternal antibodies (MatAbs) passively transferred across the placenta and into breast milk are critical for protection against infectious disease and immune development during the first year of life [1]. Passive transfer in the placenta and mammary gland (MG) is dependent on MatAbs binding to crystallizable fragment (Fc) receptors (FcRs) on polarized epithelial cells. For example, immunoglobulin G (IgG) transfers through the placenta by Fc domain binding to the Fc receptor neonatal (FcRn) on syncytiotrophoblasts [2], providing the fetus with a systemic source of protective IgG antibodies [3]. Additionally, maternal dimeric immunoglobulin A (dIgA) antibodies transfer into breast milk by binding to the polymeric immunoglobulin receptor (pIgR) on MG epithelial cells through the antibody joining chain (J-chain) [4] and provide immune protection in the gut while shaping microbiota colonization [4,5]. Yet MatAbs can interfere with the neonatal immune response, particularly after vaccination [6]. This Pearl explores the role of monomeric IgG, the only antibody isotype to cross the placenta, and polymeric IgA, the major antibody species in breast milk, and their Fc domain characteristics on passive transfer to and functional activity in the newborn. The IgG Fc domain and its effector functions in the context of MatAb passive transfer Antibodies contain 2 domains that exert a wide range of effector functions. The antigen-binding fragment (Fab) domain binds foreign antigens and drives antibody diversity [7], whereas the Fc is responsible for initiating innate immune cell activation and passive antibody transfer [8]. The classical FcRn-driven IgG transport mechanism is responsible for shuttling IgG within acidified endosomes across the syncytiotrophoblast cell barrier from maternal to fetal circulation (Fig 1A) [2]. Once in the neonate, the IgG Fc domain can engage classical type I Fc gamma (Fcγ) receptors (activating [FcγRI, FcγRIIa, FcγRIIc, FcγRIIIa, FcγRIIIb]; inhibitory [FcγRIIb]) or complement to mediate nonneutralizing functions like antibody-dependent cell-mediated cytotoxicity (ADCC) and antibody-dependent cellular phagocytosis (ADCP), or complement-dependent cytotoxicity (CDC), respectively (Fig 1A) [9]. Nonclassical type II FcRs are C-type lectin receptors, including CD209 (DC-SIGN) and CD23, which bind IgG to facilitate immune complex formation [9]. Considering each family of Fc receptors initiates distinct effector functions, the diversity of the Fc domain allows tailoring of nonneutralizing Fc-mediated activity to protect against viruses like HIV, influenza, and cytomegalovirus [10–12]. Alternatively, pathogens such as dengue virus utilize complement and FcR pathways for antibody-dependent enhancement of disease [13]. Download: PPT PowerPoint slide PNG larger image TIFF original image Fig 1. Maternal antibody passive transfer and functional activity in the neonate. (A) IgG passive transfer in the placenta influences FcγR-mediated cell cytotoxicity, phagocytosis, and complement activation in the developing fetus/newborn. (B) IgA passive transfer in the mammary gland results in FcαR- and IgA-mediated cell activation and microbiota regulation, respectively. Fab, antigen-binding fragment; Fc, crystallizable fragment; FcαR, Fc alpha receptor; FcRn, Fc receptor neonatal; FcγR, Fc gamma receptor; IgA, immunoglobulin A; IgG, immunoglobulin G; J-chain, joining chain; pIgR, polymeric immunoglobulin receptor. https://doi.org/10.1371/journal.ppat.1008303.g001 The IgG Fc domain mediates considerable