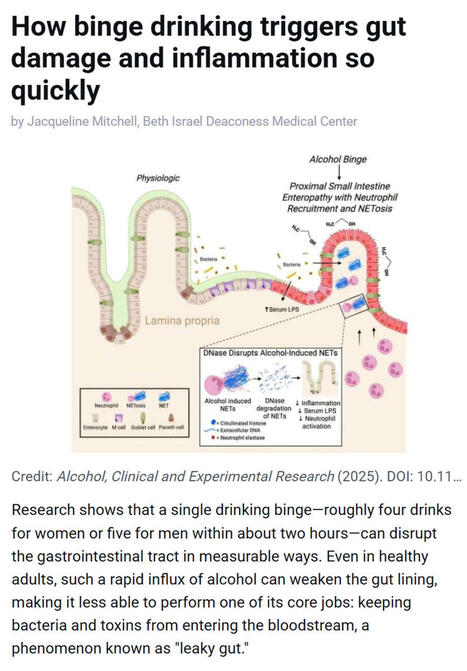
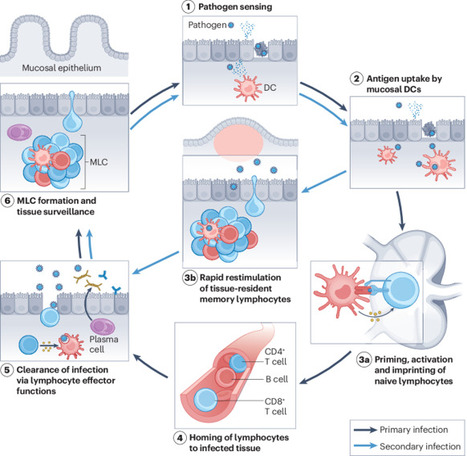
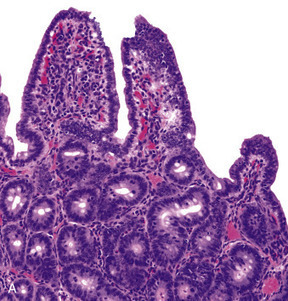
Bovine Coronavirus Related terms: View all Topics Coronaviridae In Fenner's Veterinary Virology (Fifth Edition), 2017 CORONAVIRUSES of Cattle and Horses BOVINE CORONAVIRUS Bovine coronavirus infections are associated with three distinct clinical syndromes in cattle: calf diarrhea, winter dysentery (hemorrhagic diarrhea) in adult cattle, and respiratory infections in cattle of various ages, including the bovine respiratory disease complex (shipping fever) in feedlot cattle. Coronaviruses were first reported as a cause of diarrhea in calves in the United States in 1973, and since then they have been recognized worldwide in association with the three clinical syndromes. The economic impact of respiratory disease and calf diarrhea is considerable. Although many coronaviruses have restricted host ranges, betacoronaviruses such as bovine and SARS coronaviruses (Table 24.1) can infect other animal species, including wildlife. Bovine coronavirus is closely related to the human coronavirus OC43 that causes the common cold; indeed, OC43 has been proposed to represent prior zoonotic transmission of bovine coronavirus. Bovine coronavirus has also been shown to infect dogs subclinically and to infect turkey poults, leading to fecal virus shedding, diarrhea, seroconversion, and transmission to contact controls. Genetically and/or antigenically related bovine coronavirus variants have been isolated from dogs with respiratory disease, humans with diarrhea, and captive or wild ruminants with intestinal disease similar to winter dysentery of cattle. The latter include Sambar deer (Cerous unicolor), waterbuck (Kobus ellipsiprymnus), giraffe (Giraffa camelopardalis), and white-tailed deer (Odocoileus virgineanus). Bovine coronavirus has also been linked to enteric disease in South American camelids. Interestingly, the human enteric coronavirus and wild ruminant coronaviruses both infected and caused diarrhea in experimentally exposed gnotobiotic calves, and the inoculated calves were subsequently immune to infection with bovine coronavirus. Despite the different disease syndromes and apparent interspecies transmission of bovine coronavirus and its variants, only a single serotype of bovine coronavirus is recognized, and there is little sequence diversity between the wild ruminant coronaviruses and coronaviruses associated with the different disease syndromes in cattle. Furthermore, there are few common sequence differences to explain differences in host or tissue tropism. The host cell receptor for bovine coronavirus is sialic acid, which reflects the wide tropism of this virus and explains the presence of a HE gene in the virus. Clinical Features and Epidemiology Coronavirus-induced diarrhea commonly occurs in calves under 3 weeks of age after the decline of passively acquired antibodies, but disease can occur in calves up to 3 months of age. The severity of diarrhea and dehydration depends on the infecting dose as well as the age and immune status of the calf. Coinfections with other enteric pathogens such as rotavirus, torovirus, cryptosporidia, and enterotoxigenic or enteropathogenic E. coli are common; their additive or synergistic effects increase the severity of diarrhea. Calf coronavirus diarrhea is often seasonal, being more common in winter in part because of the increased stability of the virus in the cold. Bovine coronavirus has also been implicated as a cause of winter dysentery, a sporadic, acute enteric disease of adult cattle worldwide that is especially prevalent during winter months, as the name implies. Winter dysentery is characterized by explosive, often bloody diarrhea, accompanied by decreased milk production, depression, anorexia, and frequent respiratory signs. Morbidity rates range from 20% to 100% in affected herds, but mortality rates are usually low (1–2%). A similar winter dysentery syndrome associated with bovine coronaviruses variants occurs in captive and wild ruminants. This finding suggests that certain wild ruminants (deer, elk, caribou, etc.) that share common grazing areas with cattle could