Your new post is loading...
 Your new post is loading...

|
Scooped by
Gilbert C FAURE
December 16, 2013 2:45 AM
|
Resources for DIU Immunologie et Biothérapies
DIU Immunologie et Biotherapies is a french diploma associating french universities and immunology laboratories. It is dedicated to the involvement of immunology in new biotherapies, either molecular or cellular.

|
Scooped by
Gilbert C FAURE
June 5, 1:22 PM
|
China is moving stem-cell “anti-aging” into formal clinical research.
Ruibosheng is China’s first approved mesenchymal stem-cell drug. It was originally conditionally approved for steroid-refractory acute GVHD, a severe immune complication after bone-marrow transplantation.
The therapy is priced at RMB 19,800 per infusion, roughly US$2,700–2,900. A full eight-dose course costs around US$22,000–23,000.
Now, PLA General Hospital, widely known as 301 Hospital and one of China’s most influential national medical centers, is studying the same product in aging and vascular-aging cohorts.
The studies focus on measurable aging-related endpoints, including biological age, multi-omics markers, and vascular stiffness.
Results are still pending.
#Longevity #Healthspan #StemCells #CellTherapy #RegenerativeMedicine #ChinaBiotech #AgingResearch
https://lnkd.in/gNufsMQ8

|
Scooped by
Gilbert C FAURE
May 27, 3:37 AM
|
"In a paper published on 19 February 2025 in Nature, Vinod P Balachandran, MD, David M. Rubenstein Center for Pancreatic Cancer Research, Memorial Sloan Kettering Cancer Center, New York, and colleagues examined the same 16 patients discussed in the 2023 study, 3.2 years after treatment.
The data showed:
* In 8 of 16 patients, cevumeran elicited an immune response up to three years post-administration, measured by activated T cells. The persistence of T cells was associated with a longer median recurrence-free survival in cancer vaccine responders.
* In two of the eight patients who had a strong immune response, their cancer returned.
* Seven of the eight non-responding patients had their cancers return within that 3.2-year window
The still-recruiting phase 2 clinical trial should provide more data about the effectiveness of the vaccine. It is sponsored by Genentech and BioNTech and will include 260 subjects in 14 locations in the USA. The study will compare the endpoints of patients with pancreatic cancer who receive cevumeran plus Atezolizumab and mFOLFIRINOX, two chemotherapy drugs, to patients who receive mFOLFIRINOX alone. The goal of the study is to determine the safety and efficacy of cevumeran, the mRNA vaccine, and to determine if the vaccine improves outcomes compared to chemotherapy alone.
NBC reported that the six patients who were successfully treated with the mRNA vaccine have survived for over six years. Of course, we need larger trials with more subjects to begin to confirm the effectiveness of this vaccine treatment for pancreatic cancer.
All of this is good news; we may have a powerful treatment for a scary cancer. Of course, the anti-vaccine people may block it because the vaccine is mRNA-based."

|
Scooped by
Gilbert C FAURE
May 26, 4:13 AM
|
China’s Stem Cell Wild West Is Coming to an End.
We are witnessing a massive structural shift in Chinese biotech. On May 1, 2026, China officially enforced Decree No. 818, pulling the plug on a multi-billion-dollar unapproved cell therapy market and introducing some of the tightest compliance standards our industry has seen in decades.
For years, a massive regulatory gray market allowed clinics to sell unproven stem cell injections for up to $28,000, while actual manufacturing costs hovered around $400. I've watched how this hyper-profitable, unregulated ecosystem diluted true scientific innovation.
Decree No. 818 completely dismantles this gray zone through three absolute mandates:
1. Restricting all cell therapies exclusively to top-tier Grade 3A hospitals.
2. Imposing an absolute ban on charging patients during clinical research phases.
3. Levying crippling fines up to 20 times illegal profits for non-compliance.
This is not a routine policy tweak. It is an aggressive, much-needed industry purge.
My view is that up to 80% of China's 30,000+ registered cell therapy firms will face immediate elimination. While under-qualified players are already panic-selling equipment in tech hubs like Guangzhou Nansha Medical Valley, compliant, science-backed biotech operations saw their stocks hit daily limits.
The era of selling hype is over. China’s premium longevity and biotech sectors are finally entering a mature, institutional phase.
In this new landscape, survival belongs strictly to those who prioritize clinical evidence over marketing noise.
#ChinaBiotech #CellTherapy #StemCells #BiotechRegulation #HealthcareCompliance