heterogeneity of its effector functions depending on the subclass and glycan profile. For example, each IgG subclass (IgG1-4) has one N-glycosylation site in each CH2 domain, an important binding site for FcγRs (Fig 2). Interestingly, there are up to 36 possible antibody glycan profiles that could theoretically be present on each CH2 domain. This allows for combinatorial diversity of the Fc region with 144 different potential functional states for the 4 IgG subclasses [14]. This is relevant in the context of maternal–fetal immunity, as FcRn has different binding affinities to each IgG subclass, which may reflect their placental transfer efficiency [15]. Additionally, recent data suggest that Fc glycan profiles create antibody transfer hierarchies in the placenta of both healthy and HIV-infected pregnant women. For example, in healthy pregnant women, there is a shift toward IgG galactosylated antibodies, which have higher FcRn-binding affinity, are more efficiently transferred across the placenta, and enhance natural killer (NK) cell degranulation and chemokine secretion [16]. Additionally, binding of tetanus toxoid–specific IgG to placental FcγRIIa H131, FcγRIIa R131, and FcγRIIIa F158 (but not canonical FcRn) was positively associated with placental IgG transfer efficiency in HIV-infected women, suggesting that noncanonical placental FcRs may also play a role in IgG placental transfer [17,18]. Fc-mediated differential selection of IgG antibodies in the placenta is likely an adaptive evolutionary mechanism to passively transfer the most effective antibodies to the infant, which can be altered by disease status. Download: PPT PowerPoint slide PNG larger image TIFF original image Fig 2. Schematic representation of IgA and IgG glycosylation. N-linked glycosylation is depicted as yellow circles, whereas O-linked glycosylation is depicted as green stars. IgA, immunoglobulin A; IgG, immunoglobulin G; sIgA2, secretory IgA. https://doi.org/10.1371/journal.ppat.1008303.g002 Do IgA Fc region characteristics influence IgA passive transfer or effector function in breast milk? IgA antibodies bind their own unique Fc receptors that facilitate epithelial cell transcytosis and innate immune cell activation. dIgA antibodies are composed of 2 monomers, linked by a 15-kDa J-chain. Transport of dIgA into breast milk is dependent on C-terminal binding of the J-chain to a portion of pIgR, known as the secretory component, on the basolateral surface of MG epithelial cells [19]. Without the J-chain, IgA antibodies are secreted as monomers and are not actively transported across the mucosal epithelium [20]. After transport of the J-chain/pIgR complex to the apical portion of the cell, pIgR is cleaved, releasing secretory IgA (sIgA) into breast milk and other mucosal fluids (Fig 1B) [21]. IgA also binds to Fc alpha receptor (FcαR) [22] on the surface of myeloid cells. Monomeric serum IgA induces inhibitory signals, whereas IgA immune complexes have increased avidity to and cross-link FcαRI, resulting in proinflammatory responses [23]. The dominant immunoglobulin class in breast milk sIgA has decreased affinity for FcαRI likely due to steric hindrance from the attached secretory component [24]. Although the opsonic activity of sIgA is poor compared with monomeric and dIgA [25], sIgA can initiate macrophage phagocytosis and neutrophil respiratory burst [26,27]. Further defining the anti- and proinflammatory effects of IgA subclass–FcR interactions would allow fine tuning of breast milk immunity and may represent an attractive