Your new post is loading...
 Your new post is loading...

|
Scooped by
NatProdChem
March 14, 2013 3:43 PM
|
Two new bromotyrosine alkaloids, pseudoceralidinone A (1) and aplysamine 7 (2), along with three known compounds were isolated from the Australian sponge Pseudoceratina verrucosa. Their structures were characterized by NMR and MS data and the synthetic route. Their cytotoxicity was evaluated against cancer cell lines (HeLa and PC3) and a noncancer cell line (NFF). Trong D. Tran, Ngoc B. Pham, Gregory Fechner, John N. A. Hooper, and Ronald J. Quinn*J. Nat. Prod., Article ASAPDOI: 10.1021/np300648dPublication Date (Web): March 14, 2013

|
Scooped by
NatProdChem
March 13, 2013 1:11 PM
|
High failure rate of front-line therapy puts eradication campaign at risk. One in five people treated for a serious form of leishmaniasis in Nepal relapse after a year. The finding, published in Clinical Infectious Diseases last month1, is “an alarming signal” for campaigns to eliminate the neglected disease, say researchers.

|
Scooped by
NatProdChem
March 13, 2013 12:19 PM
|
One of my colleagues had this to say about blogs: "Never before have so many people with so little to say said so much to so few…" I think there are probably only a few exceptions to that in chemistry (Derek Lowe's blog being an example). Over the years I have vaguely followed some of them, mostly through my students or through being occasionally contacted by someone that runs a blog. Practically all of my colleagues roll their eyes the minute the word "blog" is uttered for a variety of largely justified reasons. But times are clearly changing. I'm not on Facebook, linked in, I don't tweet or chirp, and I don't have time for blogs. I truly don't think I am interesting enough or important that people would care what I think or do every day. And, I barely have time to breathe these days with commitments piling up and a young family. But, I find my students and postdocs to be incredibly interesting and talented. They are brilliant artists, comedians, and wonderful human beings. On top of that they are deeply passionate about chemistry. And they generally have a lot of free time :) I think the world should know them and their inspiring stories - scientific or otherwise - without having to hide behind a cloak of anonymity with the fear that I might find out they are "one of those bloggers".
Last year I was at a dinner symposium where EJ Corey gave a brilliant impromptu talk before a toast. It was a captivating speech all about how things have rapidly changed over the span of his 80+ years. The take home message was that change is natural and you can either embrace it and adapt or be left behind. I'm no fortune teller but it is clear to me that blogging is here to stay and is gathering momentum. The purpose of this blog is to give you a no holds barred, behind the scenes look into our science.
This blog will, hopefully, be like an open flask. --Phil

|
Scooped by
NatProdChem
March 12, 2013 7:20 PM
|
Dichrocephones A (1) and B (2), two new modhephane sesquiterpenoids containing the key core skeleton as that of modhephane sesquiterpenoids rarely encountered in nature were isolated from Dichrocephala benthamii C. B. Clarke. Dichrocephone B (2) contains a new ring system formed by the [3.3.3] propellane core structure fused to a unique oxetane ring with significant cytotoxicity against HeLa, KB and A549 cell lines. Xinhui Tian RSC Adv., 2013, Accepted Manuscript DOI: 10.1039/C3RA23364B

|
Scooped by
NatProdChem
March 11, 2013 4:31 PM
|
A new lipopeptide, ciliatamide D (1), was isolated from a marine sponge Stelletta sp., collected at Oshimashinsone, together with the known compound ciliatamide A (2). Ciliatamide D (1) is a congener of 2, in which N-Me-Phe is replaced by N-Me-Met(O). Marfey’s analysis of the acid hydrolysate of 1 demonstrated that the two constituent amino acids were both in the l-form. This result prompted us to carefully investigate the configuration of 2, resulting in the assignment of the l-form for both residues. Yasufumi Imae, Kentaro Takada, Shigeru Okada, Yuji Ise, Hiroshi Yoshimura, Yasuhiro Morii, and Shigeki Matsunaga J. Nat. Prod., Article ASAP DOI: 10.1021/np300878b Publication Date (Web): March 6, 2013

