Your new post is loading...
 Your new post is loading...

|
Scooped by
NatProdChem
May 21, 2013 6:23 AM
|
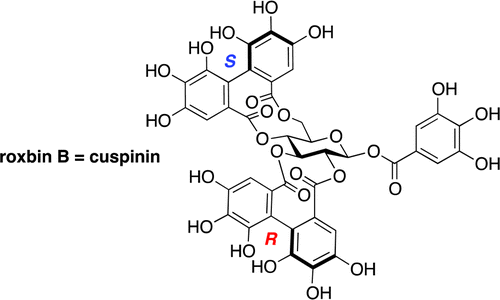
Prompted by the outcome that the synthesized roxbin B was not identical to the natural roxbin B, the structural determination process and spectral data were re-examined, with the finding that roxbin B was very likely to be 1-O-galloyl-2,3-(R);4,6-(S)-bis-O-hexahydroxydiphenoyl-β-d-glucose (cuspinin). Because the (R)-axial chirality is rare in natural products when the hexahydroxydiphenoyl group bridges the 2- and 3-oxygens, the proposed structure of cuspinin was confirmed by the total synthesis, leading to the conclusion that roxbin B is the same as cuspinin. Sayuri Yamaguchi†, Tsukasa Hirokane†, Takashi Yoshida‡, Takashi Tanaka§, Tsutomu Hatano‡, Hideyuki Ito‡, Gen-ichiro Nonaka, and Hidetoshi YamadaJ. Org. Chem., Article ASAPDOI: 10.1021/jo400562k

|
Scooped by
NatProdChem
May 14, 2013 2:15 PM
|
Chromatographic analysis of an ethyl acetate extract of Pestalotiopsis virgatula rice cultures yielded four new α-pyrone derivatives, pestalotiopyrones I–L, and the new (6S,1′S,2′S)-hydroxypestalotin, in addition to three known compounds. The planar structures of the new compounds were elucidated on the basis of extensive NMR spectroscopic and mass spectrometric analyses. The absolute configurations of the new compounds were determined on the basis of biosynthetic considerations, coupling constants, and for pestalotiopyrone L by TDDFT ECD calculations of solution conformers. David Rönsberga,Abdessamad Debbaba,Attila Mándib,Victor Wrayc,Haofu Daid,Tibor Kurtánb,Peter Prokscha,Amal H. Aly Tetrahedron Letters Volume 54, Issue 25, 19 June 2013, Pages 3256–3259 http://dx.doi.org/10.1016/j.tetlet.2013.04.03

|
Scooped by
NatProdChem
May 14, 2013 2:09 PM
|
Mbandakamines A (1) and B (2), isolated from the leaves of an as yet unidentified Congolese Ancistrocladus species, are the first dimeric naphthylisoquinoline alkaloids with an unsymmetrically coupled central biaryl axis. Their novel 6′,1″-coupling type implies a hitherto unprecedented peri–peri coupling in one of the naphthalene parts, leading to the as yet highest steric hindrance at the central axis and a total of seven elements of chirality. Mbandakamine A exhibits good antimalarial activity. Gerhard Bringmann*†, Blaise Kimbadi Lombe†‡, Claudia Steinert†, Karine Ndjoko Ioset†, Reto Brun§, Florian Turini#, Günther Heubl#, and Virima Mudogo Org. Lett., Article ASAP DOI: 10.1021/ol4005883 Publication Date (Web): May 13, 2013

|
Scooped by
NatProdChem
May 13, 2013 10:30 AM
|
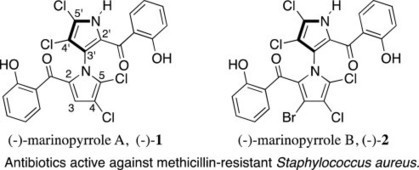
Marinopyrroles A and B are recently isolated and structurally unusual antibiotics that have high activity against methicillin-resistant bacteria. This is an important property because of the very serious1—and increasing—problems associated with drug resistance. We review here the chemistry, synthesis, biosynthesis, and biological activity of these substances, their congeners, and analogs. Derrick L.J. Clive, , Ping Cheng Tetrahedron Volume 69, Issue 25, 24 June 2013, Pages 5067–5078 http://dx.doi.org/10.1016/j.tet.2013.04.036