therapeutic strategy. Considering breast milk sIgA protects from pathogenic insult and facilitates maturation of the microbiota in early life [28], understanding breast milk antibody Fc–mediated effector functions is integral to neonatal intestinal health (Fig 1B). This is further supported by the fact that bacteria have evolved mechanisms to block IgA and FcαR interactions [29]. Additionally, it is likely that the more complex and extensive glycosylation pattern of IgA antibodies (Fig 2) impacts effector function in milk. Indeed, mucosal secretions, including breast milk, contain mostly IgA2 [30], which has 2 [IgA2m(1)] or 3 [IgA2m(2)] additional conserved N-glycans compared with IgA1 [31], which dominates in serum [32]. Recent evidence demonstrates that IgA glycan–bacteria interactions regulate gut microbiota composition and metabolism as well as retrograde transport of sIgA immune complexes back to the lamina propria independent of antibody–epitope interactions [33–35]. Additionally, the sialic acid in IgA antibody’s C-terminal tail competes with receptor binding to some viruses, providing an innate line of defense against infection [36]. This is relevant to breast milk IgA passive transfer, considering that the leading causes of severe pediatric gastroenteritis worldwide (rotavirus and norovirus) both utilize sialic acid receptors for intestinal infection [37,38]. Studies are needed to define the mechanisms of Fc-mediated IgA effector functions in breast milk, including their interactions with the developing infant microbiome and protection against intestinal viral infections. MatAb interference is influenced by MatAb Fc domain–receptor interactions Despite the well-documented benefits of MatAbs on early life immunity [3], a mounting body of evidence indicates that MatAbs can inhibit immune responses to certain infant vaccinations [6,39]. A recent meta-analysis demonstrated that MatAbs acquired transplacentally inhibited antibody responses to priming vaccinations and these effects were not overcome by administration of a booster dose [40]. This highlights the “window of susceptibility” that exists for infants when MatAbs are not high enough for seroprotection yet still interfere with infant vaccine responses. Multiple mechanisms have been proposed to describe both Fab- and Fc-mediated MatAb interference. These include live virus vaccine neutralization, inhibition of B-cell responses by epitope masking [39], and IgG Fc binding to FcγRIIB [41]. Kim and colleagues demonstrated that B-cell responses to a live attenuated measles vaccine were inhibited by passively transferred measles-specific IgG antibodies in a FcγRIIB-dependent manner, suggesting that IgG Fc region characteristics contribute to suppression of the immune response [41]. Additionally, removing the glycans from IgG2b abolished its immunosuppressive activity both in vitro and in vivo [42,43]. Considering that the mechanisms of MatAb interference likely differ depending on vaccine type (live attenuated, inactivated, subunit), route of delivery (oral, subcutaneous [SQ], intramuscular [IM]), and adjuvant formulation, defining glycan-dependent passive transfer of maternal IgG antibody subclasses in the placenta is crucial for developing effective maternal immunization strategies. Although less studied, IgA antibodies in breast milk may also contribute to interference of immune responses to oral infant vaccines such as rotavirus and poliovirus [44,45]. Indeed, the 2 oral rotavirus vaccines Rotarix and Rotateq demonstrate lower efficacy and