|
Scooped by
NatProdChem
March 5, 2013 7:45 PM
|
Since the advent of high-throughput screening (HTS) in the early 1990s, a wealth of innovative technologies have been proposed and implemented for the effective localization and characterization of bioactive constituents in complex matrices. The latest developments in this field are reviewed under the perspective of their applicability to natural product-based drug discovery. The approaches discussed here include TLC-based bioautography, HPLC-based assays with on-line, at-line and off-line detection, as well as affinity-based methods, such as frontal affinity chromatography, pulsed ultrafiltration mass spectrometry, imprinted polymers, and affinity capillary electrophoresis. Selected practical examples are given to illustrate the strengths and limitations of these approaches in contemporary natural product lead discovery. In addition, compatibility issues of natural product extracts and HTS are addressed, and selected protocols for the generation of high quality libraries are presented. Olivier Potterat and Matthias Hamburger Nat. Prod. Rep., 2013, Advance Article DOI: 10.1039/C3NP20094A

|
Suggested by
LFNOT
March 4, 2013 8:03 PM
|
The fungal family Clavicipitaceae includes plant symbionts and parasites that produce several psychoactive and bioprotective alkaloids. The family includes grass symbionts in the epichloae clade (Epichloë and Neotyphodium species), which are extraordinarily diverse both in their host interactions and in their alkaloid profiles. Epichloae produce alkaloids of four distinct classes, all of which deter insects, and some—including the infamous ergot alkaloids—have potent effects on mammals. The exceptional chemotypic diversity of the epichloae may relate to their broad range of host interactions, whereby some are pathogenic and contagious, others are mutualistic and vertically transmitted (seed-borne), and still others vary in pathogenic or mutualistic behavior. We profiled the alkaloids and sequenced the genomes of 10 epichloae, three ergot fungi (Claviceps species), a morning-glory symbiont (Periglandula ipomoeae), and a bamboo pathogen (Aciculosporium take), and compared the gene clusters for four classes of alkaloids. Results indicated a strong tendency for alkaloid loci to have conserved cores that specify the skeleton structures and peripheral genes that determine chemical variations that are known to affect their pharmacological specificities.

|
Scooped by
NatProdChem
March 1, 2013 2:50 PM
|
Natural products and related structures are essential sources of new pharmaceuticals, because of the immense variety of functionally relevant secondary metabolites of microbial and plant species. Furthermore, the development of powerful analytical tools based upon genomics, proteomics, metabolomics, bioinformatics and other 21st century technologies are greatly expediting identification and characterization of these natural products. Here we discuss the synergistic and reciprocal benefits of linking these 'omics technologies with robust ethnobotanical and ethnomedical studies of traditional medicines, to provide critically needed improved medicines and treatments that are inexpensive, accessible, safe and reliable. However, careless application of modern technologies can challenge traditional knowledge and biodiversity that are the foundation of traditional medicines. To address such challenges while fulfilling the need for improved (and new) medicines, we encourage the development of Regional Centres of 'omics Technologies functionally linked with Regional Centres of Genetic Resources, especially in regions of the world where use of traditional medicines is prevalent and essential for health. Linh T. Ngo , Joseph I. Okogun and William R. Folk Nat. Prod. Rep., 2013, Advance Article DOI: 10.1039/C3NP20120A

|
Scooped by
NatProdChem
February 27, 2013 10:51 AM
|
Discovering molecular components and their functionality is key to the development of hypotheses concerning the organization and regulation of metabolic networks. The iterative experimental testing of such hypotheses is the trajectory that can ultimately enable accurate computational modelling and prediction of metabolic outcomes. This information can be particularly important for understanding the biology of natural products, whose metabolism itself is often only poorly defined. Here, we describe factors that must be in place to optimize the use of metabolomics in predictive biology. A key to achieving this vision is a collection of accurate time-resolved and spatially defined metabolite abundance data and associated metadata. One formidable challenge associated with metabolite profiling is the complexity and analytical limits associated with comprehensively determining the metabolome of an organism. Further, for metabolomics data to be efficiently used by the research community, it must be curated in publicly available metabolomics databases. Such databases require clear, consistent formats, easy access to data and metadata, data download, and accessible computational tools to integrate genome system-scale datasets. Although transcriptomics and proteomics integrate the linear predictive power of the genome, the metabolome represents the nonlinear, final biochemical products of the genome, which results from the intricate system(s) that regulate genome expression. For example, the relationship of metabolomics data to the metabolic network is confounded by redundant connections between metabolites and gene-products. However, connections among metabolites are predictable through the rules of chemistry. Therefore, enhancing the ability to integrate the metabolome with anchor-points in the transcriptome and proteome will enhance the predictive power of genomics data. We detail a public database repository for metabolomics, tools and approaches for statistical analysis of metabolomics data, and methods for integrating these datasets with transcriptomic data to create hypotheses concerning specialized metabolisms that generate the diversity in natural product chemistry. We discuss the importance of close collaborations among biologists, chemists, computer scientists and statisticians throughout the development of such integrated metabolism-centric databases and software. Manhoi Hur , Alexis Ann Campbell , Marcia Almeida-de-Macedo , Ling Li , Nick Ransom , Adarsh Jose , Matt Crispin , Basil J. Nikolau and Eve Syrkin Wurtele Nat. Prod. Rep., 2013, Advance Article
DOI: 10.1039/C3NP20111B

