Your new post is loading...
 Your new post is loading...

|
Scooped by
Gilbert C FAURE
December 16, 2013 2:45 AM
|
Resources for DIU Immunologie et Biothérapies
DIU Immunologie et Biotherapies is a french diploma associating french universities and immunology laboratories. It is dedicated to the involvement of immunology in new biotherapies, either molecular or cellular.

|
Scooped by
Gilbert C FAURE
December 7, 4:12 AM
|
mRNA flu vaccine coming in hot! Look at the separation of those curves 🤩
Bonus points for not needing to be grown in eggs....
https://lnkd.in/eSTKsddY
#influenza #flu #fluvaccine #mRNA

|
Scooped by
Gilbert C FAURE
December 5, 9:53 AM
|
Read the Article now➡️
Immune-Related Adverse Events Associated with Atezolizumab: Insights from Real-World Pharmacovigilance Data
By Connor Frey and Mahyar Etminan
The link: https://bit.ly/4ar94bW

|
Scooped by
Gilbert C FAURE
November 30, 7:38 AM
|
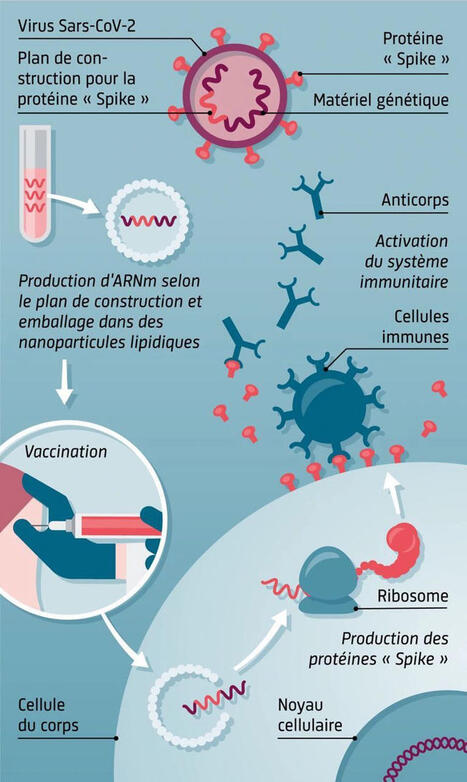
Cette illustration explique le principe des vaccins à ARNm, une technologie innovante qui a joué un rôle majeur dans la réponse contre le SARS-CoV-2.
1. Identifier la cible : la protéine Spike
Le virus SARS-CoV-2 porte une protéine appelée Spike, essentielle pour infecter les cellules humaines.
Les scientifiques récupèrent uniquement le plan génétique permettant de fabriquer cette protéine.
2. Production de l’ARN messager
Ce plan est utilisé pour fabriquer en laboratoire un ARN messager (ARNm) synthétique.
Cet ARNm est ensuite protégé et transporté par des nanoparticules lipidiques.
3. Vaccination
Lors de l’injection, les nanoparticules déposent l’ARNm dans nos cellules.
➡️ l’ARNm ne pénètre pas dans le noyau et ne modifie pas l’ADN.
4. Production de la protéine Spike
Les ribosomes de nos cellules lisent cet ARNm et produisent temporairement la protéine Spike.
Cette protéine seule est inoffensive, mais elle sert d’entraînement au système immunitaire.
5. Activation du système immunitaire
Les cellules immunitaires reconnaissent cette protéine comme étrangère et fabriquent : des anticorps et des lymphocytes mémoire.
Résultat : une protection rapide et efficace
En cas de contact ultérieur avec le vrai virus, le système immunitaire réagit immédiatement, empêchant l’infection ou réduisant sa gravité.