immunogenicity in infants from some low- and middle-income countries (LMICs) [46,47] where women tend to have higher titers of antirotavirus IgA antibodies and neutralizing activity in milk [48,49]. The high rotavirus neutralizing activity in breast milk of women from LMICs may partially explain the decrease in rotavirus vaccine efficacy; however, IgA MatAb interference is not well defined [49]. Although infant CD4+ T-cell responses are mostly unaffected by MatAb interference [6,39], recent work demonstrated that MatAbs dampen mucosal T-cell responses against commensal bacteria [50] and limit the expansion of T follicular helper (TFH) cells in the germinal center (GC) [39]. The premature decline in GC TFH cells resulted in the reduction or prevention of plasma cell and memory B-cell generation in a MatAb- and antigen dose–dependent manner [39]. Interestingly, at low or intermediate titers, MatAbs did not prevent the induction of memory B cells, suggesting a gradient effect of MatAbs on infant immune responses [39]. Defining the functional consequences of MatAb gradients will be essential for infant vaccine design and immunization timing. For example, and in addition to previously discussed mechanisms, there is evidence that preexisting antibodies can promote higher affinity antibody responses due to competitive binding in the GC [51] and increased uptake and antigen presentation through immune complexes (ICs) [52] in a Fc glycosylation–dependent manner [53,54]. However, more research is needed to determine the effects of these mechanisms in the setting of passively transferred MatAbs and infant GC responses. Harnessing MatAb Fc region characteristics and receptor interactions to fine-tune maternal immunizations that maximize infant protection Passive transfer of MatAbs is central to pathogen protection and immune system development in early life. However, MatAb-mediated interference dampens antibody responses to vaccinations, leaving children more susceptible to infections while increasing transmission rates to unvaccinated cohorts. Recent work demonstrated that maternal IgG antibodies are differentially transferred across the placenta in an Fc glycan–dependent manner [16,17]. Considering vaccination strategies could direct antigen-specific antibody glycosylation [55], defining MatAb glycan profiles represents an adaptable and powerful mechanism to fine-tune maternal immunizations that maximize infant protection while limiting MatAb interference. Future studies are needed to determine (1) how maternal vaccination and their distinct adjuvant mixtures alter IgG Fc domain glycosylation and whether certain glycan profiles are associated with IgG Fc-mediated MatAb interference and (2) whether or not the IgA Fc domain regulates passive transfer in the MG or effector function in breast milk. Defining the molecular mechanisms of Fc-mediated functional activity at the maternal–fetal/neonatal interface is critical for developing next-generation maternal vaccines and antibody-based therapeutics to improve the health of the mother–neonatal dyad. References 1. Hurley WL, Theil PK. Perspectives on immunoglobulins in colostrum and milk. Nutrients. 2011 Apr;3(4): 442–474. pmid:22254105; PubMed Central PMCID: PMC3257684. View Article PubMed/NCBI Google Scholar 2. Simister NE, Story CM, Chen HL, Hunt JS. An IgG-transporting Fc receptor expressed in the syncytiotrophoblast of human placenta. Eur J Immunol. 1996 July;26(7): 1527–1531. pmid:8766556 View Article PubMed/NCBI Google Scholar 3. Fouda GG, Martinez DR, Swamy GK, Permar SR. The Impact of IgG transplacental transfer on early life immunity. Immunohorizons. 