|
Scooped by
NatProdChem
February 23, 2013 4:20 PM
|
A personal selection of 32 recent papers is presented covering various aspects of current developments in bioorganic chemistry and novel natural products such as pseudolarenone from Pseudolarix amabilis. Robert A. Hill and Andrew Sutherland Nat. Prod. Rep., 2013, Advance Article DOI: 10.1039/C3NP90007J

|
Scooped by
NatProdChem
February 22, 2013 6:41 PM
|
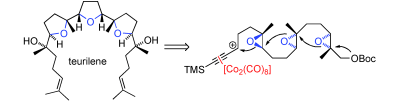
Natural inspiration: Based on the biosynthesis of squalene-derived polyethers, a total synthesis of teurilene is described. The carbocation formation in the Nicholas reaction serves to control the initiation of a polyepoxide ring-opening cascade. The three furan rings present in teurilene were obtained in excellent yield in one step. Boc=tert-butoxycarbonyl, TMS=trimethylsilyl. Julio Rodríguez-López, Dr. Fernando Pinacho Crisóstomo, Dr. Nuria Ortega, Prof. Matías López-Rodríguez, Prof. Víctor S. Martín, Dr. Tomás Martín Angewandte Chemie International Edition Article first published online: 21 FEB 2013 DOI: 10.1002/anie.201209159

|
Scooped by
NatProdChem
February 22, 2013 7:17 AM
|
The absolute configuration (AC) of the bioactive metabolites phyllostin (1) and scytolide (2), two hexahydro-1,4-benzodioxines produced by Phyllosticta cirsii, and oxysporone (3), a dihydrofuropyranone recently isolated from a strain of Diplodia africana, has been assigned by computational analysis of their optical rotatory dispersion (ORD), electronic circular dichroism (ECD), and vibrational circular dichroism (VCD) spectra. Computational prediction of ORD, ECD, and VCD allowed us to assign (3S,4aR,8S,8aR) AC to naturally occurring (−)-1, while (4aR,8S,8aR) AC was assigned to (−)-2 employing only ECD and VCD, because in this case ORD analysis turned out to be unsuitable for AC assignment. Theoretical prediction of both ORD and ECD spectra of 3 led to assignment of (4S,5R,6R) AC to (+)-3. In this case a satisfactory agreement between experimental and calculated VCD spectra was obtained only after taking into account solvent effects. This study shows that in the case of flexible and complex natural products only a concerted application of more than a single chiroptical technique permits unambiguous assignment of absolute configuration. Giuseppe Mazzeo, Ernesto Santoro, Anna Andolfi, Alessio Cimmino, Pavle Troselj, Ana G. Petrovic, Stefano Superchi, Antonio Evidente, and Nina Berova J. Nat. Prod., Article ASAP DOI: 10.1021/np300770s Publication Date (Web): February 21, 2013

|
Scooped by
NatProdChem
February 19, 2013 7:35 PM
|
|

|
Scooped by
NatProdChem
March 13, 2013 6:39 PM
|
When the white smoke puffed up the conclave chimney, all eyes turned to the Vatican. A little while later, Jorge Mario Bergoglio - now Pope Francis* - emerged. His historic election as the first Pope from South America overshadowed another first: he's a chemist.

|
Scooped by
NatProdChem
March 13, 2013 12:30 PM
|
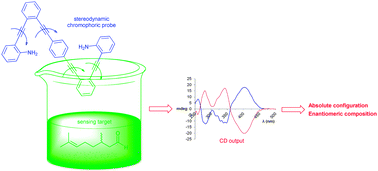
Circular dichroism (CD) spectroscopy is one of the most useful techniques for the stereochemical analysis of chiral biopolymers and fine chemicals. It has become invaluable for the assignment of the absolute configuration, the study of conformational isomers, and the determination of racemization kinetics of CD active chiral compounds. Molecular interactions between a nonracemic chiral substrate and a chromophoric, CD-silent probe that is achiral or exists as a racemic mixture of rapidly interconverting enantiomeric conformations or configurations can induce a strong, characteristic chiroptical readout. A covalent or noncovalent binding event that coincides with a well-defined asymmetric induction process can effectively imprint the chiral information of the substrate on the stereodynamic sensor and thus generate intense Cotton effects in the UV region of the latter. The probe can thus function as a stereochemical reporter unit and analysis of the CD spectrum often provides accurate information about the absolute configuration and enantiomeric composition of the substrate used. In this review, recent developments in circular dichroism analysis of chiral compounds with stereodynamic probes are described and particular emphasis is given to sensor design, chiral induction processes and applications scope. Christian Wolf and Keith W. Bentley Chem. Soc. Rev., 2013, Advance Article
DOI: 10.1039/C3CS35498A

