Your new post is loading...
 Your new post is loading...

|
Scooped by
NatProdChem
April 3, 2013 5:44 PM
|
Three prenylated indole alkaloids with a rare anti bicyclo-[2.2.2]diazaoctane core ring (5–7) were isolated from Aspergillus taichungensis. The structures including absolute configurations were elucidated based on NMR, X-ray, and CD methods. (+)-Versicolamides B and C (8–9) which contain a spiro-center, together with seven analogues (7, 10–15), were isolated as photoinduced conversion products of 6. Biological evaluation indicated that 6 and 7 exhibited significant cytotoxicities with IC50 values in the low micromolar range. Shengxin Cai, Yepeng Luan, Xianglan Kong, Tianjiao Zhu, Qianqun Gu, and Dehai Li Org. Lett., Article ASAP DOI: 10.1021/ol400694h Publication Date (Web): April 3, 2013

|
Scooped by
NatProdChem
April 3, 2013 6:20 AM
|
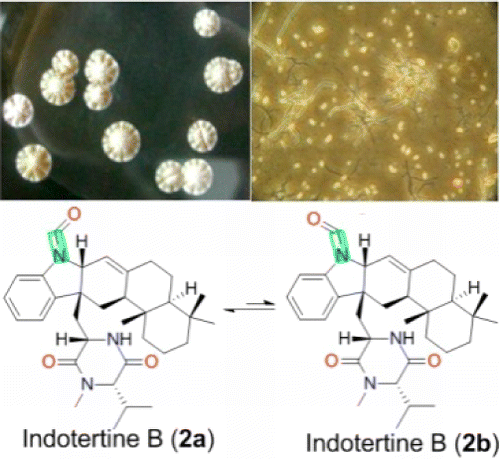
Two new hybrid isoprenoids, named indotertine B (2) and drimentine H (3), along with the known analogue drimentine C (4), were isolated from the reed rhizosphere soil derived actinomycete Streptomyces sp. CHQ-64. The structures of these compounds, including absolute configurations, were elucidated by extensive NMR, MS, and CD analyses. Indotertine B (2) exists as a pair of rotamers about the N–C(O) bond with a 2:1 ratio and displays activities against HCT-8 and A549 tumor cell lines with IC50 values of 6.96 and 4.88 μM, respectively. Qian Che†, Tianjiao Zhu†, Robert A. Keyzers‡, Xiaofang Liu†, Jing Li§, Qianqun Gu†, and Dehai Li J. Nat. Prod., Article ASAP DOI: 10.1021/np3008864

|
Scooped by
NatProdChem
April 1, 2013 8:30 PM
|
Share laboratory techniques, video protocols and advice for your life in science.

|
Scooped by
NatProdChem
March 28, 2013 7:40 AM
|
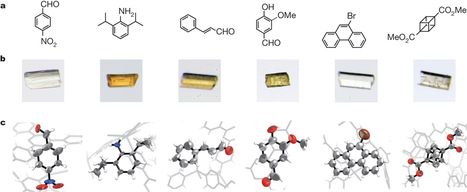
X-ray single-crystal diffraction (SCD) analysis has the intrinsic limitation that the target molecules must be obtained as single crystals. Here we report a protocol for SCD analysis that does not require the crystallization of the sample. In our method, tiny crystals of porous complexes are soaked in a solution of the target, such that the complexes can absorb the target molecules. Crystallographic analysis clearly determines the absorbed guest structures along with the host frameworks. Because the SCD analysis is carried out on only one tiny crystal of the complex, the required sample mass is of the nanogram–microgram order. We demonstrate that as little as about 80 nanograms of a sample is enough for the SCD analysis. In combination with high-performance liquid chromatography, our protocol allows the direct characterization of multiple fractions, establishing a prototypical means of liquid chromatography SCD analysis. Furthermore, we unambiguously determined the structure of a scarce marine natural product using only 5 micrograms of the compound. Yasuhide Inokuma,Shota Yoshioka,Junko Ariyoshi,Tatsuhiko Arai,Yuki Hitora,Kentaro Takada,Shigeki Matsunaga,Kari Rissanen& Makoto Fujita Nature 495,461–466 (28 March 2013) doi:10.1038/nature11990

