 Your new post is loading...
 Your new post is loading...

|
Scooped by
NatProdChem
April 26, 2013 3:22 PM
|
A lot of people (and I'm one of them) have been throwing the word "epigenetic" around a lot. But what does it actually mean - or what is it supposed to mean? That's the subject of a despairing piece from Mark Ptashne of Sloan-Kettering in a recent PNAS. He noted this article in the journal, one of their "core concepts" series, and probably sat down that evening to write his rebuttal. When we talk about the readout of genes - transcription - we are, he emphasizes, talking about processes that we have learned many details about. The RNA Polymerase II complex is very well conserved among living organisms, as well it should be, and its motions along strands of DNA have been shown to be very strongly affected by the presence and absence of protein transcription factors that bind to particular DNA regions. "All this is basic molecular biology, people", he does not quite say, although you can pick up the thought waves pretty clearly.

|
Scooped by
NatProdChem
April 23, 2013 2:40 PM
|
Five novel tigliane-type diterpenes, stelleracins A–E (3–7), a novel flavanone dimer, chamaeflavone A (8), and six known compounds were isolated from the roots of Stellera chamaejasme. Their structures were elucidated by extensive spectroscopic analyses. The isolated compounds were evaluated for anti-HIV activity in MT4 cells. New compounds 3–5 showed potent anti-HIV activity (EC90 0.00056–0.0068 μM) and relatively low or no cytotoxicity (IC50 4.4–17.2 μM). These new compounds represent promising new leads for development into anti-AIDS clinical trial candidates. Yoshihisa Asada*†, Aya Sukemori‡, Takashi Watanabe§, Kuber J. Malla, Takafumi Yoshikawa‡, Wei Li*, Xinzhu Kuang, Kazuo Koike, Chin-Ho Chen, Toshiyuki Akiyama#, Keduo Qian#, Kyoko Nakagawa-Goto#, Susan L. Morris-Natschke#, Yan Lu#, and Kuo-Hsiung Lee J. Nat. Prod., Article ASAP DOI: 10.1021/np300815t Publication Date (Web): April 23, 2013

|
Scooped by
NatProdChem
April 22, 2013 6:12 PM
|
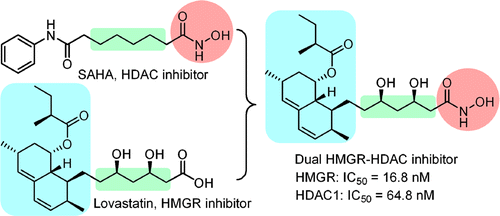
A series of dual-action compounds were designed to target histone deacetylase (HDAC) and 3-hydroxy-3-methylglutaryl coenzyme A reductase (HMGR) by having a hydroxamate group essential for chelation with the zinc ion in the active site of HDAC and the key structural elements of statin for binding with both proteins. In our study, the statin hydroxamic acids prepared by a fused strategy are most promising in cancer treatments. These compounds showed potent inhibitory activities against HDACs and HMGR with IC50 values in the nanomolar range. These compounds also effectively reduced the HMGR activity as well as promoted the acetylations of histone and tubulin in cancer cells, but were not toxic to normal cells. Jhih-Bin Chen†, Ting-Rong Chern‡, Tzu-Tang Wei§, Ching-Chow Chen§, Jung-Hsin Lin*‡#, and Jim-Min Fang J. Med. Chem., Article ASAP DOI: 10.1021/jm400179b

|
Scooped by
NatProdChem
April 20, 2013 1:14 PM
|
French pharma firm set to supply a third of the world's artemisinin next year with photochemical process

