Your new post is loading...

|
Scooped by
Juan Lama
June 24, 2025 10:48 AM
|
RetroVirox has launched a Summer Promotion with a 30% discount for antiviral and neutralization services against 4 viruses, including influenza, dengue, human metapneumovirus (HMPV) and respiratory syncytial virus (RSV). Discount applies to services initiated between July 1 and August 31 2025
Contact us at info@retrovirox.com for inquiries and additional info

|
Scooped by
Juan Lama
April 29, 12:35 PM
|
Highlights • Severe IAV infection imparts a long-lasting decline in cardiac function • Atypical GMP-independent CD319+ pro-DC3s transport infectious IAV to cardiomyocytes • Direct IFN-I signaling on cardiomyocytes drives cardiac dysfunction • mod-mRNA delivery of dominant-negative IFNAR1 on cardiomyocytes ameliorates damage Summary Abundant evidence has correlated influenza infection with cardiovascular disease, yet mechanisms linking infection with the heart remain poorly understood. Here, we show that influenza infection damaged the human and murine heart. In mice, we showed that shortly after pulmonary infection, the virus infected a circulating myeloid pro-dendritic cell 3 (pro-DC3) that expressed high concentrations of the chemokine receptor CCR2. The heart, which produces abundant CCL2, preferentially attracted infected pro-DC3. In the myocardium, the virus escaped pro-DC3, infected cardiomyocytes, and triggered production of type-I interferon (IFN-I). Engagement of the IFN-I receptor (IFNAR1) on cardiomyocytes caused tissue damage and compromised heart function. Genetically and therapeutically dampening IFNAR1 exclusively in cardiomyocytes protected the heart while preserving anti-viral immunity in the lung. Our results identify a series of host-pathogen interactions that propagate tissue damage and uncover an axis for intervention to mitigate cardiovascular risk following viral infection. Published in Immunity (March 10, 2026):

|
Scooped by
Juan Lama
April 22, 11:48 AM
|
This study investigated stroke incidence after severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection using Qatar’s nationwide stroke registry linked with national testing, vaccination, and mortality databases. A self-controlled case series design was implemented among individuals with stroke and confirmed primary SARS-CoV-2 infection between January 1, 2020, and April 11, 2023. Incidence rate ratios (IRRs) comparing stroke incidence rate during the 1–90 days post-infection with predefined control periods were estimated using conditional Poisson regression. Of 4,187 stroke cases, 338 with confirmed infection met inclusion criteria. Stroke incidence rate was elevated during the 1–90 days post-infection (IRR 1.84, 95% CI: 1.31–2.58), peaking at days 1–28 (IRR 2.22, 95% CI: 1.53–3.22) and declining thereafter (days 29–59: IRR 1.47, 95% CI: 0.97–2.24; days 60–90: IRR 0.72, 95% CI: 0.43–1.21). This corresponded to 5.9 excess strokes per 100,000 infections. Elevated risk was confined to pre-Omicron infections (IRR 2.98, 95% CI: 1.89–4.70), with no evidence of increased risk observed for Omicron infections (IRR 0.82, 95% CI: 0.46–1.45). Subtype analyses showed increased risk for ischemic stroke (IRR 1.99, 95% CI: 1.37–2.89) and cerebral venous sinus thrombosis (CVST; IRR 3.52, 95% CI: 1.15–10.78), but not hemorrhagic stroke (IRR 0.64, 95% CI: 0.16–2.51). SARS-CoV-2 infection was associated with a transient elevation in stroke risk, peaking shortly after infection and declining thereafter. Excess risk was confined to pre-Omicron infections and driven primarily by ischemic stroke, while CVST, though rare, showed the strongest relative effect. Published in Scientific Reports (April 2026)

|
Scooped by
Juan Lama
April 10, 1:42 PM
|
In 2024, a bovine H5N1 strain was first isolated from dairy cows in Texas and confirmed to transmit cross-species to humans. Therefore, research on treatments for human infection should be accelerated. In our study, the antiviral effects of baloxavir acid (BXA), oseltamivir carboxylate (OSC), EIDD-1931 (NHC), and ribavirin (RBV) against five H5N1 strains were evaluated in vitro. Cell viability and viral replication were measured to assess the antiviral effects. The results showed that the EC50 of BXA treatment was the lowest. The BXA/NHC and BXA/OSC combination treatments showed more potent inhibitory effects than each monotherapy. The 15 mg/kg baloxavir marboxil (BXM) / 125 mg/kg molnupiravir (MNP) and the 15 mg/kg BXM / 10 mg/kg oseltamivir phosphate (OSP) were tested in BALB/c mice. The mice were inoculated with 10 times the 50% mouse lethal dose (10 MLD50) of bovine H5N1 virus. Treatments began 1-day post-infection (1 dpi) and were administered orally twice daily for 5 or 7 days. Changes in body weight, clinical signs, and survival were monitored; lung and brain tissues were collected for virological, immunological, and histological analyses. Most mice died from severe neurological symptoms. Compared with the 5-day treatment, the 7-day treatment effectively inhibited viral replication and increased survival rates to 50% in BXM, BXM/MNP, and BXM/OSP treatments. Mice treated with BXM/MNP or BXM/OSP combination therapy showed lower viral yields in the lungs than those treated with BXM alone. The results provide a reference for human treatment, and extending the 7-day combination treatment should be considered. Published in Emerging Microbes and Infections (March 31, 2026): https://doi.org/10.1080/22221751.2026.2645843