|
Scooped by
Gilbert C FAURE
May 26, 3:44 AM
|
💉 𝐓𝐎𝐏 𝟏𝟎 𝐏𝐎𝐔𝐋𝐓𝐑𝐘 𝐃𝐈𝐒𝐄𝐀𝐒𝐄𝐒 & 𝐓𝐇𝐄𝐈𝐑 𝐕𝐀𝐂𝐂𝐈𝐍𝐄𝐒
Vaccination remains one of the most effective tools in modern poultry health management.
Many major poultry diseases can cause severe economic losses, reduced productivity, and high mortality when preventive vaccination programs are ignored or delayed.
This practical guide highlights some important poultry diseases along with commonly recommended vaccines used in poultry practice.
📌 Includes:
• Newcastle Disease (ND)
• Infectious Bursal Disease (IBD)
• Infectious Bronchitis (IB)
• Fowl Pox
• Marek’s Disease
• Avian Influenza (AI)
• Fowl Cholera
• Infectious Laryngotracheitis (ILT)
• Mycoplasma Gallisepticum (MG)
• Coccidiosis
A strong vaccination program helps:
✅ Reduce disease outbreaks
✅ Improve flock immunity
✅ Increase farm productivity
✅ Reduce mortality losses
✅ Support long-term farm profitability
Healthy flocks begin with prevention.
Save this guide for quick reference and share it with poultry professionals, veterinarians, students, and farm managers.
#Poultry #VeterinaryMedicine #PoultryFarming #AnimalHealth #Vaccination #PoultryHealth #VetLife #FarmManagement #Livestock #DigitalVetHub

|
Scooped by
Gilbert C FAURE
May 23, 5:24 AM
|
The activation of pattern recognition receptors (PRRs) orchestrates inflammation and regulates adaptive immunity. To test whether tuning inflammation through PRR stimulation enhanced the efficacy of mRNA vaccines, we combined an mRNA-based vaccine generated against the ancestral spike protein of SARS-CoV-2 with mannadjuvant, a formulation of fungal mannan and aluminum hydroxide targeting the PRR dectin-2. In mice and non-human primates, mannadjuvant increased the magnitude and durability of the response elicited by the mRNA-based vaccine, and it also led to the induction of neutralizing antibodies directed against variants of concern with a high escape capacity, overcoming antigenic imprinting. Mechanistically, prolonged type I interferon (IFN) production and potentiated interleukin-1 (IL-1) signaling locally within the draining lymph node in mice or in human cells were necessary and sufficient to exert the effect of mannadjuvant. Our data indicate that antifungal PRRs can be harnessed to create more potent and durable mRNA-based vaccines. Jena, Qu et al. show that mannadjuvant, a formulation of fungal mannan and aluminum hydroxide, increases the magnitude, durability and breadth of the response elicited by mRNA-based vaccines against SARS-CoV-2 variants in mice and non-human primates.

|
Scooped by
Gilbert C FAURE
May 21, 10:55 AM
|

|
Scooped by
Gilbert C FAURE
May 17, 4:10 AM
|
A new Science Immunology paper by Yuxi Wei et al. makes an important point for translational immunology and cell therapy:
What we observe ex vivo or in vitro may not fully predict what immune cells can become, or do, in vivo.
The study shows that Foxp3-transduced conventional T cells can acquire endogenous Foxp3 expression, Treg-like transcriptomic and chromatin features, and suppressive function specifically in vivo. This suggests that Foxp3 acts not only as a transcriptional regulator, but as a context-dependent driver of Treg epigenetic identity.
To me, the key message is biological context.
The in vivo environment, including reduced AKT-mTOR signaling and engagement of STAT5 and NF-κB, created permissive conditions for reprogramming that were not fully captured in vitro.
This matters for cell therapy. Phenotype alone is not enough. A cell may express the right markers, or fail to fully mature in simplified culture, but its therapeutic potential may depend on environmental cues, tissue context, and functional integration.
As we develop engineered Tregs, Foxp3-based approaches, and immune-modulating cellular therapies, we need models and assays that help distinguish appearance from function.
The question is not only: “What did we generate?”
It is: “What can this cell become, and what can it do, in the environment where it needs to function?”
Yuxi Wei et al., Science Immunology 2026
DOI: 10.1126/sciimmunol.aed2111
#Immunology #CellTherapy #TranslationalImmunology #Treg #Foxp3 #Epigenetics #Autoimmunity #ImmuneEngineering