|
Scooped by
NatProdChem
March 27, 2013 10:06 AM
|
A solvent-free intermolecular Diels–Alder cycloaddition of piperine on silica gel is described. The dimerization of piperine in the presence of silica gel gives better yields than that under the condition of only heating. The new procedure is simple, clean, and easy to operate. It has higher yields and can be completed in short time. Kun Wei, Shunxin Wang,Zhimeng Liu,Yu Du,Xuebo Shi,Ting Qi,Shen Ji Tetrahedron Letters Volume 54, Issue 18, 1 May 2013, Pages 2264–2266 http://dx.doi.org/10.1016/j.tetlet.2013.02.080

|
Scooped by
NatProdChem
March 27, 2013 9:55 AM
|

|
Scooped by
NatProdChem
March 27, 2013 6:23 AM
|
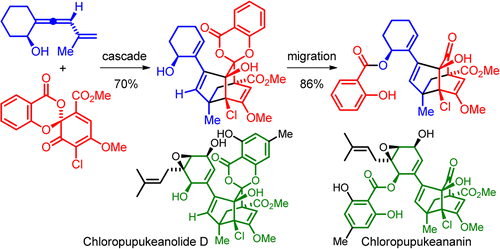
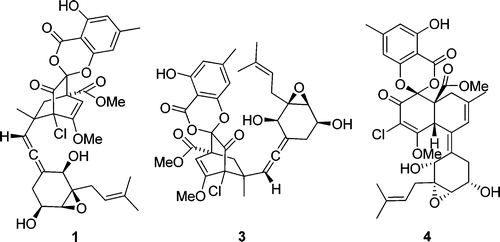
The biomimetic synthesis of the advanced model compound of chloropupukeananin has been achieved. The present synthesis features an unexpected enantiomer-differentiating Diels–Alder/carbonyl-ene cascade under high-pressure conditions and a base-promoted migration of the salicyl group. Takahiro Suzuki*†, Yuria Miyajima†, Kaname Suzuki†, Kanako Iwakiri†, Masaki Koshimizu†, Go Hirai‡, Mikiko Sodeoka‡, and Susumu KobayashiOrg. Lett., Article ASAPDOI: 10.1021/ol400549q

|
Scooped by
NatProdChem
March 25, 2013 5:25 PM
|
Two new prenylated indole alkaloids, 17-epi-notoamides Q and M (1 and 2), and two new phenyl ether derivatives, cordyols D and E (9 and 13), together with 10 known compounds (3–8, 10–12, 14) were isolated from a marine-derived Aspergillus sp. fungus. Among them, 1/5 and 2/4 were pairs of epimers. The planar structures and absolute configurations of the new compounds were determined by extensive NMR spectroscopic data as well as CD spectra. The absolute configuration of 3 was confirmed by single-crystal X-ray diffraction analysis for the first time. All isolated metabolites (1–14) and eight synthetic phenyl ether derivatives (12a, 14a–14g) were evaluated for their antibacterial activities in vitro. The polybromide phenyl ether 14g showed pronounced antibacterial activity against Staphylococcus epidermidis with an MIC value of 0.556 μM, stronger than that of the positive control ciprofloxacin (MIC = 3.13 μM). Min Chen†, Chang-Lun Shao†, Xiu-Mei Fu†, Ru-Fang Xu†, Juan-Juan Zheng†, Dong-Lin Zhao†, Zhi-Gang She*‡, and Chang-Yun WangJ. Nat. Prod., Article ASAPDOI: 10.1021/np300707xPublication Date (Web): March 25, 2013