|
Scooped by
Juan Lama
April 8, 12:03 PM
|
More than three-quarters of all cases of liver cancer worldwide are associated with chronic viral hepatitis but scientists have been limited in their ability to model how these viruses lead to cancer. In the new study, a Rockefeller team showed that mice infected with an engineered version of a rat virus develop liver inflammation, scarring, and ultimately cancer similar to that seen in humans with viral hepatitis-associated liver cancer. The new mouse model can be used to study how liver virus cause cancer.... Study published in J. Hepatology (March 2026): https://www.journal-of-hepatology.eu/article/S0168-8278(26)00085-1/fulltext

|
Scooped by
Juan Lama
March 29, 1:33 PM
|
Many viruses have adapted to persist in infected humans for life1,2. Variable host control of their ongoing abundance (viral load) can lead to clearance or disease3–5. Here we analysed the viral DNA load of 31 common viruses in human blood and saliva using whole-genome sequencing data from UK Biobank (n = 490,401), All of Us (n = 414,817) and Simons Foundation Powering Autism Research for Knowledge (SPARK; n = 12,519). Viral DNA load varied markedly with age, time of day and season; most viruses were also present at greater abundance in men than in women. Human genetic variation at dozens of loci associated with DNA load of seven viruses: Epstein–Barr virus (EBV, 45 loci), human herpesvirus (HHV)-7 (37 loci), HHV-6B, Merkel cell polyomavirus and three anelloviruses. Variation at the major histocompatibility complex (MHC) locus generated the strongest associations (P = 5.8 × 10–9 to 2.5 × 10–1459), which were specific to each virus. The HLA-B*08:01 allele also exhibited a host–virus genetic interaction with EBV subtype (P = 7.4 × 10–70). Other human genetic effects implicated genes encoding proteins that process peptides for antigen presentation, such as ERAP1 (HHV-7, P = 2.7 × 10–78) and ERAP2 (EBV, P = 4.6 × 10–111). Mendelian randomization analyses supported a strong causal effect of EBV DNA load on increased risk of Hodgkin’s lymphoma (P = 1.8 × 10–3), but not multiple sclerosis (P = 0.52). This suggests that higher chronic EBV load increases lymphoma risk, whereas associations of EBV infection with autoimmune conditions reflect host immune responses to particular viral epitopes. Analyses of biobank data show that human variation such as age, sex and genetics, particularly at the major histocompatibility complex locus, is associated with viral abundance and supports a causal link between abundance of Epstein–Barr virus and Hodgkin’s lymphoma. Published in Nature (March 25, 2026): https://doi.org/10.1038/s41586-026-10288-y

|
Scooped by
Juan Lama
March 15, 10:53 AM
|
As the U.S. flu season winds down, health officials say the flu vaccine didn’t work very well with one of its worst effectiveness rates in more than a decade. A new strain that dominated the early winter was not well matched to the vaccine, leading to an intense early onslaught of flu. The Centers for Disease Control and Prevention on Friday posted data that showed a continued decline in doctor's office and hospital visits for flu symptoms through last week. The number of states reporting high flu activity dropped to 16, many of them in a belt stretching from Colorado to Virginia. “The winter respiratory virus season is slowly coming to a close, and we’re all very grateful for that,” said Dr. William Schaffner, a Vanderbilt University vaccine expert. This season's vaccines were around 25% to 30% effective in preventing adults from getting sick enough from the flu that they had to go to a doctor’s office, clinic or hospital, according to a CDC report this week. Children who were vaccinated were about 40% less likely to get treatment at a doctor’s office or hospital. Officials generally are pleased if a flu vaccine is 40% to 60% effective. Judging from past CDC research, this season saw one of the lowest effectiveness rates in the last two decades. Flu infections surged in late December and were especially intense in some parts of the country. New York City health officials called it the most intense season in 20 years. Relatively low flu vaccination rates did not help, but experts also blamed the new flu strain that was causing most infections. The new strain belonged to a category of flu virus, called A H3N2. This new version, subclade K, seemed to spread more easily — though it did not necessarily cause more severe illness. The vaccine available for this season was built to address a different version of H3N2, and the new strain's explosion is a likely explanation for why the vaccine was less effective, Schaffner said. CDC scientists estimate there have been at least 27 million illnesses, 350,000 hospitalizations and 22,000 deaths from flu so far this season. At the same point last year, the estimates were at least 40 million illnesses, 520,000 hospitalizations, but about the same number of deaths. At least 101 children have died so far this season. For those whose vaccination status is known, about 85% were not fully vaccinated against flu. The flu vaccine may not protect everyone from getting sick, but it can prevent people from becoming severely ill and dying. That's why getting a flu shot remains worthwhile, Schaffner said. CDC data suggests adult vaccination rates are up slightly this season, to 46.5%, following an unusually bad season last year that set a record for the most child deaths this century. An estimated 48% of U.S. kids were vaccinated against flu around the end of last month. That's about the same as last year, but down from the 52% vaccinated at this point in 2024, according to CDC data. Starting in 2010, the government recommended annual flu vaccinations for Americans 6 months and older. In January, however, the Trump administration stopped broadly recommending flu shots for all children, saying instead that it’s up to parents and family doctors to decide. Meanwhile, work is already underway for next winter’s flu season. Last month, the World Health Organization announced its recommendations for which virus strains to address in the vaccines for the 2026-27 northern hemisphere flu season. The vaccines should be built to handle subclade K, the organization said. This week, a U.S. Food and Drug Administration advisory committee endorsed the WHO recommendations. CDC Report in MMWR (March 12, 2026): https://www.cdc.gov/mmwr/volumes/75/wr/mm7509a2.htm