be a reservoir for coronavirus strains transmissible to cattle, or vice versa. Bovine coronavirus also causes mild respiratory disease (coughing, rhinitis) or pneumonia in 2–6-month-old calves. An epidemiologic study of calves from birth to 20 weeks of age confirmed both fecal and nasal shedding of coronavirus, with diarrhea prominent upon initial infection. The calves subsequently shed virus intermittently via the respiratory route, with or without signs of disease, suggesting that long-term mucosal immunity in the upper respiratory tract is ineffective in mediating virus clearance. As a consequence, coronavirus may recycle among cattle of all ages and regardless of their immune status, with sporadic nasal or fecal shedding from individual animals. Alternatively, new virus strains may be introduced when cattle from different sources are comingled, or from cohabiting wild ruminants. Since 1993, bovine coronavirus has been incriminated as a precipitating cause of the bovine respiratory disease (shipping fever) complex. Both respiratory and enteric shedding of bovine coronavirus are common in affected feedlot cattle, peaking shortly after arrival at feedlots. Since its discovery, bovine coronavirus repeatedly has been identified in the lungs of feedlot cattle that died with bovine respiratory disease complex. Most feedlot cattle also seroconvert to bovine coronavirus within 3 weeks of arrival. Importantly, studies suggest that cattle arriving at feedlots with high serum titers of bovine coronavirus antibody were less likely to shed virus or to develop shipping fever. This observation suggests a role for serum antibodies in protection, or as an indicator of recent infection and active immunity. Pathogenesis and Pathology Concurrent fecal and nasal virus shedding persists for up to 10 days after coronavirus infection of calves. Coronavirus antigen is commonly detected in epithelial cells of both the upper respiratory and intestinal tracts, and occasionally in the lung. The pathogenesis of coronavirus enteritis in calves is similar to that caused by rotavirus, with the notable exception of extensive involvement of the large intestine by coronavirus. Disease occurs most commonly in calves at about 1–3 weeks of age, when virus exposure increases and antibody titers in the dam’s milk begin to wane. The pathogenesis and consequences of enteric coronavirus infection of calves are similar to those described for transmissible gastroenteritis in piglets. The destruction of the mature absorptive cells lining the intestinal villi and mucosal surface in the large intestine leads to maldigestion and malabsorption, with rapid loss of water and electrolytes. The resultant hypoglycemia, acidosis, and hypovolemia can progress to circulatory failure and death, especially in very young animals. The pathogenesis and lesions of winter dysentery of dairy and beef cattle resemble those of calf diarrhea, but often with marked intestinal hemorrhage and extensive necrosis of cells within the crypts of the large intestinal mucosa. Nasal and fecal shedding is more transient (up to 4–5 days). The anorexia and depression seen in dairy cattle with winter dysentery may explain the precipitous and sometimes prolonged decrease in milk production. The cause of the acute and often voluminous bloody diarrhea in some cattle is unexplained. Both nasal and fecal shedding of bovine coronavirus can occur soon after cattle are transported to feedlots. Coronavirus infection is probably important in predisposing cattle entering feedlots to secondary bacterial infection that results in the characteristic shipping fever pneumonia—a severe, often fatal fibrinous bronchopneumonia caused by Mannheimia haemolytica biotype A, serotype 1 infection. Bovine coronavirus antigen also has been detected in epithelial cells of the upper (trachea, bronchi) and lower (terminal bronchioles and alveoli) respiratory tract of some affected cattle, but the precise role of coronavirus in precipitating the bovine respiratory disease complex awaits definitive characterization. Diagnosis Initially, the diagnosis of enteric bovine coronavirus infections was based on the detection of virus by electron