|
Scooped by
NatProdChem
May 10, 2013 7:51 AM
|
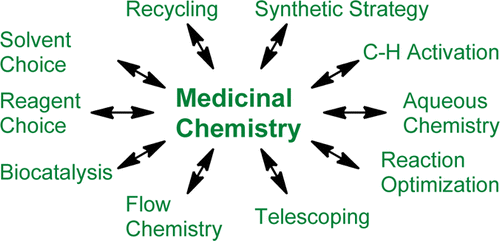
The medicinal chemistry subgroup of the American Chemical Society’s Green Chemistry Institute Pharmaceutical Roundtable (ACS GCI PR) offers a perspective on the current state of environmentally sustainable practices in medicinal chemistry with the aim of sharing best practices more widely and highlighting some potential future developments. Marian C. Bryan†, Barry Dillon‡, Lawrence G. Hamann§, Gregory J. Hughes, Michael E. Kopach, Emily A. Peterson#, Mehrnaz Pourashraf∞, Izzat Raheem×, Paul Richardson, Daniel Richter, and Helen F. Sneddon J. Med. Chem., Article ASAP DOI: 10.1021/jm400250p

|
Scooped by
NatProdChem
May 10, 2013 6:18 AM
|
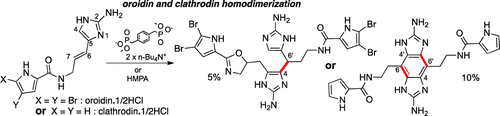
The first biomimetic homodimerization of oroidin and clathrodin was effected in the presence HMPA and diphosphonate salts, strong guanidinium and amide chelating agents. The intermolecular associations probably interfere with the entropically and kinetically favored intramolecular cyclizations. Use of oroidin·1/2HCl salt or clathrodin·1/2HCl was indicative in the presence of the ambident nucleophilic and electrophilic tautomers of the 2-aminoimidazolic oroidin and clathrodin precursors. Surprisingly, the homodimerization of oroidin led to the nagelamide D skeleton, while the homodimerization of clathrodin gave the benzene para-symmetrical structure 19. The common process was rationalized from tautomeric precursors I and III. Clarisse Lejeune, Hua Tian, Jérôme Appenzeller, Ludmila Ermolenko, Marie-Thérèse Martin, and Ali Al-Mourabit J. Nat. Prod., Article ASAP DOI: 10.1021/np400048r Publication Date (Web): May 8, 2013

|
Scooped by
NatProdChem
May 3, 2013 1:34 PM
|
Asperterpenol A (1) and asperterpenol B (2), two novel sesterterpenoids with an unusual 5/8/6/6 tetracyclic ring skeleton, were isolated from a mangrove endophytic fungus Aspergillus sp. 085242. The structures were elucidated on the basis of spectroscopic methods and the absolute configurations determined by single-crystal X-ray diffraction analysis. Compounds 1 and 2 inhibit acetylcholinesterase with IC50 values of 2.3 and 3.0 μM, respectively. Ze’en Xiao†, Huarong Huang†, Changlun Shao†, Xuekui Xia†, Lin Ma†, Xishan Huang†, Yongjun Lu‡, Yongcheng Lin†, Yuhua Long*§, and Zhigang SheOrg. Lett., Article ASAPDOI: 10.1021/ol401005jPublication Date (Web): May 3, 2013

|
Scooped by
NatProdChem
May 3, 2013 1:27 PM
|
An overview of marine natural products synthesis during 2010 is provided. As with earlier installments in this series, the emphasis is on total syntheses of molecules of contemporary interest, new total syntheses, and syntheses that have resulted in structure confirmation or stereochemical assignments. Marine natural products: synthetic aspects Jonathan C. MorrisNat. Prod. Rep., 2013, Advance Article
DOI: 10.1039/C3NP20116C