|
Scooped by
NatProdChem
April 17, 2013 7:41 AM
|
We report the development and testing of an accurate mass–time (AMT) tag approach for the LC/MS-based identification of plant natural products (PNPs) in complex extracts. An AMT tag library was developed for approximately 500 PNPs with diverse chemical structures, detected in electrospray and atmospheric pressure chemical ionization modes (both positive and negative polarities). In addition, to enable peak annotations with high confidence, MS/MS spectra were acquired with three different fragmentation energies. The LC/MS and MS/MS data sets were integrated into online spectral search tools and repositories (Spektraris and MassBank), thus allowing users to interrogate their own data sets for the potential presence of PNPs. The utility of the AMT tag library approach is demonstrated by the detection and annotation of active principles in 27 different medicinal plant species with diverse chemical constituents. Daniel J. Cuthbertsona, 1,Sean R. Johnsona,Jasenka Piljac-Žegaraca, b,Julia Kappela, c,Sarah Schäfera, c,Matthias Wüstc,Raymond E.B. Ketchuma, 2,Rodney B. Croteaua,Joaquim V. Marquesa,Laurence B. Davina,Norman G. Lewisa,Megan Rolfd,Toni M. Kutchand,D. Doel Soejartoe, f,B. Markus Langea, , Phytochemistry, in Press

|
Scooped by
NatProdChem
April 16, 2013 8:00 AM
|
The Open Chemistry project is a collection of open source, cross platform libraries and applications for the exploration, analysis and generation of chemical data. The project builds upon various efforts by collaborators and innovators in open chemistry such as the Blue Obelisk, Quixote and the associated projects. We aim to improve the state of the art, and facilitate the open exchange of ideas and exchange of chemical data leveraging the best technologies ranging from quantum chemistry codes, molecular dynamics, informatics and visualization.

|
Scooped by
NatProdChem
April 15, 2013 8:47 PM
|
Fumiquinazoline L (1), an alkaloid with a heptacyclic skeleton formed via a bridging hemiaminal linkage, was isolated from a gorgonian-derived Scopulariopsis sp. fungus. The structure and absolute configuration of the new compound were identified by comprehensive spectroscopic data and X-ray diffraction analysis. During acid hydrolysis of 1, the isomerization of the valine residue was observed and also studied in different conditions. Fumiquinazoline L (1) showed no cytotoxic or antibacterial activities. Chang-Lun Shao†, Ru-Fang Xu†, Mei-Yan Wei†‡, Zhi-Gang She*§, and Chang-Yun Wang J. Nat. Prod., Article ASAP DOI: 10.1021/np4002042 Publication Date (Web): April 15, 2013

|
Scooped by
NatProdChem
April 14, 2013 3:06 PM
|
The initial enthusiasm following the discovery of a pharmacologically active natural product is often fleeting due to the poor prospects for its ultimate clinical application. Despite this, the ever-changing landscape of modern biology has a constant need for molecular probes that can aid in our understanding of biological processes. After its initial discovery by Bristol-Myers Squibb as a microbial anti-tumor natural product, epoxomicin was deemed unfit for development due to its peptide structure and potentially labile epoxyketone pharmacophore. Despite its drawbacks, epoxomicin's pharmacophore was found to provide unprecedented selectivity for the proteasome. Epoxomicin also served as a scaffold for the generation of a synthetic tetrapeptide epoxyketone with improved activity, YU-101, which became the parent lead compound of carfilzomib (Kyprolis™), the recently approved therapeutic agent for multiple myeloma. In this era of rational drug design and high-throughput screening, the prospects for turning an active natural product into an approved therapy are often slim. However, by understanding the journey that began with the discovery of epoxomicin and ended with the successful use of carfilzomib in the clinic, we may find new insights into the keys for success in natural product-based drug discovery. Kyung Bo Kim and Craig M. Crews Nat. Prod. Rep., 2013, Advance Article DOI: 10.1039/C3NP20126K

|
Scooped by
NatProdChem
April 14, 2013 11:56 AM
|
Sacchathridine A (1) was isolated from the fermentation broth of strain Saccharothrix sp. MI559-46F5. The structure was determined as a new naphthoquinone derivative with an acetylhydrazino moiety by a combination of NMR, MS spectral analyses, and chemical degradation. Compound 1 showed inhibitory activity of prostaglandin E2 release in a concentration-dependent manner from human synovial sarcoma cells, SW982, with an IC50 value of 1.0 μM, but had no effect on cell growth up to 30 μM. Koichi Nakae*, Ikuko Kurata, Fukiko Kojima, Masayuki Igarashi, Masaki Hatano, Ryuichi Sawa, Yumiko Kubota, Hayamitsu Adachi, and Akio Nomoto J. Nat. Prod., Article ASAP DOI: 10.1021/np3006327 Publication Date (Web): April 12, 2013