|
Scooped by
Juan Lama
March 12, 12:06 PM
|
Emerging research from the University of Virginia (UVA) has unveiled a startling connection between severe viral respiratory infections and an increased risk for lung cancer development months or even years post-infection. This groundbreaking study, spearheaded by Dr. Jie Sun and colleagues at UVA’s Beirne B. Carter for Immunology Research and Comprehensive Cancer Center, elucidates how severe cases of COVID-19, influenza, and pneumonia induce long-lasting alterations in lung immune cell behavior, effectively “reprogramming” the pulmonary immune landscape to favor tumor growth. Their findings illuminate critical implications for clinical surveillance paradigms, vaccination policies, and future cancer prevention strategies. The investigation began with an acknowledgement of the longstanding gap in understanding the long-term oncogenic consequences of viral lung injuries. Researchers utilized both murine models and retrospective human clinical data to assess how severe respiratory viral infections modulate lung immunity and impact carcinogenesis. Results from murine studies demonstrated that mice afflicted with severe pulmonary infections exhibited a marked propensity to develop lung tumors and suffered higher mortality rates following cancer establishment when compared to uninfected controls. This phenomenon suggested a causative link between intense lung inflammation and an immunological milieu conducive to cancer progression. In parallel analysis, the team accessed large-scale patient datasets revealing a statistically significant association between hospitalization for severe COVID-19 and an elevated incidence of lung cancer, quantified at a 1.24-fold increased risk independent of traditional risk factors like smoking and pre-existing comorbidities. This correlation underscored the real-world clinical relevance of the murine findings and raised urgent questions about post-infection cancer monitoring protocols. Notably, mild COVID-19 cases did not present this increased risk, implying that the severity of immune perturbation is the critical determinant in carcinogenic priming. Mechanistically, the study pinpointed drastic shifts in lung-resident innate immune cells—specifically neutrophils and macrophages—that are ordinarily responsible for pathogen clearance and tissue homeostasis. Following severe viral infection, these cells adopt dysfunctional phenotypes characterized by sustained pro-inflammatory cytokine production and extracellular matrix remodeling. This aberrant activation fosters a chronic inflammatory microenvironment rich in reactive oxygen species and growth factors, creating fertile ground for malignant transformation and tumor growth. Additionally, perturbations to the epithelial lining of alveoli were observed, further compromising the lung’s integrity and enhancing vulnerability to oncogenic insults. A significant and hopeful facet of Sun’s research is the protective effect consistent vaccination confers against these detrimental alterations. Vaccinated individuals, by priming adaptive immunity and curbing viral replication, mitigate the chaotic immune activation and lung injury associated with severe infection. The data suggest that vaccines serve a dual protective role: preventing acute disease hospitalization and abrogating the long-term immune scarring responsible for increased cancer susceptibility. This finding advocates for intensified vaccination efforts not only for infectious disease control but also for cancer prevention in vulnerable populations. The clinical ramifications of these discoveries are profound. Dr. Jeffrey Sturek, a UVA physician-scientist who collaborated on the study, emphasized the need to rethink post-viral infection surveillance akin to long-established cancer risk assessments derived from smoking history. Patients recovering from severe viral pneumonia could benefit from more rigorous and routine lung cancer screening protocols, perhaps integrating low-dose computed tomography scans for early tumor detection. The insights call for personalized medicine approaches that integrate viral infection history into oncological risk stratification models. Beyond clinical implications, the study expands our fundamental understanding of how acute infectious insults can prime chronic disease pathways. It reveals a novel paradigm where immune memory of viral trauma contributes to malignancy, broadening the landscape of cancer etiology beyond genetic and environmental factors. Such knowledge opens avenues for innovative therapeutic interventions aimed at restoring immune cell function and reversing pro-tumor inflammatory imprints in post-infection lungs. UVA’s Beirne B. Carter Center continues to lead in dissecting the complex interplay between infection, immunity, and cancer, building on prior work elucidating immune cell plasticity in disease. The Center’s integrative approach, combining immunology, oncology, and infectious disease expertise, uniquely positions it to translate these findings into targeted therapies. Their efforts are complemented by the UVA Comprehensive Cancer Center’s national leadership in cancer research, recognized through the prestigious National Cancer Institute designation. Publication of this multidisciplinary research in the high-impact journal Cell not only solidifies its scientific rigor but also ensures wide dissemination among the biomedical community. The work was supported by multiple National Institutes of Health grants and UVA institutional awards, highlighting the collaborative investment in addressing pressing health challenges arising from global pandemics and endemic respiratory diseases. Dr. Sun and colleagues stress the urgency for the medical community to incorporate viral infection histories into routine cancer risk evaluation and to champion vaccination as a multifaceted health safeguard. With millions affected worldwide by severe COVID-19 and influenza annually, proactive measures based on this research could markedly reduce lung cancer morbidity and mortality. The authors anticipate that ongoing research will further clarify molecular pathways involved, fostering development of precision medicine interventions to disrupt the cancer-promoting sequelae of viral lung infections. Published in Cell (March 11, 2026): https://www.sciencedirect.com/science/article/abs/pii/S0092867426002205