2018 Jan 1;2(1): 14–25. pmid:29457151 View Article PubMed/NCBI Google Scholar 4. Rogier EW, Frantz AL, Bruno MEC, Wedlund L, Cohen DA, Stromberg AJ, et al. Secretory antibodies in breast milk promote long-term intestinal homeostasis by regulating the gut microbiota and host gene expression. Proc Natl Acad Sci U S A. 2014 Feb 25;111(8):3074–3079. pmid:24569806 View Article PubMed/NCBI Google Scholar 5. Sadeharju K, Knip M, Virtanen SM, Savilahti E, Tauriainen S, Koskela P, et al. Maternal antibodies in breast milk protect the child from enterovirus infections. Pediatrics. 2007 May;119: 941–946. pmid:17473095. View Article PubMed/NCBI Google Scholar 6. Niewiesk S. Maternal antibodies: clinical significance, mechanism of interference with immune responses, and possible vaccination strategies. Front Immunol. 2014 Sep 16;5: 446. pmid:25278941; PubMed Central PMCID: PMC4165321. View Article PubMed/NCBI Google Scholar 7. Li Z, Woo CJ, Iglesias-Ussel MD, Ronai D, Scharff MD. The generation of antibody diversity through somatic hypermutation and class switch recombination. Genes Dev. 2014 Jan 1;18(1): 1–11. pmid:14724175. View Article PubMed/NCBI Google Scholar 8. Lu LL, Suscovich TJ, Fortune SM, Alter G. Beyond binding: antibody effector functions in infectious diseases. Nat Rev Immunol. 2018 Jan;18(1):46–61. pmid:29063907 View Article PubMed/NCBI Google Scholar 9. Pincetic A, Bournazos S, DiLillo DJ, Maamary J, Wang TT, Dahan R, et al. Type I and type II Fc receptors regulate innate and adaptive immunity. Nat Immunol. 2014 Aug;15(8):707–16. pmid:25045879 View Article PubMed/NCBI Google Scholar 10. Horwitz JA, Bar-On Y, Lu CL, Fera D, Lockhart AAK, Lorenzi JCC, et al. Non-neutralizing Antibodies Alter the Course of HIV-1 Infection In Vivo. Cell. 2017 Aug 10;170(4):637–648.e10. pmid:28757252 View Article PubMed/NCBI Google Scholar 11. Henry Dunand CJ, Leon PE, Huang M, Choi A, Chromikova V, Ho IY, et al. Both Neutralizing and Non-Neutralizing Human H7N9 Influenza Vaccine-Induced Monoclonal Antibodies Confer Protection. Cell Host Microbe. 2016 Jun 8;19(6):800–813. pmid:27281570 View Article PubMed/NCBI Google Scholar 12. Nelson CS, Huffman T, Jenks JA, Cisneros de la Rosa E, Xie G, Vandergrift N, et al. HCMV glycoprotein B subunit vaccine efficacy mediated by nonneutralizing antibody effector functions. Proc Natl Acad Sci U S A. 2018 Jun 12;115(24):6267–6272. pmid:29712861 View Article PubMed/NCBI Google Scholar 13. Katzelnick LC, Gresh L, Halloran ME, Mercado JC, Kuan G, Gordon A, et al. Antibody-dependent enhancement of severe dengue disease in humans. 2017 Nov 17;358(6365):929–932. pmid:29097492 View Article PubMed/NCBI Google Scholar 14. Jennewein MF, Alter G. The Immunoregulatory Roles of Antibody Glycosylation. Trends Immunol. 2017 May;38(5):358–372. pmid:28385520. View Article PubMed/NCBI Google Scholar 15. Palmeira P, Quinello C, Silveira-Lessa AL, Zago CA, Carneiro-Sampaio M (2012) IgG placental transfer in healthy and pathological pregnancies. Clin Dev Immunol. 2012;2012: 985646. pmid:22235228; PubMed Central PMCID: PMC3251916. View Article PubMed/NCBI Google Scholar 16. Jennewein MF, Goldfarb I, Dolatshahi S, Cosgrove C, Noelette FJ, Krykbaeva M, et al. Fc Glycan-Mediated Regulation of Placental Antibody Transfer. Cell. 2019 Jun 27;178(1):202–215.e14. pmid:31204102; PubMed Central PMCID: PMC6741440. View Article PubMed/NCBI Google Scholar 17. Martinez DR, Fong Y, Li SH, Yang F, Jennewein MF, Weiner JA, et al. Fc Characteristics Mediate Selective Placental Transfer of IgG in HIV-Infected Women. Cell. 2019 Jun 27;178(1):190–201.e11. pmid:31204101; PubMed Central PMCID: PMC6727200. View Article PubMed/NCBI Google Scholar 18. Martinez DR, Fouda GG, Peng X, Ackerman ME, Permar SR. Noncanonical placental Fc receptors: What is their role in modulating transplacental transfer of maternal IgG? PLoS Pathog. 