|
Scooped by
NatProdChem
May 2, 2013 2:47 PM
|
Fungal polyketides and their hybrid non ribosomal peptide derivatives are characterized by often striking structural features and biological activities. Their diversity and their complexity arise from highly organized and programmable biosynthetic pathways and have been challenged by many synthetic chemists. This review will conceptually illustrate how complexity can be generated, starting from a general biosynthetic purpose (the fundaments of PKS-NRPS assembly lines) and finally showing how the particular class of hirsutellone compounds has emerged from such processes in relation to post-elongation and secondary tailoring events. Synthetic efforts to produce these natural products will be described with a special emphasis on complexity-generating strategies and steps. Thus, the biosynthetic and synthetic works will be analyzed in a continuous flow, focusing on both the logic of Nature and organic chemists. Xu-Wen Li,a Alexandre Eara and Bastien Nay Nat. Prod. Rep., 2013, Advance Article DOI: 10.1039/C3NP70016J

|
Scooped by
NatProdChem
May 1, 2013 5:06 PM
|
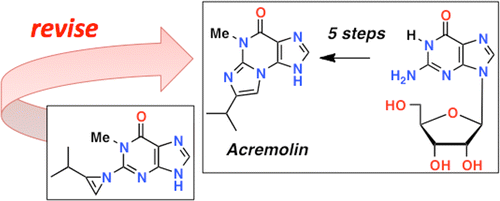
The first synthesis of the heterocyclic marine natural product, acremolin, is reported along with the revision of the structure from a 1H-azirine to a substituted N2,3-ethenoguanine (5-methyl-7-isopropyl-4,5-dihydroimidazo[2,1-b]purine). Additional properties of acremolin are also described including its 1H–15N-HMBC and fluorescence spectra. Lawrence A. Januar† and Tadeusz F. Molinski Org. Lett., Article ASAP DOI: 10.1021/ol400752s Publication Date (Web): May 1, 2013

|
Scooped by
NatProdChem
May 1, 2013 5:01 PM
|
David Dobbs enjoys a brilliant look at the making of DSM-5, the new 'psychiatrists' Bible'.

|
Scooped by
NatProdChem
April 29, 2013 8:24 PM
|
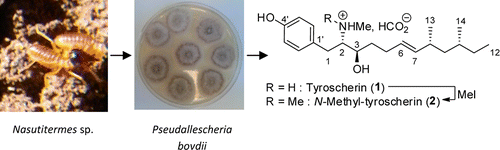
Defense mutualisms between social insects and microorganisms have been described in the literature. The present article describes the discovery of a Pseudallescheria boydii strain isolated from Nasutitermes sp. The microbial symbiont produces two antifungal metabolites: tyroscherin and N-methyltyroscherin, a compound not previously described in the literature. Methylation of tyroscherin has confirmed the structure of N-methyltyroscherin. Both compounds are effective antifungal agents with favorable selectivity indices for Candida albicans and Trichophyton rubrum. Charlotte Nirma†‡, Véronique Eparvier*†, and Didier StienJ. Nat. Prod., Article ASAPDOI: 10.1021/np4001703Publication Date (Web): April 29, 2013

|
Scooped by
NatProdChem
April 27, 2013 2:23 PM
|
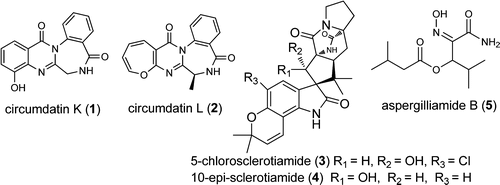
Two new benzodiazepine alkaloids, circumdatins K and L (1, 2), two new prenylated indole alkaloids, 5-chlorosclerotiamide (3) and 10-epi-sclerotiamide (4), and one novel amide, aspergilliamide B (5), together with six known alkaloids were isolated from the deep-sea-derived fungus Aspergillus westerdijkiae DFFSCS013. Their structures were elucidated by extensive spectroscopic analysis. All of the compounds were tested for cytotoxicity toward human carcinoma A549, HL-60, K562, and MCF-7 cell lines. Jiang Peng†‡, Xiao-Yong Zhang†, Zheng-Chao Tu§, Xin-Ya Xu†, and Shu-Hua Qi J. Nat. Prod., Article ASAP DOI: 10.1021/np400132m Publication Date (Web): April 26, 2013
|

|
Scooped by
NatProdChem
May 15, 2013 6:03 PM
|
Embrace mistakes, urges Mario Livio — they are portals to scientific progress.