|
Scooped by
Gilbert C FAURE
November 30, 3:31 AM
|
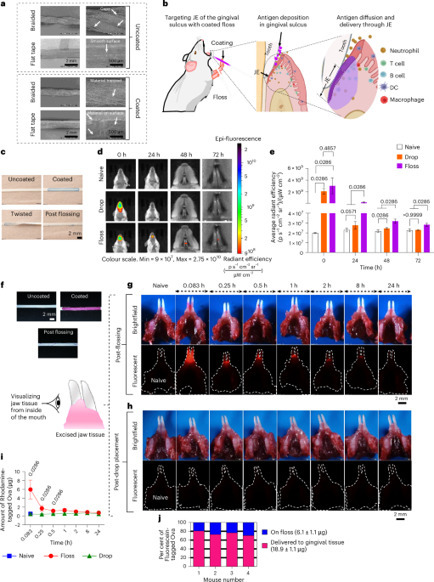
Un vaccin sans aiguille, grâce à du fil dentaire ? C’est possible !
➡️ Une étude récemment publiée dans le journal Nature Biomedical Engineering (Impact Factor 26,6) a montré qu’un fil dentaire enduit d’antigènes pouvait délivrer un vaccin via l’épithélium jonctionnel, induisant une réponse immunitaire forte et durable contre le virus de la grippe. Son efficacité est comparable à la vaccination intranasale.
💡 Pourquoi c’est une révolution ? Vaccination orale, simple, sans aiguille, sans douleur, potentiellement réalisable chez le dentiste ou à la maison.
🔗 L’étude complète ici : https://lnkd.in/dp3a5VUT

|
Scooped by
Gilbert C FAURE
November 29, 5:02 AM
|
✨ Can a new type of immune cell help us fight cancer more safely? Meet CAR-NKT cells.
In November 2024, researchers published a landmark study in Nature Biotechnology introducing a powerful new approach to engineer CAR-NKT cells a next-generation immune therapy designed to target cancer more precisely and with fewer side effects.
🧬 What makes CAR-NKT cells different?
Unlike CAR-T cells, which rely on a strong adaptive immune response, NKT cells are hybrid players part innate, part adaptive. This gives them a unique “dual identity”:
• ⚡ Rapid response to tumors
• 🎯 More targeted killing
• 🛡️ Lower risk of graft-versus-host disease
• 🌐 Potential for “off-the-shelf” manufacturing
The 2024 study demonstrated a new way to produce AlloCAR-NKT cells at scale with high purity, strong tumor control, and sustained activity in preclinical models. This brings us one step closer to universal cell therapies that don’t require personalized manufacturing.
💡 Why should the general public care?
Because this technology could eventually lead to:
• More accessible cell therapies
• Fewer side effects
• Broader applications even beyond cancer
• Faster, more affordable treatment options
We’re not there yet but the progress is real, and the momentum is strong.
The future of cell therapy might not just be smarter… it might finally become accessible to everyone.
What’s your impression of this new wave of immune-engineered therapies?
👇 Let’s talk in the comments.
Li Y-R, Zhou K, Lee D, Zhu Y, Halladay T, Yu J, et al.
Generation of allogeneic CAR-NKT cells from hematopoietic stem and progenitor cells using a clinically guided culture method for cancer immunotherapy.
Nature Biotechnology. 2024; publicado em 14 maio 2024.
#CARNKT #CellTherapy #Immunotherapy #Biotech #FutureOfMedicine #CancerResearch #Innovation #NextGenTherapies

|
Scooped by
Gilbert C FAURE
November 26, 4:21 AM
|
HOW NK CELLS IMPACT ANTIBODY RESPONSE AFTER VACCINATION
Natural killer (NK) cells play a central role in innate immunity, yet their role in the vaccine response has long been marginalized. Several studies have demonstrated NK cell activation following vaccination against influenza, tuberculosis, yellow fever, malaria, and COVID-19. Beyond their cytotoxic function, NK cells are likely to impact the response to vaccination by modulating the adaptive response, in particular by negatively affecting the T-cell response. The present Nature Immunology article also discusses their deleterious effect on the antibody response.
The authors explain how due to different activation of NK cell populations some people develop broad, variant-cross-neutralizing antibodies after SARS-CoV-2 infection — and why others don’t. They showed that
In individuals with poor antibody breadth:
--> NK cells were more abundant and strongly IFN-activated
--> They expressed high levels of ISGs, NKG2D, NKp30, and cytotoxic molecules
--> These NK cells more effectively killed TFH cells, which are essential for affinity maturation
In individuals with broad antibody breadth:
--> NK cells were fewer, less cytotoxic, and showed an immature/proliferative phenotype
--> TFH cells were preserved
The accompanying News & Views emphasizes that high inflammation and IFN drive NK cells into “super-killer” mode, undermining the development of broad antibodies.
#NKcells #Vaccin #COVID19
https://lnkd.in/eCrbwNvv