|
Scooped by
Gilbert C FAURE
May 15, 11:22 AM
|
26 years after the first approval, the ADC has crossed a structural inflection point: 19 approved agents, 10 in the last 5 years, nearly 50 in late-stage development, and 260+ in the global pipeline.
Bispecific formats, TME-directed non-internalizing conjugates, and dual-payload architectures are converging on a single objective: Decoupling antitumor potency from antigen density (historically suggested threshold: ≥10⁴ antigens per cell) & homogeneity.
Next generation ADC architectures are directed at stroma-dense, antigen-heterogeneous, and pharmacologically shielded tumors previously considered inaccessible including pancreatic, glioblastoma and colorectal.
The Top1i payload class has been pivotal to this pharmacologic expansion: Its comparatively lower intrinsic potency enables higher DARs, broader bystander killing and validated activity in antigen-low tumors, as T-DXd demonstrated in HER2-low breast cancer.
Yet only 0.1% of administered ADC reaches tumor cells due to systemic clearance and restricted access; The remaining 99.9% drives normal tissue exposure and makes toxicity the primary clinical constraint, transforming containment of systemic payload release into the decisive engineering frontier.
So the therapeutic window is no longer solely fixed by the target but engineered through payload-linker-antibody co-optimization.
The next competitive edge belongs to platforms that integrate modularity across all 3 components with biomarker-guided patient stratification. Target-centric development in the absence of this modular foundation faces attrition by volume.
#ADC #BispecificADC #DualPayload #LinkerChemistry #DrugDevelopment #Oncology
https://lnkd.in/eBXqEvze

|
Scooped by
Gilbert C FAURE
May 8, 2:03 AM
|
🔬 Antibody-guided vaccine design is shaping the next generation of immunization. By leveraging epitope-, nanoparticle-, and scaffold-based strategies, researchers aim to achieve more precise and durable immune protection.
Challenges remain across diverse pathogens — from influenza A and HIV to malaria, RSV, and SARS-CoV-2 — yet advances in immunogenetics and personalized vaccines may unlock stronger, more targeted antibody responses.
👉 How do you see the balance between universal and personalized vaccines evolving in the coming decade?
📖 Read the full article here:
https://lnkd.in/dGpXeSB3
#VaccineDesign #Immunology #AntibodyResearch #NextGenVaccines #InfectiousDiseases #BiomedicalResearch #GROWTHresearchgroup #SUMMITproject #velikovamd

|
Scooped by
Gilbert C FAURE
May 4, 7:31 AM
|
A clinical study shows how the intranasal FluMist vaccine can elicit potent antiflu immunity at mucosal sites, researchers tie persistent T cells in the liver to clinical cure of chronic hepatitis B, and more this week in #ScienceTranslationalMedicine.
https://scim.ag/4vY4WbW

|
Scooped by
Gilbert C FAURE
April 24, 7:12 AM
|
A recent publication in Nature Medicine reports 5-year follow-up data from a phase 3 trial evaluating a single-dose live attenuated tetravalent dengue vaccine (Butantan-DV). The results show an overall efficacy of about 65% against symptomatic dengue and around 80% against severe forms, with no safety signal identified over the follow-up period .
An important aspect of dengue immunology needs to be considered when looking at longterm protection. Dengue is characterized by the coexistence of four serotypes. Infection with one serotype induces protection against that specific serotype but may increase the risk of severe disease upon subsequent infection with a different serotype. This phenomenon called antibody-dependent enhancement (ADE), is explained by the impact of non-neutralizing or subneutralizing antibodies that facilitate viral entry into host cells.
A key point is that these potentially enhancing antibodies do not appear immediately after infection or vaccination. They may emerge over time as
antibody levels decline and the balance between neutralizing and non-neutralizing responses shifts. This temporal dynamic is one of the reasons
why long-term follow-up is essential in dengue vaccine studies. The vaccine is designed to induce balanced immunity against all four dengue serotypes and thereby prevent the emergence of enhancing antibodies; however, this remains a theoretical goal, as neutralizing responses are not equally induced against each serotype, which may favor the development of non-neutralizing, potentially enhancing antibodies
In this study, vaccine efficacy remained significant over five years, including in
individuals without prior dengue exposure, and no increase in severe dengue
cases was observed in vaccinated participants . These findings are particularly relevant given previous experience with dengue vaccines, where safety concerns emerged only several years after vaccination in seronegative
individuals.
Overall, these results support the importance of sustained efficacy and long-term safety assessment when evaluating dengue vaccines, in a context where immune responses can evolve over time and influence disease risk upon subsequent exposure.
#Denguevaccine #Butantan
https://lnkd.in/eMg3yrsU