|
Scooped by
NatProdChem
April 11, 2013 6:30 PM
|
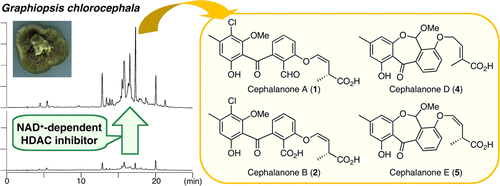
Graphiopsis chlorocephala was separated from the surface-sterilized healthy leaves of Paeonia lactiflora (Paeoniaceae) and cultivated with nicotinamide (an NAD+-dependent HDAC inhibitor). The culture conditions significantly enhanced secondary metabolite production in the fungus and led to the isolation of a structurally diverse set of new benzophenones, cephalanones A–F (1–6), and a known 2-(2,6-dihydroxy-4-methylbenzoyl)-6-hydroxybenzoic acid (7). The structures of 1–6 were determined from NMR data, single crystal X-ray diffraction, and chemical transformations. Teigo Asai*†, Sae Otsuki†, Hiroaki Sakurai‡, Kouwa Yamashita§, Tomoji Ozeki‡, and Yoshiteru Oshima Org. Lett., Article ASAP DOI: 10.1021/ol400781b Publication Date (Web): April 11, 2013

|
Scooped by
NatProdChem
April 10, 2013 6:44 AM
|
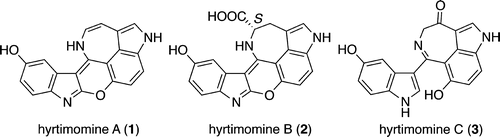
Three new alkaloids, hyrtimomines A–C (1–3), were isolated from an Okinawan marine sponge Hyrtios sp. The structures of 1–3 were elucidated on the basis of spectroscopic analysis and application of a phenylglycine methyl ester (PGME) method. Hyrtimomines A (1) and B (2) are heteroaromatic alkaloids possessing a fused hexacyclic 6/5/6/6/7/5 ring system, while hyrtimomine C (3) is an alkaloid consisting of hydroxyindole and azepino-hydroxyindole moieties. Hyrtimomine A (1) exhibited cytotoxicity against KB and L1210 cells. Rei Momose†, Naonobu Tanaka†, Jane Fromont‡, and Jun’ichi Kobayashi*† Org. Lett., Article ASAPDOI: 10.1021/ol400687bPublication Date (Web): April 9, 2013

|
Scooped by
NatProdChem
April 6, 2013 4:58 PM
|
@ The Sceptical Chymist As a recent comment in Nature pointed out (Lab life: Scientists are snobs), it’s all too easy to get carried away at big multi-session conferences and gravitate towards presentations given by high-flying scientists and those from top institutes. Encountering some down-to-earth research can be a healthy antidote.

|
Scooped by
NatProdChem
April 4, 2013 8:52 PM
|
Ternatusine A (1), a novel alkaloid with an unprecedented epoxyoxepino[4,5-c] pyrrole ring, was isolated from the roots of Ranunculus ternatus Thunb. Its unusual structure, including its absolute stereochemistry, was determined using UV, IR, HRESIMS, and 1D and 2D NMR data and through comparison of the experimental and calculated electronic circular dichroism (ECD) spectra. A possible biosynthetic pathway for ternatusine A was postulated. Zhilai Zhan, Ziming Feng, Yanan Yang, Li Li, Jianshuang Jiang, and Peicheng Zhang Org. Lett., Article ASAP DOI: 10.1021/ol400643q
|

|
Scooped by
NatProdChem
April 23, 2013 3:28 PM
|
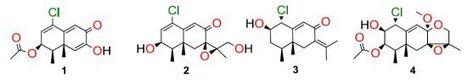
A new chloro-trinoreremophilane sesquiterpene 1, three new chlorinated eremophilane sesquiterpenes 2–4, together with a known compound, eremofortine C (5), were isolated from an Antarctic deep-sea derived fungus, Penicillium sp. PR19N-1. Structures were established using IR, HRMS, 1D and 2D NMR techniques. In addition, the plausible metabolic network of these isolated products is proposed. Compound 1 showed moderate cytotoxic activity against HL-60 and A549 cancer cell lines. Guangwei Wu † , Aiqun Lin † , Qianqun Gu, Tianjiao Zhu* and Dehai Li* Mar. Drugs 2013, 11(4), 1399-1408; doi:10.3390/md11041399