|
Scooped by
Gilbert C FAURE
November 22, 2:51 AM
|

|
Scooped by
Gilbert C FAURE
November 21, 9:41 AM
|
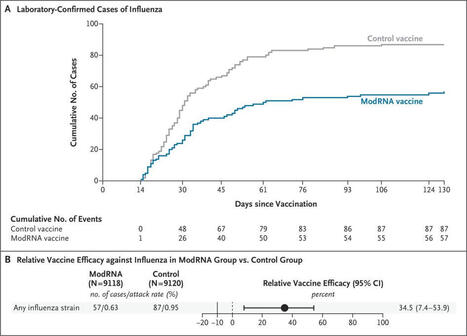
Efficacy, Immunogenicity, and Safety of Modified mRNA Influenza Vaccine | NEJM

|
Scooped by
Gilbert C FAURE
November 19, 6:24 AM
|

|
Scooped by
Gilbert C FAURE
November 16, 12:49 PM
|
Potent Neutralization of Marburg virus by a Vaccine-Elicited Antibody –
Recent study published in Nature describes the characterization of a neutralizing antibody (MARV16) with activity broad activity against Marburg virus (MARV) isolates as well as Ravn virus and Dehong virus.
The fully human antibody was engineered against a mutated version of the prefusion MARV glycoprotein (GP) ectodomain trimer to improve thermostability, and immunogenicity.
MARV16 and previously identified RBS-directed antibodies can bind MARV GP simultaneously. These antibody cocktails require multiple mutations to escape neutralization by both antibodies. The use of MARV GP stabilized antigens may lead to MARV therapeutics resilient to viral evolution.
https://lnkd.in/g_Vw4xA8
#Marburg #antibodies #filovirus #Africa #globalhealth #publichealth #medicine #biotechnology #medicine #pharmaceuticals #FDA #CDA #WHO #NIH

|
Scooped by
Gilbert C FAURE
November 8, 11:23 AM
|
The benefits are observed across patient subtypes and potentially offer the chance to avoid dialysis and implement a new standard of care.

|
Scooped by
Gilbert C FAURE
November 7, 12:37 PM
|
|

|
Scooped by
Gilbert C FAURE
December 7, 4:15 AM
|
#VAXVOICE Javier Casellas MD PID PhD @vaxvoice.org
mRNA-LNP DENV VACCINES
"We previously demonstrated that a mRNA-LNP encoding for DENV-1 prM and Envelope proteins generates homotypic neutralizing immune responses and protects against a lethal challenge. The DENV-1 vaccine avoided ADE by eliminating the fusion loop (FL), one of the dominant ADE epitopes. Using a similar design methodology, here we present mRNA-LNP prM-E vaccines for DENV 2, 3, and 4. These vaccines maintained the ∆FL modifications, but encode a chimeric E protein to improve VLP expression and stability. All three vaccines elicited neutralizing titers of serotype specific antibodies mice and protected against a lethal homotypic DENV challenge. Mutation of the FL lowered ADE in vaccinated mouse sera. We formulated monotypic vaccines into a tetravalent vaccine and evaluated for immunogenicity and protection in mice. The tetravalent vaccine elicited neutralizing humoral responses against all DENV serotypes and protected against lethal challenges. Cumulatively, our monovalent DENV mRNA-LNP vaccines against all DENV serotypes generate protective immunity and lower the potential for ADE, leading to the development of a first-generation ADE-altered tetravalent DENV vaccine. "
https://lnkd.in/dbqUWtXn
Eduardo Eugênio Bittencourt de Gomensoro
Rebecca C. Christofferson