|
Scooped by
Gilbert C FAURE
April 19, 3:59 AM
|
A new order in cell therapy
The interim futility analysis for cema-cel, just gave the lymphoma field something to think about. A 41.6% absolute difference in MRD negativity at Day 45 versus observation, with clean tolerability, availability in community hospitals and most patients managed as outpatients. It is a meaningful signal that allogeneic CAR-T can work in first-line consolidation post R-CHOP.
What about the patients who relapse after cema-cel? Go back to CD19 with an autologous product: Yescarta, Breyanzi? They all target the same CD19 as cema-cel. Would a second CD19 CAR-T be reimbursed? If cema-cel moves towards commercialization in LBCL, patients who relapse will need a subsequent differentiated option. The commercial case for CD19-targeted autologous therapies in this setting will need rethinking.
This is where eti-cel becomes relevant. A highly differentiated product also developed at Cellectis, on the same backbone as cema-cel (originated from Cellectis platform), eti-cel targets both CD20×CD22 simultaneously. Data at the current dose show 88% ORR and 63% CR in heavily pretreated patients, a strong signal for a dual allogeneic approach.
Fifteen years ago, CAR-T emerged as a revolution. The real breakthrough, the bona fide pharmaceutical product, is allogeneic. Off-the-shelf, scalable, standardized. It will establish a new order in cell therapy. Autologous therapies are a process and will, in time, disappear.
On in vivo CAR-T: the science is early, the toxicity profile remains unfavorable today, and the regulatory path for a therapy where the vector is the product is, at best, unclear.
Cellectis Allogene Therapeutics Kite Pharma Bristol Myers Squibb Novartis Johnson & Johnson Gilead Sciences
#dlcl #CART #celltherapy #allogeneic | 14 comments on LinkedIn
|

|
Scooped by
Gilbert C FAURE
June 7, 3:37 AM
|
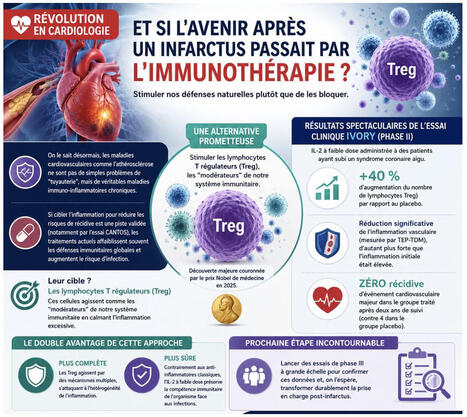
[#PARCC Highlight]: Retour sur une avancée majeure en cardiologie : l’immunothérapie post-infarctus!
On le sait désormais, les maladies cardiovasculaires comme l’athérosclérose ne sont pas de simples problèmes de "tuyauterie", mais de véritables maladies immuno-inflammatoires chroniques.
Si cibler l'inflammation pour réduire les risques de récidive est une piste validée (notamment par l'essai CANTOS), les traitements actuels affaiblissent souvent les défenses immunitaires globales et augmentent le risque d'infection.
🔬 Une alternative prometteuse émerge grâce aux travaux de l’équipe du #PARCC de Alain Tedgui, Ziad Mallat, Soraya Taleb et Hafid Ait-Oufella : stimuler nos propres cellules protectrices plutôt que de bloquer le système immunitaire.
Leur cible ? Les lymphocytes T régulateurs (Treg) — dont la découverte majeure a d'ailleurs été couronnée par le prix Nobel de médecine en 2025. Ces cellules agissent comme les "modérateurs" de notre système immunitaire en calmant l'inflammation excessive.
📈 Les résultats spectaculaires de l'essai clinique IVORY (Phase II) : En administrant de l'interleukine-2 (IL-2) à faible dose à des patients ayant subi un syndrome coronaire aigu, l'équipe a observé :
➡️ +40 % d'augmentation du nombre de lymphocytes Treg par rapport au placebo.
➡️ Une réduction significative de l'inflammation vasculaire (mesurée par TEP-TDM), d'autant plus forte que l'inflammation initiale était élevée.
➡️ Zéro récidive d’événement cardiovasculaire majeur dans le groupe traité après deux ans de suivi.
🛡️ Le double avantage de cette approche :
📈 Plus complète : Les Treg agissent par des mécanismes multiples, s'attaquant à l'hétérogénéité de l'inflammation.
📉 Plus sûre : Contrairement aux anti-inflammatoires classiques, l'IL-2 à faible dose préserve la compétence immunitaire de l'organisme face aux infections.
👉 Prochaine étape incontournable : lancer des essais de phase III à grande échelle pour confirmer ces données et, on l'espère, transformer durablement la prise en charge post-infarctus.
Faculté de Santé - Université Paris Cité INSERM médecine/sciences
👉 Pour approfondir le sujet, découvrez l'analyse complète d'Alain Tedgui et Ziad Mallat dans médecine/sciences: https://lnkd.in/e4zebMjF