microscopy. Cell culture isolation became a viable option when it was discovered that the virus could be grown when trypsin was added to the medium—virus replication is recognized by hemadsorption or cytopathogenic effects, and the presence of coronavirus is confirmed by diagnostic tests. An array of assays is now available for detection of bovine (or variant) coronaviruses in cell culture or diagnostic specimens such as feces or nasal swabs, including ELISAs that incorporate monoclonal antibodies for antigen capture, immune electron microscopy using hyperimmune antiserum, and RT-PCR using bovine coronavirus or pan-coronavirus-specific primers to detect viral RNA. The use of RT-PCR for detection of bovine coronavirus has significantly increased the detection of this agent, particularly in respiratory samples, and has also substantially increased the recognized period of virus shedding by infected animals. Postmortem diagnosis is performed on acute fresh or fixed respiratory or intestinal tissues using hyperimmune antisera or monoclonal antibodies for immunofluorescence or immunohistochemical tissue staining. Immunity, Prevention, and Control Passive Immunity to Enteric Bovine Coronavirus Infections in Calves Because coronavirus diarrhea occurs in young calves during the nursing period, maternal vaccination is required to provide immediate passive (lactogenic) immunity. Passive immunity to enteric viral infections in calves correlates with high levels of IgG1 antibodies in colostrum and milk. In ruminants, IgG1 antibodies are dominant in colostrum and milk and are selectively transported from serum. Most adult cattle are seropositive for antibodies to bovine coronavirus. Therefore, parenteral vaccination of mothers with adjuvanted inactivated bovine coronavirus vaccines effectively boosts IgG1 antibody titers in serum and mammary secretions, to provide enhanced passive immunity to calves. Immunity to Respiratory Bovine Coronavirus Infections The correlates of immunity to respiratory coronavirus infections in cattle are not clearly defined. The serum antibody titer to bovine coronavirus may be a marker for respiratory protection, as coronavirus-specific antibody titers and isotype (IgG1, IgG2, IgA) were correlated with protection of calves and feedlot cattle against subsequent occurrence of respiratory disease, pneumonia, or coronavirus shedding. However, it can be difficult to distinguish whether serum antibodies are correlates of protection, or whether they merely reflect prior enteric or respiratory coronavirus infection. Intranasal vaccination using live-attenuated enteric coronavirus vaccine has been proposed to reduce the risk of bovine respiratory disease complex (so-called “shipping fever”) in cattle entering feedlots. Respiratory System Jeff L. Caswell, Kurt J. Williams, in Jubb, Kennedy & Palmer's Pathology of Domestic Animals: Volume 2 (Sixth Edition), 2016 Bovine coronavirus. Bovine coronavirus (BCoV) is an important cause of enteric disease in young calves, and the same strains occasionally induce respiratory disease in calves 2-16 weeks of age. BCoV is a less frequent but nonetheless important cause of respiratory disease in calves, and deserves more attention than it has received in the past. Isolates from occurrences of respiratory and diarrheic diseases have similar genotypes, and a single outbreak may include both forms of disease. Respiratory signs include fever, serous nasal discharge, sneezing, and coughing. The virus replicates primarily in the nasal and tracheal epithelium, and occasionally in the lung. Many calves shed BCoV in their nasal secretions and/or seroconvert to BCoV in the first month after arrival in feedlots, but the association between seroconversion or virus shedding and increased risk of respiratory disease has been variable in the studies reported. Bronchiolar necrosis is the typical lesion seen in BCoV-infected calves (eFig. 5-83). Bronchiolar syncytia have been described in feedlot calves with concurrent BCoV infection and bacterial pneumonia, but the contribution of other viruses, such as bovine respiratory syncytial virus, to these lesions is uncertain. BCoV may be demonstrated using