|
Scooped by
Juan Lama
February 19, 12:09 PM
|
A virus that is big enough to be seen under an ordinary light microscope co-opts its host’s systems with the help of potentially purloined genes. Scientists report that a type of giant virus multiplies furiously by hijacking its host’s protein-making machinery — long-sought experimental evidence that viruses can co-opt a system typically associated with cellular life. The researchers found that the virus makes a complex of three proteins that takes over its host’s protein-production system, which then churns out viral proteins instead of the host’s own. Virologists had already suspected that viruses could perform such a feat, says Frederik Schulz, a computational biologist at the Lawrence Berkeley National Laboratory in California, who was not involved with the work. But the new findings, published in Cell on 17 February, are an important confirmation. Compared with other viruses, he says, this one “has a more powerful toolbox to really replace what the host is doing”. Big microbe Giant viruses, which are so named for their massive genomes, might seem exotic, but they are decidedly commonplace. They tend to infect single-celled organisms called protists — a group that includes amoebae and protozoa — that “are all over the place”, says Eugene Koonin, an evolutionary biologist at the US National Center for Biotechnology Information in Bethesda, Maryland. The giant DNA virus used in this study, Acanthamoeba polyphaga mimivirus, has a genome that is about five times larger than those of poxviruses, which have the biggest genomes of any virus that infects humans. Mimivirus is giant in another way too: it is large enough to be seen under a light microscope. To understand whether the virus affects its host’s protein-assembly line, the researchers isolated viral proteins that interact with host organelles called ribosomes. These structures translate RNA molecules into proteins. Viral imitation The scientists identified three viral proteins that seemed likely to be involved in hijacking host protein production. They then genetically engineered the viruses to lack these proteins and found that viruses that were missing any one of the three multiplied 1,000–100,000 times more slowly than those that did have these proteins. “It turns out that, yes, they were absolutely required for infection,” says Maximilian Fels, a virologist at Harvard Medical School in Boston, Massachusetts, and a co-author of the study. The mimivirus genome includes genes for three viral products that shut down protein synthesis in the host and redirect it towards making viral proteins. #giantvirusThe authors found that the three viral proteins form a complex that is similarly shaped to one produced by the host that attaches to the ribosome and affects which proteins are made. When mimivirus infects a host, its protein cluster replaces the host’s, and viral RNA molecules gain preferential access to the ribosome, depriving the host RNA molecules of access to the cellular machinery. The researchers also exposed the host cells to stressful conditions by adding harmful chemicals or depriving the cells of nutrients. Even in these stressed cells, which would typically respond by halting most protein production, the virus could still multiply. But it did not multiply as quickly if it was missing any one of the three crucial proteins. Gene thieves Individual viruses are so simple and have such bare-bone structures that they do not qualify as cells. So how did Acanthamoeba polyphaga mimivirus come to have proteins that can replace part of its host cell’s function? Scientists say that it’s possible that mimivirus ‘stole’ useful genes from its hosts early in its evolutionary history, and then evolved to use them. “These large viruses that have large DNA genomes are gene thieves,” says Nels Elde, an evolutionary geneticist at the University of Utah in Salt Lake City. “They collect host genes almost like trading cards.” The authors of the new paper hypothesize that mimivirus and other giant viruses are so large because they’ve taken a lot of genetic machinery on board. This might allow the viruses to thrive in a wider range of environments than can viruses with smaller genomes.

|
Scooped by
Juan Lama
February 17, 12:11 PM
|
Mucosal surfaces are primary entry sites for many infectious pathogens, yet parenteral vaccination alone often fails to elicit effective mucosal immunity. Mucosally delivered vaccines offer a promising strategy for reinforcing frontline defences and inducing localized, pathogen-specific immune responses. Recent studies indicate that mucosal vaccines elicit tissue-resident memory T and B cells, along with robust local antibody secretion, to prevent infection and transmission. However, achieving sterilizing immunity at mucosal sites proves challenging owing to the complex immune environments consisting of epithelial barriers, varying mucus composition, pH differences and hormonal influences. In this Review, we outline how specialized immune-inductive and effector mechanisms across distinct mucosal compartments contribute to protective immunity and discuss emerging strategies to harness multilayered mucosal immunity to develop safe, effective vaccines that elicit durable protection. Many clinically relevant pathogens enter the body through mucosal surfaces, yet conventional parenteral immunization is insufficient to elicit robust mucosal immunity. This Review examines the unique anatomical and immunological features of the mucosal surfaces of the body and how this knowledge can be used to develop protective mucosal vaccines. Published in Nat. Reviews Immunology (February 16, 2026): https://doi.org/10.1038/s41577-026-01273-7