|
Scooped by
NatProdChem
April 22, 2013 6:15 PM
|
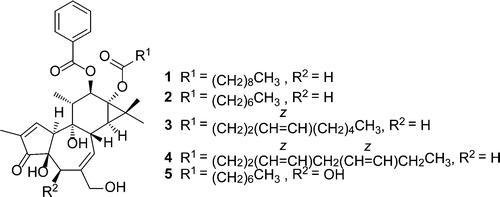
Chemical investigation of the seeds of Croton tiglium afforded eight new phorbol diesters (three phorbol diesters, 1–3, and five 4-deoxy-4α-phorbol diesters, 4–8), together with 11 known phorbol diesters (nine phorbol diesters, 9–17, and two 4-deoxy-4α-phorbol diesters, 18 and 19). The structures of compounds 1–8 were determined by spectroscopic data information and chemical degradation experiments. The cytotoxic activities of the phorbol diesters were evaluated against the SNU387 hepatic tumor cell line, and compound 3 exhibited the most potent activity (IC50 1.2 μM). Xiao-Long Zhang†§, Lun Wang†§, Fu Li†, Kai Yu‡, and Ming-Kui Wang J. Nat. Prod., Article ASAP DOI: 10.1021/np300832n

|
Scooped by
NatProdChem
April 22, 2013 6:09 PM
|
The biosynthetic gene cluster for the pyralomicin antibiotics has been cloned and sequenced from Nonomuraea spiralis IMC A-0156. The 41 kb gene cluster contains 27 ORFs predicted to encode all of the functions for pyralomicin biosynthesis. This includes nonribosomal peptide synthetases (NRPS) and polyketide synthases (PKS) required for the formation of the benzopyranopyrrole core unit, as well as a suite of tailoring enzymes (e.g., four halogenases, an O-methyltransferase, and an N-glycosyltransferase) necessary for further modifications of the core structure. The N-glycosyltransferase is predicted to transfer either glucose or a pseudosugar (cyclitol) to the aglycone. A gene cassette encoding C7-cyclitol biosynthetic enzymes was identified upstream of the benzopyranopyrrole-specific ORFs. Targeted disruption of the gene encoding the N-glycosyltransferase, prlH, abolished pyralomicin production, and recombinant expression of PrlA confirms the activity of this enzyme as a sugar phosphate cyclase involved in the formation of the C7-cyclitol moiety. Patricia M. Flatt, Xiumei Wu, Steven Perry, and Taifo Mahmud* J. Nat. Prod., Article ASAP DOI: 10.1021/np400159aPublication Date (Web): April 22, 2013

|
Scooped by
NatProdChem
April 17, 2013 7:45 AM
|
A phytochemical investigation of the fruits of Daphniphyllum longeracemosum K. Rosenthal led to the isolation of a novel fused-hexacyclic alkaloid, longeracemine (1), along with a new one, demethyl calyciphylline A (2). Their structures were established on the basis of comprehensive spectra associated with quantum chemical analysis. Bioassay firstly indicated that Daphniphyllum alkaloid 1 can stimulate wheat shoot elongation. X.-J. Hao , Yingtong Di and Qiang Zhang RSC Adv., 2013, Accepted Manuscript
DOI: 10.1039/C3RA41072B

|
Scooped by
NatProdChem
April 16, 2013 4:54 PM
|
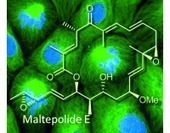
Epoxide “swing”: A family of polyketide macrolactones originating from maltepolide E was discovered in Sorangium cellulosum So ce1485. Their structure was established by NMR experiments, molecular modeling, and X-ray crystallography. Maltepolides induce rather rare morphological changes in the dividing transformed cell lines (see picture in the background). Dr. Herbert Irschik1,Dr. Peter Washausen1,Dr. Florenz Sasse1,Dr. Jörg Fohrer2,Dr. Volker Huch3,Prof. Dr. Rolf Müller1,4,Dr. Evgeny V. Prusov Angewandte Chemie International Edition Article first published online: 16 APR 2013 DOI: 10.1002/anie.201210113