|
Scooped by
Gilbert C FAURE
June 5, 11:18 AM
|
Are you in London this Sunday afternoon? Join us to learn about the amazing work on Plant-Powered Vaccines Tsepo Tsekoa Sibongile Mtimka Maabo Moralo Rahwa Osman PhD.Katty Wadda, PhDChanjo Hub Liselo Labs Academy of Medical Sciences
https://lnkd.in/eE-hckpT
See you there 😎 😎 😎

|
Scooped by
Gilbert C FAURE
May 27, 3:34 AM
|
mRNA Vaccines in Poultry: Questions We Must Ask Before the Future Arrives
Q1: Why are mRNA vaccines important for poultry?
Because poultry pathogens such as avian influenza virus and infectious bronchitis virus can evolve rapidly. mRNA platforms may allow faster vaccine redesign once new genetic sequences are identified.
Q2: How do mRNA vaccines work?
They deliver messenger RNA encoding a selected protective antigen. Host cells translate this mRNA into antigen protein, which can stimulate antibody responses, cellular immunity, and immune memory.
Q3: Do mRNA vaccines use live pathogens?
No. They do not require live pathogen replication inside the vaccine product, which may reduce biosafety risks during development and production.
Q4: What diseases could be future poultry targets?
Major candidates include H5/H7 avian influenza, infectious bronchitis virus, Newcastle disease virus, and infectious bursal disease virus.
Q5: Are mRNA vaccines already routine in poultry?
Not yet. They remain mainly experimental or early-stage veterinary technologies. Routine poultry use still requires strong field validation, regulatory approval, and cost-effective manufacturing.
Q6: What are the main advantages?
Rapid redesign, precision antigen targeting, potential DIVA compatibility, reduced need for live-virus culture, and possible induction of humoral and cellular immune responses.
Q7: What are the biggest challenges?
Cost, cold-chain stability, mass delivery, mucosal immunity, reduction of shedding, long-term protection, and performance under real commercial farm conditions.
Q8: What is the real question for the future?
Not only whether mRNA can produce an antigen, but whether it can become affordable, stable, scalable, and protective at the farm level.
The future of poultry vaccination may move from classical vaccine matching toward sequence-based precision immunity.
#Poultry #mRNA #Vaccines #AvianInfluenza #IBV #NDV #IBDV #AnimalHealth #Biosecurity #PrecisionImmunology #OneHealth

|
Scooped by
Gilbert C FAURE
May 26, 3:47 AM
|
Having worked on gene and cell therapies for the past 14 years, it’s been fascinating to help drive the evolution from purely viral approaches (adenoviruses, AAVs, lentiviruses) to non-viral platforms (lipid nanoparticles/liposomes, polymer nanoparticles, electroporation methods). Yet, the fundamental biology and end goal remain the same: delivering nucleic acids to the right cells, while avoiding off-target effects and excessive innate immune activation. The latter is especially tricky, as LNPs and other nanoparticles trigger various anti-viral and tissue damage pathways due to their resemblance with viruses (as depicted in this illustration).
The current momentum behind in vivo CAR-T delivery will put these viral and non-viral platforms into direct competition. Ultimately, the winners of that race won't just be determined by delivery efficiency, but by who can achieve the most precise tissue targeting with an acceptable, manageable safety profile, especially when treating patients with inflammatory and autoimmune diseases.
And, perhaps, the key principles we learn and the limitations we overcome in this space will define how we develop all other in vivo therapeutics in the future.