|
Scooped by
Juan Lama
February 15, 9:08 AM
|
Staff members at the United States’s premier infectious-disease research institute have been instructed to remove the words “biodefense” and “pandemic preparedness” from the institute’s web pages, according to e-mails Nature has obtained. The directive comes amid a broader shake-up at the US National Institute of Allergy and Infectious Diseases (NIAID), one of 27 institutes and centres at the National Institutes of Health (NIH). The NIAID is expected to deprioritize the two topics in an overhaul of its funded research projects, according to four NIAID employees who spoke to Nature on the condition of anonymity, because they are not authorized to speak to the press. NIH director Jay Bhattacharya explained the restructure at an event with other top agency officials on 30 January. “It’s a complete transformation of [the NIAID] away from this old model” that has historically prioritized research on HIV, biodefence and pandemic preparedness, he said. The institute will focus more on basic immunology and other infectious diseases currently affecting people in the United States, he added, rather than on predicting future diseases. About one-third of the NIAID’s US$6.6-billion budget currently funds projects involving emerging infectious diseases and biodefence. The research studies pathogens of concern and monitors their spread, and develops medical countermeasures against threats from radiation exposure, chemicals and infectious diseases. Nahid Bhadelia, director of Boston University’s Center on Emerging Infectious Diseases in Massachusetts, says the decision to deprioritize these areas will leave people in the United States more vulnerable to pathogens that are constantly evolving in wildlife around the world and spilling into human populations, sometimes sparking outbreaks. “Just because we say we’re going to stop caring about these issues doesn’t make the issues go away — it just makes us less prepared,” she says. A spokesperson for the NIH, the world’s largest public funder of biomedical science, based in Bethesda, Maryland, says, “NIAID’s new vision sharpens its focus on the interconnected pillars of infectious diseases and immunology, expanding opportunities for research that address the most pressing challenges to Americans’ health today.” The spokesperson declined to respond to Nature's queries about the agency’s specific plans to restructure the institute. Political heat
The NIAID is currently under the leadership of acting director Jeffery Taubenberger, after its previous director, infectious-disease physician Jeanne Marrazzo, was fired by the administration of US President Donald Trump after less than two years into the post. Her predecessor, Anthony Fauci, held the job for 38 years. Fauci and the institute have been scrutinized by Trump and other Republican politicians as a result of public-health measures used during the COVID-19 pandemic — such as lockdowns and school closures — which they say resulted in people losing trust in the country’s health agencies. (During the pandemic, Fauci offered recommendations on how to prevent the spread of the virus, but neither Fauci nor the NIAID set policy for public-health measures.)
To restore trust, Bhattacharya, Taubenberger and Taubenberger’s senior adviser, John Powers, outlined a “new vision” for the institute in a commentary1 published in Nature Medicine on 16 January. “NIAID’s work clearly neither prevented the pandemic nor prevented Americans from experiencing among the highest levels of all-cause excess mortality in the developed world during that time,” they wrote. “Given the increasing prevalence of allergic and autoimmune disorders and the burden of common infections in the population over the past few decades, the NIAID must focus research on these conditions with a greater sense of urgency.” New direction The instructions to agency staff members to rebrand the institute’s language are only the first step towards implementing this new vision, according to the NIAID employees. NIH principal deputy director Matthew Memoli has ordered more changes, including the review of the portfolio of grants funding biodefence and pandemic preparedness, in the coming weeks and months, they say. If funds are allocated to other topics, “that’s a very big deal”, Bhadelia says. Few other US agencies have the budget or infrastructure to fund basic research into these topics, says Gigi Gronvall, a biosecurity specialist at the Johns Hopkins Bloomberg School of Public Health in Baltimore, Maryland. Taubenberger hinted during the 30 January event that a reallocation is coming. “To better prepare for the future [we need to] better deal with what we’re facing today,” he said. “Maybe a better way to look at this is ‘people preparedness’. One way for people to be prepared is to be healthier, eat better and exercise, so you’re less likely to get sick or have a poor outcome,” Powers added. This priority is important, Bhadelia says, but “the new vision is remarkable not for what’s included — but what’s excluded. It almost paints a picture of ‘one or the other’. In reality, these things interact with each other”. For example, people with chronic conditions are at greater risk during a pandemic, she says. Gronvall adds that this approach is “full of hubris”. “We know that there are groups of viruses that are more likely to cause illness, epidemics and pandemics,” she says, so it makes sense to study them. For example, Bhadelia adds, the anticipatory basic research that the NIAID has funded helped make it possible to develop COVID-19 vaccines in record time, which “allowed us to reduce mortality” from the pandemic. The restructure is also expected to target the NIAID division focusing on HIV/AIDS research, which oversees a $1.5-billion portfolio of projects developing therapeutics and vaccines against the virus. The division’s 33 branches will likely be consolidated, one NIAID employee tells Nature. But it’s not clear whether the total number of projects or amount of money the division doles out will be impacted, the employee adds. Almost 20% of the NIH’s 2024 workforce of 21,000 have been laid off or have left voluntarily since Trump took office last January. The NIH spokesperson declined to say whether there will be further layoffs of NIAID staff members as part of the restructure. “Everyone is worried about what comes next,” an institute employee says.

|
Scooped by
Juan Lama
February 10, 11:51 AM
|
Incidence patterns of post-acute sequelae, characterised by persistence or delayed onset after the acute phase of an infection, are not well documented after infectious disease outbreaks. Nipah virus was first detected in the 1998-1999 Malaysia outbreak and remains a significant public health concern due to its high epidemic potential and recurrent outbreaks in South Asia. We conducted a systematic review on the prevalence, incidence, duration, and characteristics of post-acute sequelae in survivors of Nipah virus infection. We searched PubMed and Web of Science for studies published up to November 17, 2025. We included 8 articles, and extracted prevalence for 34 potential neurological, psychiatric or non-specific post-acute sequelae. The pooled prevalence of total residual neurological deficits was 24% (95% CI 9-49) among total survivors of Nipah infection, and 45% (95% CI 11-85) among the subset of survivors with acute Nipah encephalitis (5 articles). In the single controlled study, total residual neurological deficits, fatigue and daytime somnolence were significantly more prevalent in Nipah infection survivors than population-based controls. We estimated that 10% (95% CI 4-20) of Nipah infection survivors also experience late-onset or relapsing neurological symptoms after initial recovery. These findings demonstrate a substantial long-term disease burden following Nipah virus infection, which should be accounted for in mathematical modelling studies. However, the estimates are mainly based on data from the Malaysia/Singapore outbreak and may not be generalisable to the Bangladeshi and Indian setting, where current outbreaks occur and are caused by a different viral strain. Further limitations relate to subjective outcome assessment and heterogeneous populations of total Nipah infection survivors, which could have biased our estimates. Preprint inmedRxiv ( Feb. 09, 2026): https://doi.org/10.64898/2026.02.03.26345343