immunohistochemistry or RT-PCR, or isolated using specific rectal tumor cell lines. Further reading Bidokhti MR, et al. Tracing the transmission of bovine coronavirus infections in cattle herds based on S gene diversity. Vet J 2012;193:386-390. Fulton RW, et al. Bovine coronavirus (BCV) infections in transported commingled beef cattle and sole-source ranch calves. Can J Vet Res 2011;75:191-199. Park SJ, et al. Dual enteric and respiratory tropisms of winter dysentery bovine coronavirus in calves. Arch Virol 2007;152:1885-1900. Storz J, et al. Isolation of respiratory bovine coronavirus, other cytocidal viruses, and Pasteurella spp from cattle involved in two natural outbreaks of shipping fever. J Am Vet Med Assoc 2000;216:1599-1604. Storz J, et al. Coronavirus and Pasteurella infections in bovine shipping fever pneumonia and Evans' criteria for causation. J Clin Microbiol 2000;38:3291-3298. Diseases of the Respiratory System In Veterinary Medicine (Eleventh Edition), 2017 Bovine Coronavirus Bovine coronavirus (BoCV) is one of the more newly identified viral respiratory pathogens of cattle, being first described in 1993. As a consequence, the clinical significance of BoCV in bovine respiratory disease, and enzootic pneumonia in particular, is still being determined. The current evidence indicates that BoCV plays a primary and important role in enzootic pneumonia. BoCV was the most commonly identified viral pathogen identified in nasal swabs from calves with respiratory disease in Ireland, being present in 23% of calves.3 BoCV was identified throughout the year, but at a much lower rate in summer.3 BoCV was the only virus detected in approximately 75% of respiratory disease outbreaks in two to 3-month calves in Italy.4 Biology and Diseases of Ruminants (Sheep, Goats, and Cattle) Wendy J. Underwood DVM, MS, DACVIM, ... Adam Schoell DVM, DACLAM, in Laboratory Animal Medicine (Third Edition), 2015 Coronavirus Bovine coronavirus, of the family Coronaviridae, produces a more severe, long-lasting disease compared to rotavirus. Clinical signs in lambs and calves are similar to above, although the incubation period tends to be shorter (20–36 h). In addition, mild respiratory disease may be noted (Janke, 1989). Coronavirus infections may be complicated by parasite infestation (e.g., Cryptosporidia, Eimeria) or bacterial infections (e.g., E. coli, Salmonella). Treatment is aimed at correcting dehydration, electrolyte imbalances, and acidosis. Strict hygiene and effective passive transfer by developing good colostrum-management protocols are critical. Bovine vaccines are available both for delivery to pre-partum dams and for the neonate. Rotaviruses, coronavirus, and adenoviruses affect neonatal goats; however, little has been documented on the pathology and significance of these agents in this age group. Unlike calves, it appears that bacteria play a more important role in neonatal kid diarrheal diseases than in neonatal calf diarrheas. Parvovirus and BVDV also may cause diarrhea in neonatal calves. Viral Diseases of the Bovine Respiratory Tract Robert W. Fulton, in Food Animal Practice (Fifth Edition), 2009 BOVINE CORONAVIRUS Bovine coronaviruses (BCVs) were initially associated with neonatal calf diarrhea.15 Then BCVs were identified with “winter dysentery” in adult dairy cattle.7,15 Later BCVs were detected in respiratory secretions of infected calves with subsequent isolation from cattle with BRD signs. This isolation of BCVs from calves with “shipping fever” pneumonias led to the assumption that BCVs were a major etiology for BRD. In some studies other agents such as BRSV, BVDV, and PI-3V along with bacteria were also found in these severely ill cattle. No doubt BCVs are found in conjunction with other respiratory tract infections, yet their sole or primary BRD role has not been clearly established. Including BCV along with other bovine respiratory tract viruses contributing to BRD is best. Clearly, experimental reproduction of detectable and severe respiratory tract disease such as pneumonia would better make the case for BCV as a significant primary pathogen in respiratory tract disease in cattle. Etiology/Epidemiology BCVs are RNA viruses of