|
Scooped by
NatProdChem
March 23, 2013 7:17 AM
|
Chloropestolides B–G (1–6), six new metabolites featuring the chlorinated spiro[benzo[d][1,3]dioxine-2,7′-bicyclo[2.2.2]octane]-4,8′-dione (1–3) and spiro[benzo[d][1,3]dioxine-2,1′-naphthalene]-2′,4-dione (4–6) skeletons, and their putative biosynthetic precursor dechloromaldoxin (7) were isolated from the scale-up fermentation cultures of the plant endophytic fungus Pestalotiopsis fici. The structures of 1–7 were determined mainly by NMR experiments. The absolute configurations of 1–3 were deduced by analogy to the previously isolated metabolites from the same fungus (9 and 13–18), whereas those of 4, 5, and 7 were assigned by electronic circular dichroism (ECD) calculations. Structurally, the spiroketal skeletons found in 1–3 and 4–6 could be derived from 2,6-dihydroxy-4-methylbenzoic acid with chlorinated bicyclo[2.2.2]oct-2-en-5-one and 4a,5,8,8a-tetrahydronaphthalen-2(1H)-one, respectively. Biogenetically, compounds 1–6 were derived from the same Diels–Alder precursors as the previously isolated 9 and 12–18. In addition, compounds 2 and 3 were proposed as the biosynthetic intermediates of 17 and 16, respectively. Compound 1 was cytotoxic to three human tumor cell lines. Ling Liu†, Yan Li‡, Li Li§, Ya Cao*, Liangdong Guo†, Gang Liu†, and Yongsheng Che J. Org. Chem., Article ASAP DOI: 10.1021/jo302804h

|
Scooped by
NatProdChem
March 22, 2013 5:52 AM
|
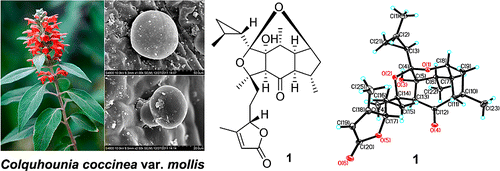
A new class of unique sesterterpenoids, colquhounoids A–C (1–3), were identified from the peltate glandular trichomes of Colquhounia coccinea var. mollis (Lamiaceae) through precise laser-microdissection coupled with UPLC/MS/MS and spectroscopic analyses and X-ray diffraction. Very interestingly, their structural features and defensive function are closely related to leucosceptroid-class sesterterpenoids harbored by the glandular trichomes of another Lamiaceae taxon, Leucosceptrum canum, even though this is morphologically distinct and taxonomically distant. Chun-Huan Li†§, Shu-Xi Jing†§, Shi-Hong Luo†, Wei Shi†§, Juan Hua†, Yan Liu†, Xiao-Nian Li†, Bernd Schneider‡, Jonathan Gershenzon‡, and Sheng-Hong Li*† Org. Lett., Article ASAP DOI: 10.1021/ol4004756 Publication Date (Web): March 21, 2013

|
Scooped by
NatProdChem
March 18, 2013 4:43 PM
|
Microorganisms produce and secrete secondary metabolites to assist in their survival. We report that the gold resident bacterium Delftia acidovorans produces a secondary metabolite that protects from soluble gold through the generation of solid gold forms. This finding is the first demonstration that a secreted metabolite can protect against toxic gold and cause gold biomineralization. Chad W Johnston,Morgan A Wyatt,Xiang Li,Ashraf Ibrahim,Jeremiah Shuster,Gordon Southam& Nathan A Magarvey Nature Chemical Biology 9, 241–243 (2013) doi:10.1038/nchembio.1179

|
Scooped by
NatProdChem
March 18, 2013 4:31 PM
|
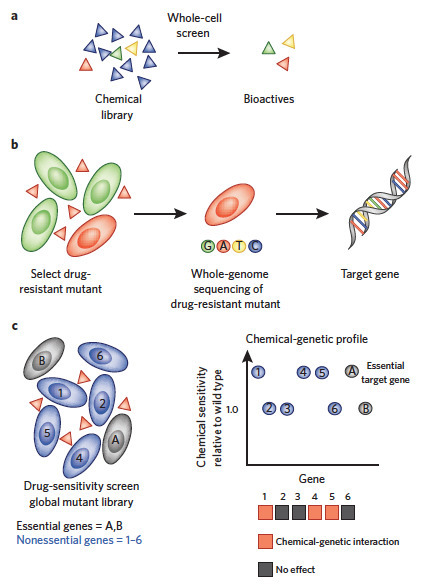
Here, we review the 'target-centric' genomic strategy to antimicrobial discovery and share our perspective on identification, validation and prioritization of potential antimicrobial drug targets in the context of emerging chemical biology, genomics and phenotypic screening strategies. We propose that coupling the dual processes of antimicrobial small-molecule screening and target identification in a whole-cell context is essential to empirically annotate 'druggable' targets and advance early stage antimicrobial discovery. We also advocate a systems-level approach to annotating synthetic-lethal genetic interactions comprehensively within yeast and bacteria models. The resulting genetic interaction networks provide a landscape to rationally predict and exploit drug synergy between cognate inhibitors. We posit that synergistic combination agents provide an important and largely unexploited strategy to 'repurpose' existing chemical space and simultaneously address issues of potency, spectrum, toxicity and drug resistance in early stages of antimicrobial drug discovery. Terry Roemer& Charles Boone Nature Chemical Biology 9, 222–231 (2013) doi:10.1038/nchembio.1205