|
Scooped by
Juan Lama
February 4, 12:35 PM
|
A panzootic caused by highly pathogenic avian influenza (HPAI) A(H5N1) virus, clade 2.3.4.4b, has affected many animal species around the world since 2021. In March 2024, genotype B3.13 of this virus was identified in dairy cattle in the United States, following a spillover event from wild birds. Mammary gland lesions were a key finding in infected cows, with infectious virus detected in their milk. Raw milk is sold legally in retail establishments in multiple US states, including California. In November 2024, HPAI A(H5N1) virus, clade 2.3.4.4b, genotype B3.13, was detected in raw milk sold commercially in California and then in bulk milk tanks. The affected product later was recalled. We describe an 8-mo-old cat with a history of severe illness after consuming this raw milk before it was recalled. The cat was euthanized and submitted for postmortem examination and diagnostic workup. Autopsy and histopathology revealed icterus, nasal discharge, hydrothorax, gliosis, and necrotizing pneumonia, hepatitis, and salpingitis, among other lesions. Immunohistochemistry for influenza A virus revealed intralesional immunolabeling in many organs. Molecular detection was positive for HPAI A(H5N1) virus, clade 2.3.4.4b, genotype B3.13. To our knowledge, HPAI A(H5N1) virus has not been reported previously in cats after consuming raw milk purchased from a retail establishment, nor has salpingitis been associated with HPAI A(H5N1) virus infection in a mammal. Hepatic damage and icterus were prominent findings in our case rather than primary involvement of the CNS.
|

|
Scooped by
Juan Lama
April 30, 12:05 PM
|
J. Craig Venter, a scientist and entrepreneur who raced to decode the human genome, died on Wednesday in San Diego. He was 79. His death was announced by the J. Craig Venter Institute, a nonprofit research organization founded by Dr. Venter and based in San Diego and Rockville, Md. The institute said in a statement that Dr. Venter had been hospitalized recently for side effects from cancer treatment. In the 1990s, Dr. Venter, a risk-taker and intense competitor, made a bold move when he decided that the Human Genome Project, a $3 billion government program for decoding the human genome, was moving slowly enough that he could enter the race late and beat it with a much faster method. His gamble paid off. In 2000, his company, Celera, made a joint announcement with a rival group saying that they had assembled the first human genomes, a landmark step toward uncovering the genetic basis of human disease and origins. Dr. Venter had a powerful ego. That was clear when he let slip that the anonymous donor whose genome Celera had sequenced was none other than his own. But his drive and management skills helped him inspire loyalty and assemble teams of exceptional scientists, including the Nobel Prize-winning microbiologist Hamilton O. Smith. Together, they achieved one landmark after another in the nascent field of genomics. In 1995, Dr. Venter revolutionized microbiology when he published the sequence of DNA letters in the bacterium Haemophilus influenzae, the first bacterial genome to be decoded, along with annotations of all the organism’s genes. The moment electrified science. For the first time, researchers could see all the genetic components of a free-living organism, giving microbiologists a manual for bacterium’s genetic tool kit. It also set off a race to sequence the genomes of known pathogens, with the aim of identifying their genetic arsenals and devising countermeasures. His team next turned to the fruit fly genome to test whether their approach, known as a whole-genome shotgun sequencing, could work on its biggest target: the human genome. The fruit fly genome was successfully decoded in 2000, providing a wealth of information that would help scientists study both the fly and human genomes. Decoding the human genome, Dr. Venter’s next big challenge, became the focus of a competition between his team at Celera against a consortium of academic rivals led by the United States and Britain. The National Institutes of Health, a major patron of the effort, refused to cooperate with Dr. Venter, who instead secured private funding from the Celera Corporation. Dr. Venter’s whole genome shotgun method enabled him to erase his rivals’ head start. With ample time and money, he might have seized this greatest of scientific prizes. Instead, he agreed, reluctantly, to accept the consortium’s offer of a formal draw, with the trappings of a White House ceremony with President Bill Clinton. For his contributions to sequencing the human genome, Dr. Venter received the Nierenberg Prize for Science in the Public Interest from the Scripps Institution of Oceanography in 2007. President Barack Obama presented him with the National Medal of Science in 2009.