|
Scooped by
NatProdChem
April 16, 2013 7:52 AM
|
A team of researchers has now developed a nuclear magnetic resonance technique that can monitor the activity of this enzyme in real time (Anal. Chem., DOI: 10.1021/ac4004002). Learning more about the mechanism of alanine racemase could lead to new antibiotics, the researchers say. Philippe Lesot of the University of Paris-South, in Orsay, and his colleagues previously reported a way to discriminate between the L- and D-alanine enantiomers using NMR (Chem. Commun., DOI: 10.1039/C1CC15097A). They placed a mixture of the amino acids in an NMR sample tube that included short DNA strands. These strands form chiral liquid crystals that orient the two enantiomers differently within the sample, giving these compounds distinct chemical fingerprints.

|
Scooped by
NatProdChem
April 15, 2013 8:44 PM
|
Five new xanthoquinodins, A4–A6 (1–3), B4 (4), and B5 (5), were isolated from the crude extract of the endolichenic fungal strain Chaetomium elatum (No. 63-10-3-1), along with three known xanthoquinodins, A1–A3 (6–8). Their structures were determined by detailed spectroscopic analysis and comparison of the NMR data with those of the closely related compounds previously reported. The absolute configuration of 1 was established by X-ray crystallographic analysis and ECD calculation. The cytotoxic activity of all compounds was tested against HL-60, SMMC-7721, A-549, MCF-7, and SW480 human cancer cell lines. Guo-Dong Chen†, Ying Chen†, Hao Gao*†, Li-Qing Shen†, Yang Wu†, Xiao-Xia Li†, Yan Li‡, Liang-Dong Guo§, Ying-Zhou Cen, and Xin-Sheng Yao J. Nat. Prod., Article ASAP DOI: 10.1021/np400041y Publication Date (Web): April 15, 2013

|
Scooped by
NatProdChem
April 14, 2013 12:27 PM
|
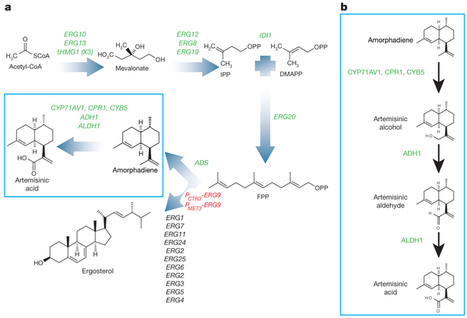
In 2010 there were more than 200 million cases of malaria, and at least 655,000 deaths1. The World Health Organization has recommended artemisinin-based combination therapies (ACTs) for the treatment of uncomplicated malaria caused by the parasite Plasmodium falciparum. Artemisinin is a sesquiterpene endoperoxide with potent antimalarial properties, produced by the plant Artemisia annua. However, the supply of plant-derived artemisinin is unstable, resulting in shortages and price fluctuations, complicating production planning by ACT manufacturers2. A stable source of affordable artemisinin is required. Here we use synthetic biology to develop strains of Saccharomyces cerevisiae (baker’s yeast) for high-yielding biological production of artemisinic acid, a precursor of artemisinin. C. J. Paddon,P. J. Westfall,D. J. Pitera,K. Benjamin,K. Fisher,D. McPhee,M. D. Leavell,A. Tai,A. Main,D. Eng,D. R. Polichuk,K. H. Teoh,D. W. Reed,T. Treynor,J. Lenihan,M. Fleck,S. Bajad,G. Dang,D. Diola,G. Dorin,K. W. Ellens,S. Fickes,J. Galazzo,S. P. Gaucher,T. Geistlingeret al. Nature (2013) doi:10.1038/nature12051

|
Scooped by
NatProdChem
April 14, 2013 11:50 AM
|
Two new alkaloids, polycarpathiamines A and B (1 and 2), were isolated from the ascidian Polycarpa aurata. Their structures were unambiguously determined by 1D, 2D NMR, and HRESIMS measurements and further confirmed by comparison with a closely related analogue, 3-dimethylamino-5-benzoyl-1,2,4-thiadiazole (4), that was prepared by chemical synthesis. Compounds 1 and 2 both feature an uncommon 1,2,4-thiadiazole ring whose biosynthetic origin is proposed. Compound 1 showed significant cytotoxic activity against L5178Y murine lymphoma cells (IC50 0.41 μM). Cong-Dat Pham†, Horst Weber‡, Rudolf Hartmann§, Victor Wray, Wenhan Lin, Daowan Lai*†, and Peter Proksch Org. Lett., Article ASAP DOI: 10.1021/ol400791n Publication Date (Web): April 12, 2013