|
Scooped by
NatProdChem
May 14, 2013 2:13 PM
|
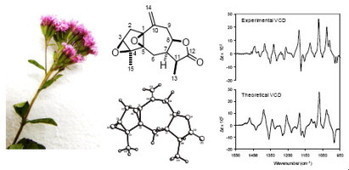
The aerial parts of Stevia tomentosa afforded a new guaianolide which was characterized by physical and spectroscopic data as (−)-1,5:3,4-diepoxyguaia-10(14)-en-12,8-olide (1). The absolute configuration of this unusual compound containing two β-oriented epoxide groups in the five-membered carbocyclic ring, one located at the C-1 and C-5 ring junction, was established as (1S,3S,4S,5R,7R,8R,11S)-1 by vibrational circular dichroism spectroscopy in combination with density functional theory calculations, and by evaluation of Flack and Hooft X-ray parameters. Alejandro Valdez-Calderóna,J. Martín Torres-Valenciaa, , ,J. Jesús Manríquez-Torresa,René Velázquez-Jiméneza,Luisa U. Román-Marínb,Juan D. Hernández-Hernándezb,Carlos M. Cerda-García-Rojasc,Pedro Joseph-Nathan Tetrahedron Letters Volume 54, Issue 25, 19 June 2013, Pages 3286–3289 http://dx.doi.org/10.1016/j.tetlet.2013.04.056

|
Scooped by
NatProdChem
May 13, 2013 10:33 AM
|
Chrysanolide A, a novel guaianolide-type sesquiterpenoid trimer(3), along with its biogenetically related monomer (Chrysanolide B, 1) and dimer (Chrysanolide C, 2), were simultaneously isolated from Chrysanthemum indicum L flowers. Their structures and absolute configurations were elucidated via spectroscopic and computational methods. All isolated compounds were evaluated for their anti-HBV activities. Qiong Gu, Yaoyao Chen, Tianzeng Hui, Dane Huang, Jingwei Zhou, Taizong Wu, Yiping Chen, Lina Shi and Jun Xu RSC Adv., 2013, Accepted Manuscript
DOI: 10.1039/C3RA23172K

|
Scooped by
NatProdChem
May 12, 2013 11:43 AM
|
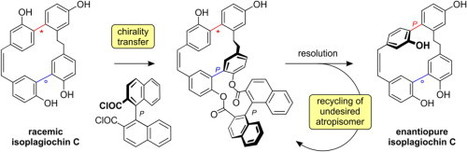
Isoplagiochin C 1 was prepared for the first time in enantiopure form from synthetic racemic material, using a novel stereochemical concept. This macrocyclic bisbibenzyl contains two biaryl axes. Of these, axis A is configurationally stable by its fixation within the macrocyclic framework, while axis B is stereochemically unstable. By diesterification of rac-1 with enantiopure (P)-1,1′-binaphthyl-2,2′-dicarboxylic acid, axis B is locked in its P-configuration, thus allowing the resolution and separate saponification of dilactones (PA,PB,P)-3 and (MA,PB,P)-3 to give the pure enantiomers of 1. This concept permits recycling of any undesired enantiomer of 1 by thermal equilibration of the respective diastereomer of 3. Gerhard Bringmanna, , ,Dominik Hagera,Anu Schaumlöffela,Matthias Grohb,Andreas Speicher Tetrahedron: Asymmetry Volume 24, Issues 9–10, 31 May 2013, Pages 575–581 http://dx.doi.org/10.1016/j.tetasy.2013.03.012

|
Scooped by
NatProdChem
May 10, 2013 6:21 AM
|
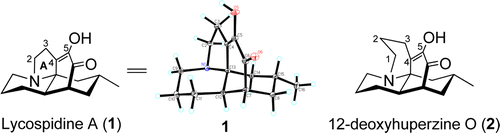
Lycospidine A (1), the first example of a Lycopodium alkaloid which contains an unprecedented five-membered A ring, was isolated from Lycopodium complanatum. The unique five-membered A ring in 1 indicates that carbons 2–5 in 1 are presumably derived from proline instead of the lysine biosynthetically, which suggests that 1 represent a new class of Lycopodium alkaloid. In addition, the unique structural feature and biosynthetic origin of 1 shed new insight into the structural diversity of Lycopodium alkaloid analogue libraries potentially accessible by engineered biosynthesis. Jin-Tang Cheng†‡, Fei Liu†‡, Xiao-Nian Li†, Xing-De Wu†, Liao-Bin Dong†‡, Li-Yan Peng†, Sheng-Xiong Huang†, Juan He*†, and Qin-Shi Zhao Org. Lett., Article ASAP DOI: 10.1021/ol400907v