|
Scooped by
Juan Lama
April 24, 12:04 AM
|
The Food and Drug Administration on Thursday approved Regeneron’s Otarmeni, the first gene therapy for genetic hearing loss. The drugmaker said it will offer the drug for free to U.S. patients. Otarmeni is approved to treat a very rare form of hearing loss that affects about 50 babies born in the U.S. each year and is caused by a mutation in a gene called OTOF. The approval was granted under the Commissioner’s National Priority Voucher, an FDA pilot program intended to fast-track drug reviews. “This really is life-changing for families with children with hearing loss,” Dr. Eliot Shearer, a pediatric otolaryngologist at Boston Children’s Hospital and a principal investigator on the Regeneron trial. The only other treatment option for children with genetic deafness is cochlear implants, Shearer said, which restore the ability to hear speech and music, but reduce the fidelity of the sound.With gene therapy, the improved hearing is “on 24/7 and doesn’t rely on batteries,” he added. The drug works by replacing the faulty OTOF gene, which is supposed to work by giving the body instructions to make a protein called otoferlin. This protein plays a role in transmitting pulses from cochlear cells within the ear to nearby nerves, which carry the signals on to the brain. Without otoferlin, sound never reaches the brain. Thursday’s approval was based on a trial of 20 children diagnosed with the OTOF mutation who got a single dose of the therapy in one or both ears. The trial found that it improved hearing in 16 of the 20 children, with five kids developing the ability to detect whispers. Side effects included infection or inflammation of the middle ear, vomiting, nausea and dizziness. The approval was touted by President Donald Trump during an Oval Office announcement Thursday afternoon about a drug pricing deal with Regeneron to provide many of its drugs at a discount on the self-pay platform TrumpRx. While the company is providing the drug at no cost, it has no say over the cost of administering the drug. Shearer said the procedure is similar to surgery for cochlear implants, where doctors access the inner ear to deliver the drug while the patient is under general anesthesia. The drug is not approved outside the U.S., so Regeneron does not have a list price for people abroad who want to get the treatment. Gene therapy prices can be in the millions. Sarah Emond, president and CEO of the Institute for Clinical and Economic Review, a nonprofit group that evaluates the cost and effectiveness of drugs, said Regeneron should be commended on its pricing. “Regeneron has shown us that one option we can consider that will ensure affordable access for patients to these therapies is to not charge the health system for the therapy,” Emond said.

|
Scooped by
Juan Lama
April 17, 1:44 PM
|
A study conducted at the University of São Paulo (USP) in Brazil reveals that tissues such as the tonsils and adenoids can serve as hiding places for the rhinovirus, which causes the common cold and is responsible for most respiratory infections worldwide. Using samples from 293 children who underwent surgery to remove these tissues, the study showed that the pathogen can infect immune cells known as lymphocytes and remain there for long periods without causing symptoms. This allows the virus to potentially be transmitted to others without warning. The findings are published in the Journal of Medical Virology. "The virus has a 'date' with the child population. Every year, about two or three weeks after school starts in temperate regions, there's a rhinovirus outbreak. And children pass it on to their parents and grandparents. We've always wondered: What does the start of school have to do with it? Well, children gather in closed spaces, and some of them with the virus in their throats can spark an outbreak at school, even if they're asymptomatic," comments rhinovirologist Eurico de Arruda Neto, a professor at the Ribeirão Preto School of Medicine (FMRP-USP) and coordinator of the research. As the researcher explains, it was already known that the rhinovirus infects the epithelium (the outermost layer of the mucosa) of the nose and throat, hijacks the cellular machinery to multiply, and causes the host cell to rupture once this process is complete, releasing progeny capable of generating new infections. For this reason, scientists consider it a lytic virus, one that causes cell lysis (rupture). This rapid and destructive cycle quickly draws the attention of the immune system, which, in most cases, eliminates the virus from the body within about five to seven days. The major finding of the study was that the rhinovirus can reach the deeper layers of tonsil and adenoid tissues in addition to the epithelium. There, it can infect B lymphocytes, which produce antibodies, and CD4 T lymphocytes, which conduct the local immune response. These cells have a long lifespan and store the "memory" of the immune system. Rather than killing them, the rhinovirus remains inside these cells for extended periods, in a state of persistence similar to that seen with herpes viruses, HPV, and cytomegalovirus. "The samples we analyzed are from children who underwent surgery due to snoring, sleep apnea, or recurrent infections related to tonsillar and adenoid hypertrophy. At the time of surgery, they were necessarily asymptomatic. Nevertheless, we detected the rhinovirus in a large number of participants," says Arruda. In addition to the tonsils and adenoids, the children's nasal secretions were analyzed as well. According to the study, the virus was present in at least one of the three sites (tonsil, adenoid, or secretion) in 46% of the volunteers. Viral proteins and other signs that the rhinovirus was replicating—and therefore capable of infecting another person—were also observed in these tissues. The research was conducted in collaboration with Ronaldo Martins, a virologist from the Ribeirão Preto School of Pharmaceutical Sciences (FCFRP-USP), as well as professors Wilma Anselmo-Lima, Edwin Tamashiro, and Fabiana Valera from the FMRP-USP. 'Virus garden' In previous studies, Arruda's team had detected adenovirus (another cause of the common cold), influenza A (flu), and SARS-CoV-2 (COVID-19) in samples of tonsils and adenoids from children who had undergone surgery. The latter two are known to cause longer-lasting infections in some patients. In the case of the rhinovirus, however, this came as a surprise. "I get the impression that no matter what common virus we look for, we'll find it. And not just in the tonsils and adenoids, but in other lymphoid tissues throughout the body, such as lymph nodes. We already have some preliminary evidence that lymphoid tissues are a sort of 'garden' for viruses. And our hypothesis is that this is a good thing. It acts as a booster for immune memory, meaning antibodies continue to be produced even long after initial exposure," says Arruda. However, in the case of people with asthma, this can be problematic. One hypothesis raised by the authors of the article is that infectious viruses in tonsil CD4 T lymphocytes may release inflammatory substances that act on the lungs and cause asthma attacks. It is already known that colds and the flu are among the most common causes of asthma attacks, especially in young children. Additionally, a previous study by the group detected respiratory viruses in normal adenoids (without hypertrophy), which are located next to the Eustachian tube. This may explain why some children suffer from recurrent otitis media. "This virus can pass from the adenoids to the middle ear and cause inflammation there. The child won't sneeze or cough, but the ear will become inflamed, closing the narrow Eustachian tube and leading to a buildup of fluid in which the local bacterial flora begins to proliferate," the researcher explains. Clinical implications Based on these findings, Arruda believes that pediatricians should be mindful of the possibility of diagnostic confusion regarding the causes of childhood illnesses. "For example, a child with hypertrophic tonsils arrives at the emergency room with a respiratory infection and bronchiolitis symptoms caused by respiratory syncytial virus, but the throat swab test detects rhinovirus from a previous infection. In other words, tests performed on secretions may not always reflect what's actually happening in the lungs," says the researcher. "We have evidence that this viral persistence can also occur in people with normal-sized tonsils and adenoids." Another hypothesis to be investigated, Arruda says, is whether viruses that persist in lymphoid tissues can cause problems for immunosuppressed patients. "Patients who undergo bone marrow transplants, for example, frequently develop lung infections and bronchiolitis. Doctors, nurses, and medical students are usually blamed for bringing the virus into the high-risk ward. But could it be that the virus was already present in the patient's tonsils or adenoids and has now been reactivated due to low immunity? It doesn't have to be transmission from outside to inside. That's what we've started to investigate in mice," he explains. Study published Jan. 2026: https://onlinelibrary.wiley.com/doi/10.1002/jmv.70809