|
Scooped by
NatProdChem
April 11, 2013 5:51 PM
|
As of early 2013, over 200 natural products are known to contain a nitrogen–nitrogen (N–N) bond. This report categorizes these compounds by structural class and details their isolation and biological activity. Lachlan M. Blair and Jonathan Sperry J. Nat. Prod., Article ASAP DOI: 10.1021/np400124nPublication Date (Web): April 11, 2013

|
Scooped by
NatProdChem
April 10, 2013 6:41 AM
|
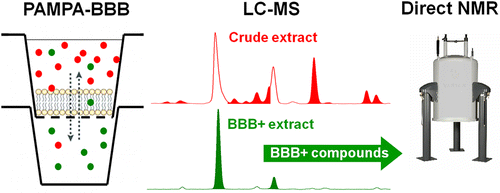
While numerous natural products (NPs) possess activity on central nervous system (CNS) targets, there has been no analytical approach to effectively identify compounds with high brain penetration potential in complex mixtures at the early stage of drug discovery. To overcome this issue, the performance of an in vitro parallel artificial membrane permeability assay for the blood–brain barrier (PAMPA-BBB) for natural products and for plant extracts has been validated and characterized. It was found that the PAMPA-BBB assay preserves its predictive power in the case of natural products and provides high phytochemical selectivity, which enables its use as a unique filtering tool in terms of selecting brain-penetrable compounds from plant extracts. Moreover, the present study has demonstrated that simple modifications in the assay design allow the direct use of PAMPA-BBB filtered samples in a dereplication process, as performed by NMR and LC-MS. The applicability of this procedure was demonstrated using extracts prepared from Tanacetum parthenium, Vinca major, Salvia officinalis, and Corydalis cava, representing different types of chemical diversity and complexity. Thus, the proposed protocol represents a potentially valuable strategy in the NP-based CNS drug discovery environment with a high-throughput screening platform. Árpád Könczöl†, Judit Müller†‡, Emília Földes‡, Zoltán Béni§, Krisztina Végh, Ágnes Kéry, and György T. Balogh J. Nat. Prod., Article ASAP DOI: 10.1021/np300882f Publication Date (Web): April 8, 2013

|
Scooped by
NatProdChem
April 6, 2013 5:51 AM
|
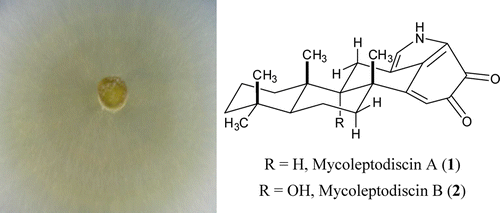
Two novel reddish-orange alkaloids, mycoleptodiscin A (1) and mycoleptodiscin B (2), were isolated from liquid cultures of the endophytic fungus Mycoleptodiscus sp. that had been isolated from Desmotes incomparabilis in Panama. Elucidation of their structures was accomplished using 1D and 2D NMR spectroscopy in combination with IR spectroscopic and MS data. These compounds are indole-terpenes with a new skeleton uncommon in nature. Mycoleptodiscin B (2) was active in inhibiting the growth of cancer cell lines with IC50 values in the range 0.60–0.78 μM. Humberto E. Ortega†, Paul R. Graupner‡, Yumi Asai§, Karen TenDyke, Dayong Qiu, Young Yongchun Shen, Nivia Rios, A. Elizabeth ArnoldΔ, Phyllis D. ColeyO#, Thomas A. KursarO#, William H. Gerwick□, and Luis Cubilla-Rios J. Nat. Prod., Article ASAP DOI: 10.1021/np300792tPublication Date (Web): April 5, 2013
|



 Your new post is loading...
Your new post is loading...






















Derek Lowe discusses semantics of epigenetics.