the viral family Coronaviridae.6,15 They are enveloped viruses, thus sensitive to disinfectants and the environment. It is not unexpected that cattle would have a coronavirus with tropism for the respiratory tract. Coronaviruses infect the respiratory tract of other species including humans, pigs, turkeys, and chickens. BCV infections in cattle are worldwide. Initially implicated in neonatal calf diarrhea, BCVs were also reported with etiology in “winter dysentery” of adult cattle. Subsequently BCVs have been isolated from the nasal samples of cattle undergoing respiratory tract disease.62-66 Thus this virus has a purported role in both respiratory tract disease and enteric diseases. Only one serotype is recognized, but likely there is some antigenic variability.15 The dilemma for working with BCV experimentally and diagnostic laboratories attempting to isolate the virus is that BCV replicates poorly or is quite difficult to isolate in standard cell cultures. A specialized cell line, a human rectal adenocarcinoma line, is permissive for BCV and has been used for virus isolation from feces and nasal swabs by selected laboratories. The BCV is considered relatively common in enteric infections in both beef and dairy operations. The virus has been isolated from cells with disease including calf pneumonias, as well as beef cattle entering feedlots in various U.S. regions. The BCV was isolated from both healthy and sick cattle in these BRD episodes. And BCV was detected by seroconversions during the first month in feedlots in transported cattle. Clinical Disease The association of BCV with BRD has been primarily by the isolation of virus from nasal swabs of cattle with BRD signs and seroconversions to BCV. The virus has been found in healthy calves as well. Likewise, antibody testing has detected seroconversions in cattle in BRD cases. The clinical signs in the BRD cases are not unlike other BRD cases with viral etiologies present such as BVDV; PI-3V; BRSV; and other viruses with fever, nasal and ocular discharges, anorexia, and coughing. Typically these BCV isolations and seroconversions occur soon after arrival to the feedlot. As expected there is often involvement of secondary bacteria such as M. haemolytica and/or P. multocida. Attempts have been made to demonstrate the pathogenicity of BCV for the bovine respiratory tract. After experimental challenge in young calves, the virus could be found in feces of diarrheic calves and nasal swabs for up to 5 days.15 Respiratory disease signs occurred in only a few calves. Lesions of emphysema and interstitial pneumonia were evident in only a few calves.67,68 For other studies, there are mixed reports of BCV detected in lung tissues of cattle with BRD, one report with no BCV detection in lungs of cattle with BRD,6 and another detecting BCV antigen by immunofluorescence in respiratory tissues.69 Diagnosis The virus can be isolated in cell culture provided that a unique cell culture is available to the diagnostic laboratory, the human rectal adenocarcinoma line (HRT-18).15 The nasal swabs collected appear to be the choice of collections from live cattle for testing. An antigen capture ELISA originally used for detecting BCV antigen in fecal samples is also used by some diagnostic laboratories for BCV detection in respiratory disease samples. Also, BCV immunofluorescence is available to detect BCV antigen. Selected diagnostic laboratories and research units have used PCR to detect BCV in diagnostic samples. Use of electron microscopy could detect BCV in respiratory samples similar to the use of EM for fecal samples. Selected research laboratories have used ELISA tests for BCV antibodies, and in some selected studies they found seroconversions when paired samples were available. Clinicians should consult with their respective diagnostic laboratory for their testing for BCV. Prevention and Control Although there are licensed BCV vaccines for enteric disease protection, there are no licensed BCV vaccines in the United States to control respiratory tract disease in cattle. Treatment focuses on the use of antimicrobials to control the bacterial secondary infections. As in prevention of the neonatal