2018 Aug 30;14(8):e1007161. pmid:30161231; PubMed Central PMCID: PMC6117057. View Article PubMed/NCBI Google Scholar 19. Johansen FE, Kaetzel CS. Regulation of the polymeric immunoglobulin receptor and IgA transport: new advances in environmental factors that stimulate pIgR expression and its role in mucosal immunity. Mucosal Immunol. 2011 Nov;4(6):598–602. pmid:21956244; PubMed Central PMCID: PMC3196803. View Article PubMed/NCBI Google Scholar 20. Johansen FE, Braathen R, Brandtzaeg P. Role of J chain in secretory immunoglobulin formation. Scand J Immunol. 2000 Sep;52(3): 240–248. pmid:10972899 View Article PubMed/NCBI Google Scholar 21. Johansen F-E, Braathen R, Brandtzaeg P. The J Chain Is Essential for Polymeric Ig Receptor-Mediated Epithelial Transport of IgA. J Immunol. 2001 Nov 1;167(9):5185–5192. pmid:11673531. View Article PubMed/NCBI Google Scholar 22. Aleyd E, Heineke MH, van Egmond M. The era of the immunoglobulin A Fc receptor FcalphaRI; its function and potential as target in disease. Immunol Rev. 2015 Nov;268(1):123–138. pmid:26497517. View Article PubMed/NCBI Google Scholar 23. Hansen IS, Baeten DLP, den Dunnen J. The inflammatory function of human IgA. Cell Mol Life Sci. 2019 Mar;76(6):1041–1055. pmid:30498997; PubMed Central PMCID: PMC6513800 View Article PubMed/NCBI Google Scholar 24. Bonner A, Furtado PB, Almogren A, Kerr MA, Perkins SJ (2008) Implications of the near-planar solution structure of human myeloma dimeric IgA1 for mucosal immunity and IgA nephropathy. J Immunol. 2008 Jan 15;180(2):1008–1018. pmid:18178841. View Article PubMed/NCBI Google Scholar 25. Bakema JE, van Egmond M. Immunoglobulin A: A next generation of therapeutic antibodies? mAbs. 2011 Jul 1;3(4): 352–361. pmid:21691145 View Article PubMed/NCBI Google Scholar 26. Stewart WW, Kerr MA. The specificity of the human neutrophil IgA receptor (Fc alpha R) determined by measurement of chemiluminescence induced by serum or secretory IgA1 or IgA2. Immunology. 1990 Nov;71(3): 328–334. pmid:2269470; PubMed Central PMCID: PMC1384427. View Article PubMed/NCBI Google Scholar 27. Gorter A, Hiemstra PS, Leijh PC, van der Sluys ME, van den Barselaar MT, van Es LA, et al. IgA- and secretory IgA-opsonized S. aureus induce a respiratory burst and phagocytosis by polymorphonuclear leucocytes. Immunology. 1987 Jul; 61(3): 303–309. pmid:3610212; PubMed Central PMCID: PMC1453393. View Article PubMed/NCBI Google Scholar 28. Fadlallah J, El Kafsi H, Sterlin D, Juste C, Parizot C, Dorham, K, et al. Microbial ecology perturbation in human IgA deficiency. Sci Transl Med. 2018 May 2;10(439): eaan1217. pmid:29720448 View Article PubMed/NCBI Google Scholar 29. Woof JM. The human IgA-Fc alpha receptor interaction and its blockade by streptococcal IgA-binding proteins. Biochem Soc Trans. 2002 Aug;30(4):491–494. pmid:12196121. View Article PubMed/NCBI Google Scholar 30. Kett K, Brandtzaeg P, Radl J, Haaijman JJ. Different subclass distribution of IgA-producing cells in human lymphoid organs and various secretory tissues. J Immunol. 