|
Scooped by
NatProdChem
March 13, 2013 12:15 PM
|
Efforts to tackle the threat will need new thinking, but their may be no easy solutions or short cuts to new drugs

|
Scooped by
NatProdChem
March 12, 2013 7:08 PM
|
Six unprecedented bisindole alkaloids, trigolutesins A and B (1–2) with a unique polycyclic skeleton and trigolutes A–D (3–6) with another polycyclic skeleton, were isolated from the twigs of Trigonostemon lutescens. Their structures and relative configurations were elucidated by spectroscopic data and single-crystal X-ray diffraction crystallography. Trigolutesin A (1) showed weak AChE inhibitory activity. Shan-Shan Ma†‡, Wen-Li Mei*†, Zhi-Kai Guo†, Shou-Bai Liu†, You-Xing Zhao†, De-Lan Yang†, Yan-Bo Zeng†, Bei Jiang‡, and Hao-Fu Dai Org. Lett., Article ASAP DOI: 10.1021/ol4002619 Publication Date (Web): March 12, 2013

|
Scooped by
NatProdChem
March 11, 2013 4:24 PM
|
A new pyrroloiminoquinone alkaloid, named atkamine, with an unusual scaffold was discovered from a cold, deep water Alaskan sponge Latrunculia sp. collected from the Aleutian Islands. Olefin metathesis was utilized to determine the location of the double bond in the hydrocarbon chain. The absolute configuration was determined by using computational approaches combing with the ECD (electronic circular dichroism) spectroscopy. Yike Zou and Mark T. Hamann Org. Lett., Article ASAP DOI: 10.1021/ol400294v Publication Date (Web): March 8, 2013

|
Scooped by
NatProdChem
March 5, 2013 7:29 PM
|
(+)-Laburnamine (1), a rare alkaloid extracted from Laburnum anagyroides seeds (4 mg from 1 kg), was shown to bind with high affinity (Ki, 293 nM) to the α4β2 nicotinic receptor subtype, which is, respectively, 126 and 136 times higher than to the α3β4 (Ki 37 μM) and α7 subtypes (Ki 40 μM). When its ability to release [3H]-dopamine from striatal slices was tested in a functional assay, compound 1 behaved as a partial agonist with an EC50 of 5.8 μM and an Emax that was 43% that of nicotine. When incubated with nicotine in the same assay, 1 prevented a maximal effect from being reached. Bruno Tasso*†, Federica Novelli†, Fabio Sparatore†, Francesca Fasoli‡, and Cecilia Gotti‡ J. Nat. Prod., Article ASAP DOI: 10.1021/np3007028 Publication Date (Web): March 5, 2013

|
Scooped by
NatProdChem
March 1, 2013 3:04 PM
|
Where once there was the genome, now there are thousands of ’omes. Nature goes in search of the ones that matter.

|
Scooped by
NatProdChem
February 28, 2013 8:18 AM
|
The marine bacterium Pseudoalteromonas sp. S2B, isolated from the Gulf of Mexico after the Deepwater Horizon oil spill, was found to produce lystabactins A, B, and C (1–3), three new siderophores. The structures were elucidated through mass spectrometry, amino acid analysis, and NMR. The lystabactins are composed of serine (Ser), asparagine (Asn), two formylated/hydroxylated ornithines (FOHOrn), dihydroxy benzoic acid (Dhb), and a very unusual nonproteinogenic amino acid, 4,8-diamino-3-hydroxyoctanoic acid (LySta). The iron-binding properties of the compounds were investigated through a spectrophotometric competition. Hannah K. Zane and Alison Butler* J. Nat. Prod., Article ASAP DOI: 10.1021/np3008655 Publication Date (Web): February 27, 2013