enteric disease, it is assumed that adequate colostrum is available to provide protection in the young calf. Diseases of the Alimentary Tract–Ruminant In Veterinary Medicine (Eleventh Edition), 2017 Synopsis Etiology Bovine coronavirus. Epidemiology Northern climates. Adult lactating dairy cows, usually during winter months when housed. Immunity develops and lasts variable periods. High morbidity with outbreaks; low mortality. Transmitted by fecal–oral route. Signs Sudden onset of diarrhea affecting almost entire herd within several days. Mild fever, decline in milk production, inappetence. Recover in few days. Some coughing. Clinical pathology None routinely. Lesions Crypt atrophy on intestinal mucosa; enterocolitis. Diagnostic confirmation Detection of virus in feces. Serology. Treatment None required. Control No specific control measures available. Hygiene. Minimize overcrowding in dairy housing. Alimentary System Francisco A. Uzal, ... Jesse M. Hostetter, in Jubb, Kennedy & Palmer's Pathology of Domestic Animals: Volume 2 (Sixth Edition), 2016 Bovine coronavirus. In neonatal calves, Bovine coronavirus (BCoV) infection is a common cause of diarrhea, either alone or in combination with other agents, particularly Rotavirus and Cryptosporidium. The disease may be severe in combination with BVDV infection. BCoV is capable of infecting absorptive epithelium in the full length of the small intestine, and in the large bowel. Viral antigen is also found in macrophages in the lamina propria of villi and in mesenteric lymph nodes. In field infections, microscopic lesions are found most consistently in the lower small intestine and colon. Calves with BCoV infection usually develop mild depression, but continue to drink milk despite developing profuse diarrhea. With progressive dehydration, acidosis, and hyperkalemia, the animals become weak and lethargic; death can ensue as a result of hypovolemia, hypoglycemia, and potassium cardiotoxicosis. Diarrhea in survivors resolves in 5-6 days. At autopsy, affected animals have the nonspecific lesions of undifferentiated neonatal calf diarrhea. Rarely, mild fibrinonecrotic typhlocolitis is recognized in calves with coronaviral infection. Mesenteric lymph nodes may be somewhat enlarged and wet. Virus replication is cytocidal and initially occurs throughout the length of the villi in all levels of the small intestine, eventually spreading throughout the large intestine up to the end of the large colon and rectum, causing a malabsorptive diarrhea. Large concentration of BCoV can be typically found in the spiral colon. Infected epithelial cells die, slough off, and are replaced by immature cells. The microscopic lesions of coronaviral infection in calves vary with the severity and duration of the infection; villus atrophy in combination with mild colitis is typical (Fig. 1-114). In the calf small intestine, villus atrophy is rarely as severe as that seen in neonatal swine with TGE. Rather, villi are moderately shortened, or have subtotal atrophy with stumpy, club-shaped, or pointed tips, and villus fusion may be common. In the early phase of the clinical disease, villi are often pointed and covered by cuboidal to squamous epithelium. Exfoliation of epithelium and microerosion may be evident. Later, the epithelium is cuboidal to low columnar, basophilic, with irregular nuclear polarity and an indistinct brush border. Cryptal epithelium is hyperplastic. The lamina propria may contain a moderate infiltrate of mainly mononuclear inflammatory cells, some of which may have pyknotic or karyorrhectic nuclei. In the early stages of infection, necrosis of cells in mesenteric lymph nodes is associated with viral replication. Peyer's patches in animals examined after 4-5 days of clinical illness often appear involuted, and are dominated by histiocytic cells. Whether this is the result of viral activity or the effect of endogenous glucocorticoids is unclear. In the colon during the early phase of infection, surface epithelium may be exfoliating, flattened, and squamous or eroded in patchy areas. Some colonic glands may be dilated, lined by flattened epithelium and contain exfoliated cells and necrotic debris. A