|
Scooped by
NatProdChem
May 10, 2013 6:14 AM
|
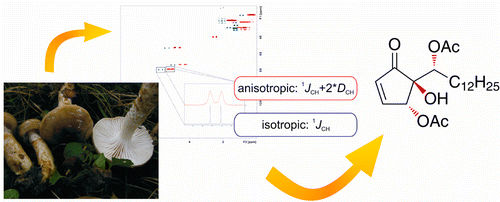
The hygrophorones, a class of cyclopentenones isolated from fruiting bodies of the genus Hygrophorus (basidiomycetes), show promising antifungal activity. While the constitution of 4,6-diacetylhygrophorone A12 (3) and the relative configuration of the stereogenic centers in the cyclopentenone ring were elucidated using standard NMR and MS techniques, the relative configuration of the exocyclic stereogenic center could not be assigned. By introducing a sample of 3 into an alignment medium and measuring anisotropic NMR parameters, namely, residual dipolar couplings, we were able to unambiguously determine the relative configuration of all three stereogenic centers in 4,6-diacetylhygrophorone A12 simultaneously by fitting several structure proposals to the experimental data. Volker Schmidts†, Maic Fredersdorf†, Tilo Lübken‡, Andrea Porzel‡, Norbert Arnold‡, Ludger Wessjohann*‡, and Christina M. Thiele J. Nat. Prod., Article ASAP DOI: 10.1021/np300728b Publication Date (Web): May 9, 2013

|
Scooped by
NatProdChem
May 3, 2013 1:30 PM
|
Structurally diverse alkaloids containing five-membered heterocyclic subunits, such as imidazole, oxazole, thiazole, as well as their saturated congeners, are widely distributed in terrestrial and marine organisms and microorganisms. These naturally occurring secondary metabolites often exhibit extensive and pharmacologically important biological activities. The latest progress involving isolation, biological activities, chemical synthetic studies, and biosynthetic pathways of these natural products has been summarized in this review. Zhong JinNat. Prod. Rep., 2013, Advance Article DOI: 10.1039/C3NP70006 B

|
Scooped by
NatProdChem
May 3, 2013 6:43 AM
|
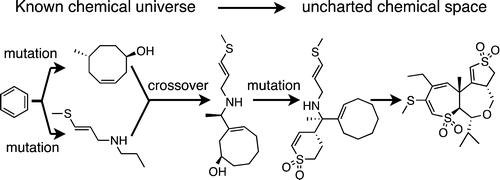
The “small molecule universe” (SMU), the set of all synthetically feasible organic molecules of 500 Da molecular weight or less, is estimated to contain over 1060 structures, making exhaustive searches for structures of interest impractical. Here, we describe the construction of a “representative universal library” spanning the SMU that samples the full extent of feasible small molecule chemistries. This library was generated using the newly developed Algorithm for Chemical Space Exploration with Stochastic Search (ACSESS). ACSESS makes two important contributions to chemical space exploration: it allows the systematic search of the unexplored regions of the small molecule universe, and it facilitates the mining of chemical libraries that do not yet exist, providing a near-infinite source of diverse novel compounds. Aaron M. Virshup†§, Julia Contreras-García†§, Peter Wipf‡§, Weitao Yang*†§, and David N. Beratan J. Am. Chem. Soc., Article ASAP DOI: 10.1021/ja401184g Publication Date (Web): April 2, 2013