|
Scooped by
NatProdChem
March 15, 2013 9:11 PM
|
Four new 4-hydroxy-2-pyridone alkaloids, didymellamides A–D (1–4), were isolated from the marine-derived fungus Stagonosporopsis cucurbitacearum. The structures of 1–4 were elucidated from spectroscopic data (NMR, MS, and IR), and the absolute configuration of 1 was determined by X-ray diffraction analysis. Didymellamide A (1) exhibited antifungal activity against azole-resistant Candida albicans. Asami Haga†, Hiroki Tamoto†, Masahiro Ishino†, Eriko Kimura†, Takashi Sugita‡, Kaoru Kinoshita†, Kunio Takahashi†, Motoo Shiro§, and Kiyotaka KoyamaJ. Nat. Prod., Article ASAPDOI: 10.1021/np300876tPublication Date (Web): March 15, 2013
|

|
Scooped by
NatProdChem
April 3, 2013 5:39 PM
|
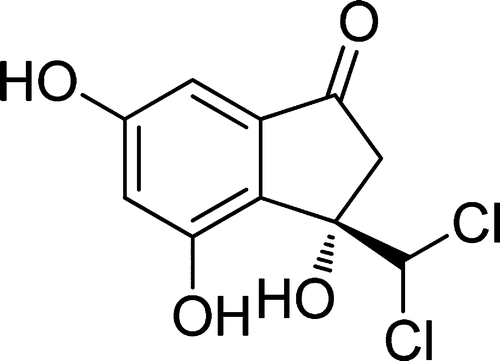
Tripartin (1), a new dichlorinated indanone, was isolated from the culture broth of the Streptomyces sp. associated with a larva of the dung beetle Copris tripartitus Waterhouse. The planar structure of tripartin (1) was identified by the spectroscopic analyses of NMR, mass, UV, and IR data. The structure was confirmed, and the absolute configuration of 1 was determined by X-ray crystallography. Tripartin displayed specific activity as an inhibitor of the histone H3 lysine 9 demethylase KDM4 in HeLa cells. Seong-Hwan Kim†, So Hee Kwon‡, Seon-Hui Park†, Jae Kyun Lee§, Hea-Son Bang, Sang-Jip Nam, Hak Cheol Kwon#, Jongheon Shin†, and Dong-Chan Oh Org. Lett., Article ASAP DOI: 10.1021/ol4004417 Publication Date (Web): April 3, 2013

|
Scooped by
NatProdChem
April 2, 2013 6:16 PM
|
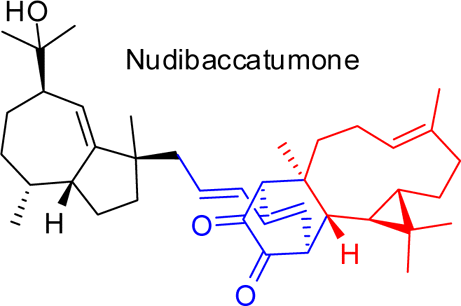
A new complex natural product with a C39 skeleton, named nudibaccatumone, and the known sesquiterpenes (+)-spathulenol, (−)-4β,10α-aromadendranediol, and ent-T-muurolol, as well as the phenylpropanoid hydroxychavicol, were isolated from the aerial parts of Piper nudibaccatum. The structure and absolute configuration of nudibaccatumone were elucidated using spectroscopic methods and ECD calculations. A 1,8-Michael addition reaction and an intermolecular, inverse electron demand Diels–Alder reaction are proposed as the key steps in the biosynthesis of nudibaccatumone. Hong-Xin Liu†‡, Kai Chen§, Qian-Yun Sun, Fu-Mei Yang, Guang-Wan Hu†, Yue-Hu Wang*†, and Chun-Lin LongJ. Nat. Prod., Article ASAPDOI: 10.1021/np300703u

|
Scooped by
NatProdChem
March 28, 2013 7:46 AM
|
Crystalline 'sponges' offer a way to impose order on small molecules so that their structures can be solved by X-ray crystallography. This enables nanogram quantities of material to be analysed using the technique.