|
Scooped by
Gilbert C FAURE
May 25, 11:32 AM
|

|
Scooped by
Gilbert C FAURE
May 22, 4:00 AM
|
Global vaccine development.
Abstract
Since the inception of the World Health Organization (WHO) Extended Programme on Immunization in 1974, vaccines have saved the lives of approximately 150 million children, and they continue to save 2–5 million lives each year, according to WHO estimates. The consensus around the benefit and funding of childhood vaccination in low- and middle-income countries has unraveled, and the hard-won gains of the past quarter century are at risk. At the same time, in some high-income countries, vaccine hesitancy—sometimes enabled by government policy pronouncements—has led to reductions in vaccination, resulting in outbreaks of measles in countries that had eliminated this disease. Despite these setbacks, advances in vaccine platform technologies, new immunogens, and new and expanding target populations have the potential to extend the direct and indirect effects of vaccination across the life course. In this Perspective, we discuss how each of these advances could shape the future vaccine landscape. We also highlight cross-cutting issues, including those related to equity, manufacturing, funding and vaccine hesitancy, which make successful implementation more complex.
https://lnkd.in/e38Spimq

|
Scooped by
Gilbert C FAURE
May 19, 4:34 AM
|
Newly published in Gut:
Our updated meta-analysis on hepatocellular carcinoma attributable to HBV and HCV across global, regional, and national levels.
Key message: a large share of HCC remains preventable through HBV vaccination, HBV treatment, and HCV testing and cure, but the burden and incidence attributable to these infections vary markedly by region, highlighting persistent inequities in prevention and control.
Pleasure to have worked with @maomao cao Catherine de Martel Nienke Catharina J. Alberts Feixue Wei Damien Georges
📄 Read here:
https://lnkd.in/em5S3D2D
#HepatitisB #HepatitisC #Hepatology #CancerPrevention

|
Scooped by
Gilbert C FAURE
May 17, 4:05 AM
|
Pediatric CSU just got its first FDA-approved targeted therapy.
The biologic era in A/I is moving fast.
With Dupixent now added to the CSU landscape in children, the treatment map in allergy-immunology keeps expanding across diseases, age groups, and mechanisms.
Here is a clean reference for current FDA-approved targeted therapies in U.S. allergy practice, organized by indication and age.
A few realities stand out:
1️⃣ One drug can now live in multiple disease spaces
2️⃣ Age cutoffs are not a footnote
3️⃣ “Best biologic” is usually the wrong conversation
The better question is:
Which target fits this patient’s disease pattern, biomarkers, comorbidities, age, dosing preference, and access reality.
That is where the real decision-making lives.
Which targeted therapy has changed allergy practice the most in your view? | 12 comments on LinkedIn