moderate mixed inflammatory reaction is present in the lamina propria, and neutrophils may be in damaged glands or effusing into the lumen through superficial microerosions. Later in infection, some dilated debris-filled colonic glands will remain, but other glands will be lined by hyperplastic epithelium, and the surface epithelium will be restored to a cuboidal or low columnar cell type. Goblet cells are usually relatively uncommon. Colonic lesions may be recognizable in tissues from animals submitted dead, even though postmortem change has obscured changes in the small intestine. Live calves in the early stages of clinical disease are the best subjects for confirmation of an etiologic diagnosis. In calves becoming ill <7 days of age, enterotoxigenic Escherichia coli is the main alternative diagnosis. Rotavirus, Cryptosporidium, and combined infections must be considered in calves 5-15 days of age. Infectious bovine rhinotracheitis, salmonellosis, and bovine viral diarrhea must also be considered. Both salmonellosis and bovine viral diarrhea may be associated with depletion of Peyer's patches and colitis that can be confused with that of coronaviral infection; neither is common in the strictly neonatal age group (<7-14 days of age). Respiratory tract infection also occurs in calves and feeders infected with BCoV. The virus replicates in the epithelium of the nasal turbinates and tracheobronchial tree, and respiratory infection may precede, be concurrent with, or follow enteric infection. Calf pneumonia caused by BRCoV can be observed in calves 6-9 months of age. Affected animals may develop fever, serous to mucopurulent nasal discharge, coughing, tachypnea, and dyspnea. Respiratory infections may play a role in maintaining the virus within a herd, and significant, but poorly characterized, pneumonia has been reported in some experimentally infected calves. In addition, coronaviral infection may predispose to subsequent respiratory bacterial infections or contribute to more severe respiratory disease as part of the shipping fever syndrome. Virus may be identified in tissue or nasal secretions by immunofluorescence or immunohistochemistry. Winter dysentery is a syndrome in adult cattle that has been associated with BCoV in a number of areas around the world. Animals develop blood-tinged diarrhea, nasolacrimal discharge or cough, anorexia, and drop in milk production. Mortality is rare, but may occur. The disease is characterized by a high morbidity rate ranging from 50-100%, but usually low mortality rate, typically <2%. Winter dysentery outbreaks are predominantly seen in young postpartum dairy cows, which then experience a drop of 25-95% in milk production. Occasional cases are also observed in adult dairy and beef cattle. Despite its name, cases of winter dysentery can be observed, albeit infrequently, during the warmer season. The pathophysiologic characteristics of winter dysentery are mostly attributed to lesions of the colonic mucosa. Grossly, the colon of affected animals has linear congestion and hemorrhage along the crests of mucosal folds and there may be a large amount of blood mixed with colonic contents (Fig. 1-115). The histologic lesions are similar to those seen in calves with classical BCoV diarrhea, although they are mostly restricted to the colon with only occasional lesions seen in the terminal small intestine. Large amount of BCoV can be detected in colonic epithelium by immunohistochemistry. Coronaviruses are commonly demonstrated in the feces of cattle with winter dysentery; seroconversions occur, and seroprevalence increases in affected herds. Coronavirus antigen is found in the colonic glands of affected animals, in which there is necrosis and exfoliation of epithelial cells. Certain management practices, notably housing animals in stanchions and use of equipment that handles both manure and feed, have been associated with the development of winter dysentery. Further reading Blanchard PC. Diagnostics of dairy and beef cattle diarrhea. Vet Clin North Am Food Anim Pract 2012;28:443-464. Boileau MJ, et al. Bovine coronavirus associated syndromes. Vet Clin North Am Food Anim Pract 2010;26:123-146. Cho KO, et