|
Scooped by
NatProdChem
March 28, 2013 7:20 AM
|
After nearly 400 years in the slow-moving world of print, the scientific publishing industry is suddenly being thrust into a fast-paced online world of cloud computing, crowd sourcing and ubiquitous sharing. Long-established practices are being challenged by new ones – most notably, the open-access, author-pays publishing model. In this special issue, Nature takes a close look at the forces now at work in scientific publishing, and how they may play out over the coming decades.

|
Scooped by
NatProdChem
March 27, 2013 9:58 AM
|
Grow the next generation of ideas

|
Scooped by
NatProdChem
March 27, 2013 9:54 AM
|
Scientists are beginning to use crowdfunding to support their work, but don’t stop filling out those grant applications just yet. Lauren Kuehne wanted to branch into a new area of research characterizing the soundscape near freshwater lakes. Ethan Perlstein’s postdoctoral fellowship was about to expire, but he was keen to continue his work on the pharmacology of methamphetamine. And Kay Holekamp knew her project to use hyenas as indicators of ecosystem health was important but too applied to get funding from the National Science Foundation (NSF). Faced with research goals that fell between the funding cracks, all of these researchers turned to an emerging fundraising channel—crowdfunding—to get the dollars they needed. Crowdfunding involves asking members of the public to chip in money for projects that interest them, and a number of crowdfunding Web sites now offer researchers a page to pitch their idea, typically including a short video. Much like donating to public radio, backers receive specified perks in return for different levels of support. The best known crowdfunding hub is Kickstarter, which raised over $300 million for over 18,000 projects last year. Kickstarter does not support scientific research projects, but other sites such as Indiegogo and RocketHub have filled the gap. Specialized, research-specific sites such as Microryza, founded by graduates of the University of Washington, Seattle, are also popping up.

|
Scooped by
NatProdChem
March 26, 2013 10:00 AM
|
Five new polyketides, plakortoxides A (1) and B (2), simplextones C (3) and D (4), and plakorsin D (5), together with six known analogues (6–11) were isolated from the South China Sea sponge Plakortis simplex. Their structures were identified by spectroscopic and chemical methods, including NMR, MS, and IR. Experimental and calculated ECD spectra and the modified Mosher’s method were used to determine the absolute configurations. Structurally, both plakortoxides A and B feature a butenolide coupled to an epoxide moiety, while simplextones C and D consist of γ-butyrolactone and cyclopentane moieties, and plakorsin D is a furan acetic acid polyketide. The cytotoxic activities of the isolates were tested, and compounds 8, 10, and 11 showed potent cytotoxicity against both K562 and HeLa tumor cell lines with IC50 values ranging from 0.8 to 5.3 μM. Compound 3 showed significant inhibitory activity against c-Met kinase. Jinrong Zhang†, Xuli Tang‡, Jing Li†, Peifeng Li‡, Nicole J. de Voogd§, Xiaoqin Ni†, Xiaojie Jin, Xiaojun Yao, Pinglin Li*†, and Guoqiang Li J. Nat. Prod., Article ASAP DOI: 10.1021/np300771p

|
Scooped by
NatProdChem
March 25, 2013 5:11 PM
|
A biomimetic synthesis of zeylanone and zeylanone epoxide, which are natural dimeric naphthoquinones, has been accomplished starting from plumbagin, a natural monomeric naphthoquinone. The key features of our synthesis are cascade intermolecular and intramolecular Michael reactions, followed by epoxidation of the resultant hydroquinone with molecular oxygen. Sayako Maruo†, Kazuyuki Nishio†, Takahiro Sasamori‡, Norihiro Tokitoh‡, Kouji Kuramochi*†, and Kazunori Tsubaki Org. Lett., Article ASAP DOI: 10.1021/ol400335s Publication Date (Web): March 25, 2013