|
Scooped by
NatProdChem
May 2, 2013 2:41 PM
|
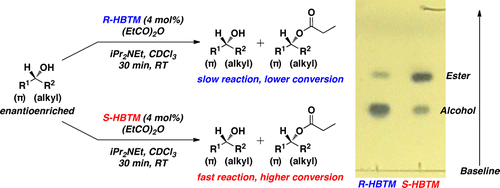
A new implementation of the competing enantioselective conversion (CEC) method was developed to qualitatively determine the absolute configuration of enantioenriched secondary alcohols using thin-layer chromatography. The entire process for the method requires approximately 60 min and utilizes micromole quantities of the secondary alcohol being tested. A number of synthetically relevant secondary alcohols are presented. Additionally, 1H NMR spectroscopy was conducted on all samples to provide evidence of reaction conversion that supports the qualitative method presented herein. Alexander J. Wagner and Scott D. RychnovskyJ. Org. Chem., Article ASAPDOI: 10.1021/jo400432qPublication Date (Web): April 17, 2013

|
Scooped by
NatProdChem
May 1, 2013 5:04 PM
|
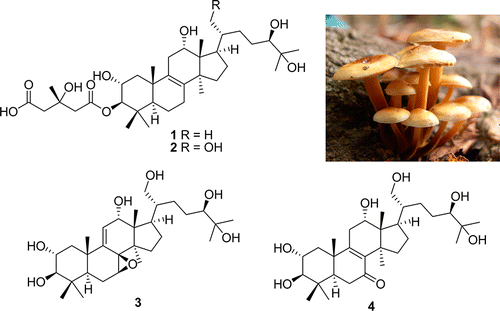
In our continuing search for structurally interesting and bioactive metabolites from Korean wild mushrooms, bioassay-guided fractionation and a chemical investigation of the MeOH extracts of the fruiting bodies of the mushroom Naematoloma fasciculare resulted in the isolation of four new lanostane triterpenoids (1–4), together with 11 known compounds (5–15). The structures of 1–5 were determined by a combination of 1D and 2D NMR and HRMS. The absolute configuration of the 3-hydroxy-3-methylglutaryl group as a side chain in 1 and 2 was determined by the alkaline methanolysis method. The full NMR data assignment of the known compound fasciculol G (5) is reported for the first time. Compounds 1–15 were tested for their antiproliferative activities against four human cancer cell lines (A549, SK-OV-3, SK-MEL-2, and HCT-15) and evaluated for their inhibitory effects on nitric oxide production in a lipopolysaccharide-activated murine microglial cell line. Ki Hyun Kim†, Eunjung Moon‡, Sang Un Choi§, Sun Yeou Kim, and Kang Ro Lee J. Nat. Prod., Article ASAP DOI: 10.1021/np300801x Publication Date (Web): May 1, 2013

|
Scooped by
NatProdChem
April 29, 2013 8:29 PM
|
Be More Unfocused Discover what innovations can come from having a broader scientific perspective.

|
Scooped by
NatProdChem
April 28, 2013 10:15 AM
|
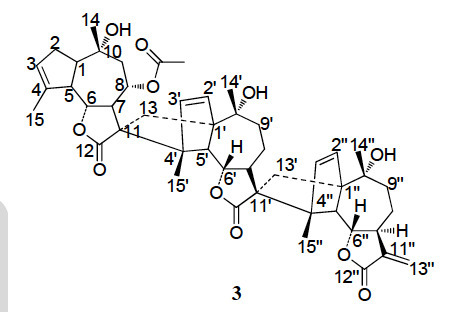
Thirteen highly oxygenated daphnane diterpenoids, including six known compounds, were isolated from the stems of Trigonostemon lii. The structures were elucidated by extensive spectroscopic analyses including 2D NMR spectroscopy (HSQC, 1H–1H COSY, HMBC, and ROESY) and mass spectrometry. The absolute stereochemistries of compounds were established on the basis of CD spectra. Four of the compounds showed modest anti-HIV-1 activity (EC50 = 2.04, 9.17, 11.42, and 9.05 μg/ml, TI = 26.49, >21.81, 9.32, and 9.56, respectively) in vitro. Shi-Fei Lia, b,Yu Zhanga,Ning Huangb, c,Yong-Tang Zhengc,Ying-Tong Dia,Shun-Lin Lia,Yuan-Yuan Chenga, b,Hong-Ping Hea, , ,Xiao-Jiang Hao Phytochemistry Available online 27 April 2013 In Press, Corrected Proof http://dx.doi.org/10.1016/j.phytochem.2013.03.003
|



 Your new post is loading...
Your new post is loading...