|
Scooped by
NatProdChem
February 26, 2013 8:10 PM
|
Natural product drug discovery programs often rely on the use of silica (Si) gel, reversed-phase media, or size-exclusion resins (e.g., RP-C18, Sephadex LH-20) for compound purification. The synthetic polymer-based sorbent Diaion HP20SS (cross-linked polystyrene matrix) is used as an alternative to prepare purified natural product libraries. To evaluate the impact of chromatographic media on the isolation of biologically active, yet chromatographically unstable natural products, Diaion HP20SS was evaluated side-by-side with normal-phase sorbents for irreversible binding of extract constituents and their effects on bioactivity. An array of chemically diverse natural product-rich extracts was selected as a test panel, and a cell-based reporter assay for hypoxia-inducible factor-1 (HIF-1) was employed to monitor potential change(s) in bioactivity. Silica gel caused significant irreversible binding of three out of 10 extracts. Curcuma longa, Saururus cernuus, and Citrus reticulata extracts showed decreased HIF-1 inhibitory activity after elution through Si gel. An additional nonpolar column wash of HP20SS with EtOAc retained considerable bioactivities of active extracts. In general, Si gel produced the greatest loss of bioactivity. However, HP20SS elution reduced significantly HIF-1 inhibitory activity of certain extracts (e.g., Asimina triloba). Sandipan Datta†, Yu-Dong Zhou*†, and Dale G. Nagle*†§ †Department of Pharmacognosy and §Research Institute of Pharmaceutical Sciences, School of Pharmacy, University of Mississippi, University, Mississippi 38677, United States J. Nat. Prod., Article ASAP DOI: 10.1021/np300858c Publication Date (Web): February 26, 2013

|
Scooped by
NatProdChem
February 23, 2013 6:01 AM
|
Herqueiazole (1), herqueioxazole (2), and herqueidiketal (3), polyaromatic metabolites with a novel skeletal class, were isolated from the marine-derived fungus Penicillium sp. Based on the combined spectroscopic analyses, the structures of 1 and 2 were determined to be the first examples of pyrrole- and oxazole-containing phenalenone compounds, respectively, whereas 3 possessed a novel skeleton with a highly oxidized naphthoquinone moiety. Compound 3 exhibited moderate cytotoxicity and significant inhibitory activity against sortase A. Elin Julianti†, Jung-Ho Lee†, Lijuan Liao†, Wanki Park‡, Sunghyouk Park†, Dong-Chan Oh†, Ki-Bong Oh*‡, and Jongheon Shin*† Org. Lett., Article ASAP DOI: 10.1021/ol4002174 Publication Date (Web): February 22, 2013

|
Scooped by
NatProdChem
February 22, 2013 2:00 PM
|
We report 12 cyanobactin cyclic peptides, the aestuaramides, from the cultivated cyanobacterium Lyngbya aestuarii. We show that aestuaramides are synthesized enzymatically as reverse O-prenylated tyrosine ethers that subsequently undergo a Claisen rearrangement to produce forward C-prenylated tyrosine. These results reveal that a nonenzymatic Claisen rearrangement dictates isoprene regiochemistry in a natural system. They also reveal one of the mechanisms that organisms use to generate structurally diverse compound libraries starting from simple ribosomal peptide pathways (RiPPs). John A. McIntosh, Zhenjian Lin, Ma. Diarey B. Tianero, and Eric W. SchmidtACS Chem. Biol., Article ASAPDOI: 10.1021/cb300614cPublication Date (Web): February 15, 2013

|
Scooped by
NatProdChem
February 19, 2013 8:02 PM
|
Chemistry is progressively unraveling the processes that underlie the evolution of matter towards states of higher complexity and the generation of novel features along the way by self-organization under the pressure of information. Chemistry has evolved from molecular to supramolecular to become adaptive chemistry by way of constitutional dynamics, which allow for adaptation, through component selection in an equilibrating set. Dynamic systems can be represented by weighted dynamic networks that define the agonistic and antagonistic relationships between the different constituents linked through component exchange. Such networks can be switched through amplification/up-regulation of the best adapted/fittest constituent(s) in a dynamic set. Accessing higher level functions such as training, learning, and decision making represent future lines of development for adaptive chemical systems. Prof. Dr. Jean-Marie Lehn Angewandte Chemie International Edition Article first published online: 18 FEB 2013 DOI: 10.1002/anie.201208397
|



 Your new post is loading...
Your new post is loading...




![Dichrocephones A and B, Two Cytotoxic Sesquiterpenoids with the Unique [3.3.3] Propellane Nucleus Skeleton from Dichrocephala benthamii C. B. Clarke | Natural Products Chemistry Breaking News | Scoop.it](https://img.scoop.it/3EwPYhwygyMdfKHeCBd7_Tl72eJkfbmt4t8yenImKBVvK0kTmF0xjctABnaLJIm9)