|
Scooped by
Gilbert C FAURE
May 11, 7:32 AM
|
𝗔 𝗽𝗹𝘂𝗴-𝗶𝗻 𝘀𝘆𝘀𝘁𝗲𝗺 𝗳𝗼𝗿 𝗺𝘂𝗹𝘁𝗶𝘀𝗽𝗲𝗰𝗶𝗳𝗶𝗰 𝗮𝗻𝘁𝗶𝗯𝗼𝗱𝗶𝗲𝘀
🧠 𝗔 𝗽𝗮𝗽𝗲𝗿 𝗮 𝗱𝗮𝘆 𝗸𝗲𝗲𝗽𝘀 𝗯𝗿𝗮𝗶𝗻 𝗱𝗲𝗰𝗮𝘆 𝗮𝘄𝗮𝘆 🧠
Monday 11 May 2026
𝘉𝘺 𝘦𝘯𝘨𝘪𝘯𝘦𝘦𝘳𝘪𝘯𝘨 𝘢 𝘴𝘱𝘦𝘤𝘪𝘧𝘪𝘤 𝘪𝘯𝘵𝘦𝘳𝘮𝘰𝘭𝘦𝘤𝘶𝘭𝘢𝘳 𝘥𝘪𝘴𝘶𝘭𝘧𝘪𝘥𝘦 𝘣𝘰𝘯𝘥 𝘣𝘦𝘵𝘸𝘦𝘦𝘯 𝘯𝘢𝘵𝘪𝘷𝘦 𝘐𝘨𝘎 𝘍𝘤 𝘢𝘯𝘥 𝘍𝘤γ𝘙𝘐𝘐𝘐𝘢, 𝘳𝘦𝘴𝘦𝘢𝘳𝘤𝘩𝘦𝘳𝘴 𝘩𝘢𝘷𝘦 𝘥𝘦𝘷𝘦𝘭𝘰𝘱𝘦𝘥 𝘢 𝘩𝘪𝘨𝘩𝘭𝘺 𝘴𝘵𝘢𝘣𝘭𝘦, 𝘱𝘭𝘶𝘨-𝘢𝘯𝘥-𝘱𝘭𝘢𝘺 𝘱𝘭𝘢𝘵𝘧𝘰𝘳𝘮 𝘧𝘰𝘳 𝘨𝘦𝘯𝘦𝘳𝘢𝘵𝘪𝘯𝘨 𝘮𝘪𝘹𝘦𝘥-𝘷𝘢𝘭𝘦𝘯𝘤𝘺 𝘮𝘶𝘭𝘵𝘪𝘴𝘱𝘦𝘤𝘪𝘧𝘪𝘤 𝘵𝘩𝘦𝘳𝘢𝘱𝘦𝘶𝘵𝘪𝘤𝘴, 𝘴𝘶𝘤𝘩 𝘢𝘴 𝘢𝘷𝘪𝘥𝘪𝘵𝘺-𝘵𝘶𝘯𝘦𝘥 𝘛-𝘤𝘦𝘭𝘭 𝘦𝘯𝘨𝘢𝘨𝘦𝘳𝘴 𝘢𝘯𝘥 𝘤𝘰𝘯𝘥𝘪𝘵𝘪𝘰𝘯𝘢𝘭𝘭𝘺 𝘢𝘤𝘵𝘪𝘷𝘢𝘵𝘦𝘥 𝘤𝘺𝘵𝘰𝘬𝘪𝘯𝘦𝘴, 𝘵𝘩𝘢𝘵 𝘦𝘯𝘵𝘪𝘳𝘦𝘭𝘺 𝘣𝘺𝘱𝘢𝘴𝘴𝘦𝘴 𝘵𝘳𝘢𝘥𝘪𝘵𝘪𝘰𝘯𝘢𝘭, 𝘤𝘰𝘮𝘱𝘭𝘦𝘹 𝘩𝘦𝘵𝘦𝘳𝘰𝘥𝘪𝘮𝘦𝘳𝘪𝘻𝘢𝘵𝘪𝘰𝘯 𝘤𝘰𝘯𝘴𝘵𝘳𝘢𝘪𝘯𝘵𝘴
💡 𝗧𝗮𝗸𝗲 𝗵𝗼𝗺𝗲 𝗺𝗲𝘀𝘀𝗮𝗴𝗲
• A single Fc A330C–FcR IIIa I106C disulfide creates stable, mammalian-expressed antibody docking complexes without post-expression modification.
• The platform converts IgG antibodies into mixed-valency multispecifics carrying CD3, CD8, or IL-2 payloads.
• Tuned hEGFR2 affinity preserved killing of high-antigen cells while reducing low-antigen activity.
🔥 𝗜𝗺𝗽𝗮𝗰𝘁
• This could simplify multispecific antibody discovery while enabling modular immune engagement, avidity tuning, and conditional cytokine delivery.
❓ 𝗢𝗽𝗲𝗻 𝗾𝘂𝗲𝘀𝘁𝗶𝗼𝗻𝘀
• Does the unpaired cysteine on the second Fc homodimer critically impact long term manufacturing stability?
• Could this disulfide docking mechanism be optimized for FcγRI to exploit distinct therapeutic receptor affinities?
• What maximal stoichiometric payload size can be achieved before spatial constraints compromise immune synapse formation?
𝗚𝗲𝗻𝗲𝗿𝗮𝘁𝗶𝗼𝗻 𝗼𝗳 𝗺𝗶𝘅𝗲𝗱-𝘃𝗮𝗹𝗲𝗻𝗰𝘆, 𝗺𝗼𝗱𝘂𝗹𝗮𝗿 𝗺𝘂𝗹𝘁𝗶𝘀𝗽𝗲𝗰𝗶𝗳𝗶𝗰 𝗮𝗻𝘁𝗶𝗯𝗼𝗱𝗶𝗲𝘀 𝘂𝘀𝗶𝗻𝗴 𝗱𝗶𝘀𝘂𝗹𝗳𝗶𝗱𝗲-𝗹𝗶𝗻𝗸𝗲𝗱 𝗙𝗰–𝗙𝗰γ𝗥 𝗰𝗼𝗺𝗽𝗹𝗲𝘅𝗲𝘀
Miso Park et al.
Nature Communications, May 2026
Corresponding author: John Williams
🔗 𝘓𝘪𝘯𝘬 𝘵𝘰 𝘵𝘩𝘦 𝘱𝘶𝘣𝘭𝘪𝘤𝘢𝘵𝘪𝘰𝘯 𝘪𝘯 𝘤𝘰𝘮𝘮𝘦𝘯𝘵𝘴
𝘐𝘭𝘭𝘶𝘴𝘵𝘳𝘢𝘵𝘪𝘰𝘯 𝘨𝘦𝘯𝘦𝘳𝘢𝘵𝘦𝘥 𝘸𝘪𝘵𝘩 𝘍𝘪𝘨𝘶𝘳𝘦𝘓𝘢𝘣𝘴.
𝘛𝘩𝘦 𝘰𝘱𝘪𝘯𝘪𝘰𝘯𝘴 𝘴𝘩𝘢𝘳𝘦𝘥 𝘰𝘯 𝘓𝘪𝘯𝘬𝘦𝘥𝘐𝘯 𝘢𝘳𝘦 𝘮𝘺 𝘰𝘸𝘯.