1986 May 15;136(10):3631–3635. pmid:3517160. View Article PubMed/NCBI Google Scholar 31. Mattu TS, Pleass RJ, Willis AC, Kilian M, Wormald MR, Lellouch AC, et al. The glycosylation and structure of human serum IgA1, Fab, and Fc regions and the role of N-glycosylation on Fcα receptor interactions. J Biol Chem. 1998 Jan 23;273(4):2260–2272. pmid:9442070. View Article PubMed/NCBI Google Scholar 32. Delacroix DL, Dive C, Rambaud JC, Vaerman JP. IgA subclasses in various secretions and in serum. Immunology. 1982 Oct;47(2):383–375. pmid:7118169; PubMed Central PMCID: PMC1555453. View Article PubMed/NCBI Google Scholar 33. Nakajima A, Vogelzang A, Maruya M, Miyajima M, Murata M, Son A, et al. IgA regulates the composition and metabolic function of gut microbiota by promoting symbiosis between bacteria. J Exp Med. 2018 Aug 6;215(8):2019–2034. pmid:30042191; PubMed Central PMCID: PMC6080902. View Article PubMed/NCBI Google Scholar 34. Rochereau N, Drocourt D, Perouzel E, Pavot V, Redelinghuys P, et al. Dectin-1 is essential for reverse transcytosis of glycosylated SIgA-antigen complexes by intestinal M cells. PLoS Biol. 2013 Sep;11(9):e1001658. pmid:24068891; PubMed Central PMCID: PMC3775721. View Article PubMed/NCBI Google Scholar 35. Mathias A, Corthesy B. Recognition of gram-positive intestinal bacteria by hybridoma- and colostrum-derived secretory immunoglobulin A is mediated by carbohydrates. J Biol Chem. 2011 May 13;286(19):17239–17247. pmid:21454510; PMCID: PMC3089566. View Article PubMed/NCBI Google Scholar 36. Maurer MA, Meyer L, Bianchi M, Turner HL, Le NPL, et al. Glycosylation of Human IgA Directly Inhibits Influenza A and Other Sialic-Acid-Binding Viruses. Cell Rep. 2018 Apr 3;23(1):90–99. pmid:29617676; PMCID: PMC5905402. View Article PubMed/NCBI Google Scholar 37. Graziano VR, Wei J, Wilen CB (2019) Norovirus Attachment and Entry. Viruses. 2019 May 30;11(6). pii: E495. pmid:31151248; PMCID: PMC6630345. View Article PubMed/NCBI Google Scholar 38. Stencel-Baerenwald JE, Reiss K, Reiter DM, Stehle T, Dermody TS. The sweet spot: defining virus-sialic acid interactions. Nat Rev Microbiol. 2014 Nov;12(11):739–749. pmid:25263223; PMCID: PMC4791167. View Article PubMed/NCBI Google Scholar 39. Vono M, Eberhardt CS, Auderset F, Mastelic-Gavillet B, Lemeille S, Christensen D, et al. Maternal Antibodies Inhibit Neonatal and Infant Responses to Vaccination by Shaping the Early-Life B Cell Repertoire within Germinal Centers. Cell Rep. 2019 Aug 13;28(7):1773–1784.e5. pmid:31412246. View Article PubMed/NCBI Google Scholar 40. Voysey M, Kelly DF, Fanshawe TR, Sadarangani M, O'Brien KL, Perera R, et al. The Influence of Maternally Derived Antibody and Infant Age at Vaccination on Infant Vaccine Responses: An Individual Participant Meta-analysis. JAMA Pediatr. 2017 Jul 1;171(7):637–646. pmid:28505244; PMCID: PMC5710349. View Article PubMed/NCBI Google Scholar 41. Kim D, Huey D, Oglesbee M, Niewiesk S. Insights into the regulatory mechanism controlling the inhibition of vaccine-induced seroconversion by maternal antibodies. 2011 Jun 9;117(23):6143–51. pmid:21357766; PMCID: PMC3122939. View Article PubMed/NCBI Google Scholar 42. Heyman B, Nose M, Weigle WO. Carbohydrate chains on IgG2b: a requirement for efficient feedback immunosuppression. J Immunol. 1985 Jun;134(6):4018–4023. pmid:3872908. View Article PubMed/NCBI Google Scholar 43. Heyman B, Wigzell H. Immunoregulation by monoclonal sheep erythrocyte-specific IgG antibodies: suppression is correlated to level of antigen binding and not to isotype. J Immunol. 