|
Scooped by
Gilbert C FAURE
December 5, 10:23 AM
|
Over the past several decades, the pharmaceutical industry has progressed from identifying small-molecule natural products with favourable pharmacological properties mediated by unknown molecular mechanisms, to deliberate engineering of chemical compounds and macro-molecules that alter the activities of prespecified targets in predefined ways. The past quarter century has seen the emergence of an entirely new drug category: multispecific molecular drugs that are prospectively designed to engage two or more entities to exert their pharmacological effect. This design elicits emergent properties that endow the drugs with capabilities that are inaccessible to monospecific therapies. Furthermore, these properties enable multispecific drugs to circumvent biological barriers to pharmacology, including rapid clearance, functional redundancy, on-target/off-tissue toxicity, and lack of druggable features. Here, I describe how a new wave of approved multispecific drugs is recalibrating expectations of what can be achieved through pharmacotherapy. Multispecific drugs are designed to engage two or more entities to exert their pharmacological effect. This Perspective discusses how a new wave of FDA-approved multispecific molecules have been transformative in overcoming barriers to drug development such as toxicity, rapid clearance, undruggable protein features, and functional redundancy.

|
Scooped by
Gilbert C FAURE
December 2, 4:30 AM
|
Encouraging to see the rapidly evolving translational landscape of CAR-T cell therapy in autoimmune diseases highlighted in Nature. The field is progressing from first-in-human studies in lupus toward phase I/II trials in rheumatoid arthritis, systemic sclerosis, and myositis - including our ongoing efforts at Charité. These developments point toward a potential shift from continuous immunosuppression to time-limited immune reset with the prospect of sustained, treatment-free remission.
Nature: “They don’t have symptoms”: CAR-T therapies send autoimmune diseases into remission
https://lnkd.in/dc2QEThC #AutoimmuneDiseases #Rheumatology #TranslationalMedicine #Immunotherapy

|
Scooped by
Gilbert C FAURE
November 30, 4:51 AM
|

|
Scooped by
Gilbert C FAURE
November 29, 10:06 AM
|
CAR-T therapy, which engineers a patient’s own immune cells, is showing promising potential to recalibrate dysregulated immunity in autoimmune disease. Early studies report reduced pathogenic B-cell activity and sustained remission in systemic lupus erythematosus and rheumatoid arthritis. By selectively targeting autoreactive pathways rather than broadly suppressing the immune system, CAR-T introduces a more precise, disease-modifying strategy that may ultimately transform current approaches to autoimmune treatment.