|
Scooped by
Gilbert C FAURE
May 5, 9:01 AM
|
Recent advances in koala vaccination against chlamydia are providing valuable insights for human vaccine development. Koalas are the first wildlife species to receive conditional approval for a chlamydia vaccine, enabling researchers to observe real-world disease outcomes and vaccine effectiveness over time. Field studies have demonstrated reduced disease rates and mortality among vaccinated koalas, without evidence of bacterial resistance. This research highlights the potential of wildlife vaccination programs to inform human health strategies, particularly for complex pathogens, and underscores the importance of alternative models when traditional clinical trials are not feasible.

|
Scooped by
Gilbert C FAURE
April 30, 11:39 AM
|
T-cell engaging therapies just took a great leap forward: a single injection of a modified self-amplifying RNA (saRNA) can mediate long-term clearance of malignant B cells, effectively bypassing the need for continuous intravenous infusions.
This new preprint originates from the collaborative research at Boston University. The study was driven by researchers Chloe (Kexin) Li and Joshua McGee, under the leadership of Dr. Wilson Wong and Dr. Mark Grinstaff.
Bispecific T cell engagers (BiTEs), such as the FDA-approved Blinatumomab, are highly effective cancer immunotherapies but are severely bottlenecked by their pharmacokinetics. With a remarkably short half-life of around 2 hours, conventional BiTEs require continuous daily intravenous infusions across multiple weeks, which drastically limits patient access and drives up healthcare costs. Further, the rapid peak of BiTEs can trigger severe adverse events like cytokine release syndrome (CRS). While utilizing mRNA to express BiTEs in vivo circumvents manufacturing hurdles, transient expression still dictates weekly dosing regimens.
To overcome this, the team engineered a 5-methylcytidine (m5C)-modified saRNA-BiTE system formulated in lipid nanoparticles (LNPs) targeting CD19. By leveraging the self-replicating nature of saRNA, the platform acts as an in situ bioreactor, achieving a durable, steady-state pharmacokinetic profile from a single systemic administration.
Key takeaways:
• saRNA-BiTE induces robust, antigen-specific lysis of CD19+ target cells (Nalm6 and Raji). saRNA-BiTE maintains target cell killing for a full week post-transfection across all tested doses, whereas the efficacy of the mRNA-BiTE counterpart rapidly declines over the same period.
• Systemically administered saRNA avoids the initial burst exposure typical of standard mRNA therapies. In murine models, saRNA-encoded proteins maintained higher serum concentrations beyond day 14 compared to mRNA
• To simulate the incomplete leukemic cell depletion often seen in human patients, the team utilized a Nalm6 acute lymphoblastic leukemia (ALL) rechallenge model with 8 successive tumor injections. A single 3 µg dose of saRNA-BiTE completely eradicated malignant cells and prevented disease recurrence for 3 months. By the end of the study, mice treated with mRNA-BiTE and Blinatumomab had all relapsed.
•Ex vivo serum killing assays confirmed that mice treated with saRNA-BiTE maintained functional BiTE expression in circulation for up to 6 to 7 weeks post-administration.
This innovative research establishes saRNA as a highly robust platform for extended in situ therapeutic protein production. By translating continuous BiTE infusions into a single, off-the-shelf LNP injection, this approach mitigates manufacturing bottlenecks associated with complex recombinant proteins.
You can read the cool preprint here: https://lnkd.in/e2M_b2Fy
#BITE #TCE #saRNA #Immunotherapy

|
Scooped by
Gilbert C FAURE
April 22, 9:32 AM
|
Exciting developments for CAR-T in solid tumors "urokinase-type plasminogen activator receptor (uPAR) CAR-T cells"

|
Scooped by
Gilbert C FAURE
April 18, 11:25 AM
|
|






 Your new post is loading...
Your new post is loading...