1984 Mar;132(3):1136–1143. pmid:6363534. View Article PubMed/NCBI Google Scholar 44. Chan J, Nirwati H, Triasih R, Bogdanovic-Sakran N, Soenarto Y, Hakimi M, et al. Maternal antibodies to rotavirus: could they interfere with live rotavirus vaccines in developing countries? Vaccine. 2011 Feb 1;29(6):1242–1247. pmid:21147127. View Article PubMed/NCBI Google Scholar 45. Plotkin SA, Katz M, Brown RE, Pagano JS. Oral poliovirus vaccination in newborn African infants. The inhibitory effect of breast feeding. Am J Dis Child. 1966 Jan;111(1):27–30. pmid:5900282. View Article PubMed/NCBI Google Scholar 46. Ruiz-Palacios GM, Perez-Schael I, Velazquez FR, Abate H, Breuer T, Clemens SC, et al. Safety and efficacy of an attenuated vaccine against severe rotavirus gastroenteritis. N Engl J Med. 2006 Jan 5;354(1):11–22. pmid:16394298. View Article PubMed/NCBI Google Scholar 47. Vesikari T, Uhari M, Renko M, Hemming M, Salminen M, et al. Impact and effectiveness of RotaTeq(R) vaccine based on 3 years of surveillance following introduction of a rotavirus immunization program in Finland. Pediatr Infect Dis J. 2013 Dec;32(12):1365–1373. pmid:24051998. View Article PubMed/NCBI Google Scholar 48. Moon SS, Tate JE, Ray P, Dennehy PH, Archary D, Coutsoudis A, et al. Differential profiles and inhibitory effect on rotavirus vaccines of nonantibody components in breast milk from mothers in developing and developed countries. Pediatr Infect Dis J. 2013 Aug;32(8):863–870. pmid:23584581; PMCID: PMC4610365. View Article PubMed/NCBI Google Scholar 49. Moon SS, Wang Y, Shane AL, Nguyen T, Ray P, Dennehy P, et al. Inhibitory effect of breast milk on infectivity of live oral rotavirus vaccines. Pediatr Infect Dis J. 2010 Oct;29(10):919–23. pmid:20442687; PMCID: PMC3704726. View Article PubMed/NCBI Google Scholar 50. Koch MA, Reiner GL, Lugo KA, Kreuk LS, Stanbery AG, Ansaldo E, et al. Maternal IgG and IgA Antibodies Dampen Mucosal T Helper Cell Responses in Early Life. Cell. 2016 May 5;165(4):827–41. pmid:27153495; PMCID: PMC4866587. View Article PubMed/NCBI Google Scholar 51. Zhang Y, Meyer-Hermann M, George LA, Figge MT, Khan M, et al. Germinal center B cells govern their own fate via antibody feedback. J Exp Med. 2013 Mar 11;210(3):457–464. pmid:23420879; PMCID: PMC3600904. View Article PubMed/NCBI Google Scholar 52. Garg AK, Desikan R, Dixit NM. Preferential Presentation of High-Affinity Immune Complexes in Germinal Centers Can Explain How Passive Immunization Improves the Humoral Response. Cell Rep. 2019 Dec 17;29(12):3946–3957.e5. pmid:31851925. View Article PubMed/NCBI Google Scholar 53. Heesters Balthasar A, Chatterjee P, Kim Y-A, Gonzalez Santiago F, Kuligowski Michael P, et al. Endocytosis and Recycling of Immune Complexes by Follicular Dendritic Cells Enhances B Cell Antigen Binding and Activation. Immunity. 2013 Jun 27;38(6):1164–75. pmid:23770227; PMCID: PMC3773956. View Article PubMed/NCBI Google Scholar 54. Lofano G, Gorman MJ, Yousif AS, Yu W-H, Fox JM, Dugast AS, et al. Antigen-specific antibody Fc glycosylation enhances humoral immunity via the recruitment of complement. Sci Immunol. 2018 Aug 17;3(26). pii: eaat7796. pmid:30120121; PMCID: PMC6298214. View Article PubMed/NCBI Google Scholar 55. Mahan AE, Jennewein MF, Suscovich T, Dionne K, Tedesco J, et al. Antigen-Specific Antibody Glycosylation Is Regulated via Vaccination. PLoS Pathog. 2016 Mar 16;12(3):e1005456. pmid:26982805; PMCID: PMC4794126. View Article PubMed/NCBI Google Scholar

|
Scooped by
Gilbert C FAURE
onto Mucosal Immunity December 9, 2020 9:09 AM
|
No comment yet.
Sign up to comment


 Your new post is loading...
Your new post is loading...