|
Scooped by
Gilbert C FAURE
November 27, 3:30 AM
|
🎉 NEW PAPER PUBLISHED 🔬
Over a century after its introduction, the BCG vaccine continues to surprise us. Despite being given to billions of people, we still know remarkably little about what actually happens in the skin and blood in the hours and days right after intradermal BCG vaccination. Our team set out to change that.
In this study, conducted in Guinea-Bissau, we developed and validated a completely new experimental pipeline—bringing cutting-edge “omics” technologies into a difficult low-resource setting—to map the local (skin) and systemic (blood) immunological events following BCG.
Here are some of the most exciting methodological advances:
✨ 1. Spatial transcriptomics on 2 mm skin punch biopsies from healthy volunteers with and without a BCG scar
Using Nature’s 2020 Method of the Year, we assessed gene expression within the actual tissue architecture of the skin. For the first time, we could identify which cell types in the epidermis, dermis, and hypodermis respond to BCG, and how.
✨ 2. Liquid biopsy: Cell-free RNA (cfRNA) Profiling
By capturing cell-free RNA from plasma, we non-invasively traced molecular signals released from the skin into the bloodstream. This offers the prospect of blood-based biomarkers that reflect tissue-level vaccine responses—a major step toward scalable immune monitoring.
✨ 3. Multiomics integration
We combined spatial transcriptomics, whole-blood transcriptomics, epigenetics, proteomics, metabolomics, & advanced computer-vision analysis of dermatoscopic images.
✨ 4. Precision study in a low-resource setting
From dermatoscopic imaging (using a smartphone-based system) to standardized storage/processing workflows (see Fig. 2 in the paper), we demonstrated that highly advanced immunology is possible in West Africa—opening the door for larger trials in populations where BCG’s effects matter most.
✨ 5. Capturing early immune events never previously characterized
By sampling on day 1, 7, & 14, and stratifying participants by presence/absence of pre-existing BCG scars, this project may shed light on why BCG given at birth has such profound survival benefits—and why revaccination responses differ.
Why this matters:
Most new TB vaccines build on BCG, yet the early steps of how BCG “trains” the immune system have never been fully mapped. Understanding these local and systemic events may be important for designing better vaccines—not only against TB, but potentially against a wide range of infections where BCG has shown non-specific (heterologous) benefits.
A huge thank you to our incredible collaborators across Guinea-Bissau, Denmark and Canada. And especially to the participants in Bissau who made this study possible.
If you’re interested in immunology, systems biology, vaccine innovation or (of course) vaccine epidemiology, let me hear your thoughts and/or connect with me here on LinkedIn.
📄 The full paper is available open access (link in comments) - & we have more interesting to come from this project.

|
Scooped by
Gilbert C FAURE
November 23, 5:07 AM
|

|
Scooped by
Gilbert C FAURE
November 22, 2:50 AM
|
While the development of preventive vaccines is hindered by the sporadic nature of IFDs in vulnerable individuals, developing therapeutic vaccines for fungal diseases could find an easier road. In this context, IFDs tend to be chronic, with prolonged courses, which would allow time for vaccination...

|
Scooped by
Gilbert C FAURE
November 21, 4:26 AM
|
In his first interview, Argento gives 'Vanity Fair' an exclusive look into the booming blood industry. From stem cells, exosomes, and vitamin infusions to platelet-rich plasma and adaptogens, it’s all about a bespoke recipe of biology and branding that’s become equal parts science experiment and...

|
Scooped by
Gilbert C FAURE
November 19, 5:43 AM
|

|
Scooped by
Gilbert C FAURE
November 14, 5:54 AM
|
Allogeneic natural killer (NK) cell immunotherapy is emerging as a promising and scalable, off-the-shelf platform for treating relapsed and refractory…

|
Scooped by
Gilbert C FAURE
November 7, 1:31 PM
|
Décidément, l'imagination des inventeurs de #pseudothérapies n'a pas de limites.
Vous n'avez peut-être jamais entendu parler de la micro-immunothérapie, mais honnêtement, c'est un cas d'école.
Aucun fondement scientifique, une organisation et un marketing millimétrés pour faire croire que tout est bien démontré (c'est faux), et beaucoup, beaucoup, beaucoup de clients (je n'ose pas dire patients) plumés, en France mais aussi ailleurs en Europe.
Ce serait drôle (version immuno du foie de canard ultra-dilué) si justement ce n'étaient pas des malades qui étaient ciblés.
Bref, ne passez pas à côté de l'excellente enquête d'Antoine Beau pour L'Express sur ce business lucratif mais méconnu👇
Spoiler : grâce à son article le Conseil national de l'Ordre des médecins la Direction Générale de la Santé - DGS ou encore l'Agence Régionale de Santé (ARS) Île-de-France sont sur le coup.
Pierre de Bremond d'Ars Gascan Hugues Mathieu Molimard Mathieu Repiquet Herve Maisonneuve Association Française pour l'Information Scientifique (AFIS) Dominique Costagliola Stephanie RIST
https://lnkd.in/eARfJrzM

|
Scooped by
Gilbert C FAURE
October 26, 10:09 AM
|
|






 Your new post is loading...
Your new post is loading...