al. Detection and isolation of coronavirus from feces of three herds of feedlot cattle during outbreaks of winter dysentery-like disease. J Am Vet Med Assoc 2000;217:1191-1194. Heckert RA, et al. Epidemiologic factors and isotype-specific antibody responses in serum and mucosal secretions of dairy calves with bovine coronavirus respiratory tract and enteric tract infections. Am J Vet Res 1991;52:845-851. Kanno T. Bovine coronavirus infection: pathology and interspecies transmission. J Disast Res 2012;7:293-302. Kapil S, et al. Experimental infection with a virulent pneumoenteric isolate of bovine coronavirus. J Vet Diagn Invest 1991;3:88-89. Natsuaki S, et al. Fatal winter dysentery with severe anemia in an adult cow. J Vet Med Sci 2007;69:957-960. Park SJ, et al. Dual enteric and respiratory tropisms of winter dysentery bovine coronavirus in calves. Arch Virol 2007;152:1885-1900. Saif LJ, et al. Winter dysentery in dairy herds: electron microscopic and serological evidence for an association with coronavirus infection. Vet Rec 1991;128:447-449. Smith DR, et al. Epidemiologic herd-level assessment of causative agents and risk factors for winter dysentery in dairy cattle. Am J Vet Res 1998;59:994-1001. Traven M, et al. Experimental reproduction of winter dysentery in lactating cows using BCV—comparison with BCV infection in milk-fed calves. Vet Microbiol 2001;81:127-151. Zhang Z, et al. Application of immunohistochemistry and in situ hybridization for detection of bovine coronavirus in paraffin-embedded, formalin-fixed tissues. J Clin Microbiol 1997;35:2964-2965. Infectious Diseases of the Gastrointestinal Tract Simon F. Peek, ... Kevin J. Cummings, in Rebhun's Diseases of Dairy Cattle (Third Edition), 2018 Etiology Based on seroprevalence studies, the bovine coronavirus (BCoV) responsible for calf diarrhea is quite prevalent in U.S. cattle herds, as is rotavirus. There is much debate among researchers at this point as to whether BCoV isolates obtained from calf and adult diarrhea cases are the same virus or distinct from those that have been incriminated in respiratory disease outbreaks in feedlot and dairy calves. Whether or not there are antigenic or genomic differences in BCoV strains that mediate different organ tropism is similarly unclear. Winter dysentery in adult cattle has been associated with BCoV, and the same strain that causes diarrhea in calves has been used to experimentally create winter dysentery in adult cattle. Therefore, the upper age limit of susceptibility to infection by this agent is apparently longer than traditionally thought. Although not as common as rotavirus as a cause of viral enteritis in dairy calves, coronavirus has been identified in neonatal calf diarrhea outbreaks, especially in the winter months and with mixed infections. A number of studies indicate that clinical disease associated with BCoV in calves is more severe than rotavirus, with higher mortality rates. Affected calves tend to be slightly older than calves infected with pure ETEC or pure rotavirus. They average 7 to 10 days of age at onset, with some observed as late as 3 weeks of age. The virus causes a severe enterocolitis characterized by villous enterocyte destruction in the small intestine and destruction of both ridges and crypts in the large intestine. Maldigestion, malabsorption, and inflammation all contribute to the pathophysiology of coronavirus diarrhea in calves. The virus is cytolytic, and affected villous enterocytes in the small intestine are replaced by cuboidal cells from the crypts, but the colonic lesions leave denuded mucosa in affected areas of the colon. The severity of this damage helps explain why coronavirus enteritis, unlike rotavirus, may cause some flecks of blood to appear in the stool and can kill calves even in a germ-free isolation facility. Thus, in the natural setting, coronavirus enteritis creates a severe clinical diarrhea and can also be associated with > 50% mortality when combined with other viral, bacterial and C. parvum infections.

|
Scooped by
Gilbert C FAURE
onto Mucosal Immunity March 8, 2020 4:59 AM
|
No comment yet.
Sign up to comment


 Your new post is loading...
Your new post is loading...