|
Scooped by
NatProdChem
March 22, 2013 5:56 AM
|
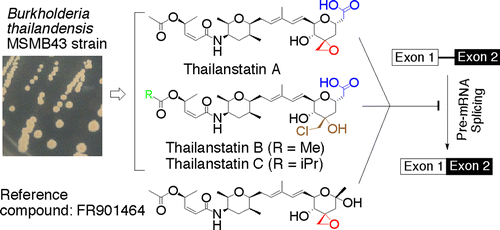
Mining the genome sequence of Burkholderia thailandensis MSMB43 revealed a cryptic biosynthetic gene cluster resembling that of FR901464 (4), a prototype spliceosome inhibitor produced by Pseudomonas sp. No. 2663. Transcriptional analysis revealed a cultivation condition in which a regulatory gene of the cryptic gene cluster is adequately expressed. Consequently, three new compounds, named thailanstatins A (1), B (2), and C (3), were isolated from the fermentation broth of B. thailandensis MSMB43. Thailanstatins are proposed to be biosynthesized by a hybrid polyketide synthase–nonribosomal peptide synthetase pathway. They differ from 4 by lacking an unstable hydroxyl group and by having an extra carboxyl moiety; those differences endow thailanstatins with a significantly greater stability than 4 as tested in phosphate buffer at pH 7.4. In vitro assays showed that thailanstatins inhibit pre-mRNA splicing as potently as 4, with half-maximal inhibitory concentrations in the single to sub-μM range. Cell culture assays indicated that thailanstatins also possess potent antiproliferative activities in representative human cancer cell lines, with half-maximal growth inhibitory concentrations in the single nM range. This work provides new chemical entities for research and development and new structure–activity information for chemical optimization of related spliceosome inhibitors. Xiangyang Liu†‡, Sreya Biswas†, Michael G. Berg§, Christopher M. Antapli†, Feng Xie, Qi Wang, Man-Cheng Tang, Gong-Li Tang, Lixin Zhang, Gideon Dreyfuss§, and Yi-Qiang Cheng J. Nat. Prod., Article ASAP DOI: 10.1021/np300913h Publication Date (Web): March 21, 2013

|
Scooped by
NatProdChem
March 18, 2013 7:02 PM
|
Naomi L. Kuehnbaum and Philip Britz-McKibbin Chem. Rev., Article ASAP DOI: 10.1021/cr300484s Publication Date (Web): March 18, 2013

|
Scooped by
NatProdChem
March 18, 2013 4:35 PM
|
This review focuses on the chemical efforts aimed at achieving
the isolation and total synthesis of specific marine macrolides,
such as macrolides that contain a fused or bridged THF ring, up
to 2012. Macrolides containing a fused THF ring share two
common carbons with the macrolide, while macrolides
containing a bridged THF ring share three or four common
atoms with the macrolide. The revision starts with those
compounds for which only isolation and structure determination
were described and follows with the isolation, structure
determination, and synthesis of the rest of the families. Adriana Lorente†‡, Janire Lamariano-Merketegi†‡, Fernando Albericio†‡§#, and Mercedes Álvarez Chem. Rev., Article ASAP DOI: 10.1021/cr3004778 Publication Date (Web): March 18, 2013

|
Scooped by
NatProdChem
March 15, 2013 9:29 PM
|
Chlorophyll f (1) is the most red-shifted absorbing natural chlorophyll reported, and it is assigned the structure [2-formyl]-chlorophyll a (C55H70O6N4Mg). This structural assignment is confirmed based on the relative retention time on HPLC, mass spectroscopy, UV/vis absorption, and CD spectroscopy, and proton and carbon NMR of chlorophyll f purified from Halomicronema hongdechloris. Org. Lett., Article ASAP Robert D. Willows*†, Yaqiong Li‡, Hugo Scheer§, and Min Chen‡ DOI: 10.1021/ol400327j Publication Date (Web): March 15, 2013
|



 Your new post is loading...
Your new post is loading...