|
Scooped by
Juan Lama
April 10, 1:19 PM
|
The acting director of the Centers for Disease Control and Prevention has delayed publication of a CDC report showing the covid-19 vaccine cut the likelihood of emergency department visits and hospitalizations for healthy adults last winter by about half, according to two scientists familiar with the decision. The scientists spoke on the condition of anonymity for fear of retaliation. The move has raised concerns among current and former officials that information about the vaccine’s benefits are being downplayed because they conflict with the views of Health Secretary Robert F. Kennedy Jr., who has been an outspoken critic of the shots. The delay, which has not been previously reported, offers a window into how vaccine policy is being shaped behind the scenes, even as the Trump administration has sought to soften its public posture on controversial actions ahead of the midterm elections. The report had been scheduled for publication March 19 in the CDC’s flagship scientific journal, the Morbidity and Mortality Weekly Report, the scientists said. Between September and December last year, healthy adults who received the vaccine reduced their likelihood of emergency department and urgent care visits by 50 percent and cut the likelihood of covid-associated hospitalizations by 55 percent, compared with those not receiving a 2025-2026 vaccine dose, according to a summary of the report obtained by The Washington Post. The report had cleared the agency’s scientific-review process, the scientists said, but now it has been delayed by acting CDC director Jay Bhattacharya over concerns about the methodology, the scientists said. The same methodology has long been used by the CDC to evaluate vaccine effectiveness for respiratory viruses, including influenza. A report about flu vaccine effectiveness this past winter — using the same methodology — was published in the MMWR a week earlier. That methodology was also used in a 2021 study on covid vaccine effectiveness in clinics and hospitals published in the New England Journal of Medicine. Vaccine effectiveness estimates using the same methodology have also been published in other peer-reviewed journals, including JAMA Network Open, the Lancet and Pediatrics....

|
Scooped by
Juan Lama
April 3, 12:31 PM
|
AI predicted that a forgotten breast cancer drug could be repurposed to treat many respiratory and gastrointestinal viruses, and subsequent animal tests suggests it may be right. A single drug has been found to inhibit a range of common viruses in lab studies, including coronaviruses, respiratory syncytial virus (RSV), norovirus, and influenza and hepatitis viruses. It will be tested in a clinical trial next year, raising hopes that the pill could one day be taken at home to relieve unpleasant symptoms or even limit infections if there were another viral pandemic. “As far as we can tell, this is the first drug that’s ever demonstrated activity across all these viral families,” says Daniel Haders, co-founder of Model Medicines, the California-based company leading its development. If it is approved, Haders envisages it being a pill that people could take if, for example, they have a flu-like illness but don’t know if it is influenza, covid-19, RSV or something else...

|
Scooped by
Juan Lama
March 26, 12:39 PM
|
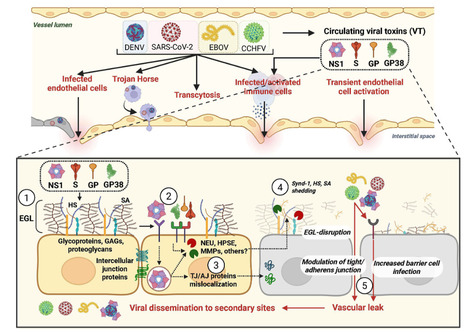
The process by which viruses cause disease, viral pathogenesis, is the result of both infection of cells and the host immune response. A less studied but equally important contributor to viral pathogenesis is viral dissemination, the capacity of a virus to move from the primary site of infection, traverse physiological barriers, and gain access to secondary sites of infection. This dictates viral tropism and pathogenesis, but the mechanisms governing barrier crossing are incompletely understood. While the presence of viral receptors on cells is a major determinant of viral tropism and a prerequisite for infection, it does not completely explain the capacity of viruses to enter a tissue. Our recent work has begun to characterize the contribution of soluble viral proteins, acting as “viral toxins,” to viral dissemination, tissue tropism, and overall pathogenesis within an infected host. In this review, we discuss the characteristics of these viral toxins, which are soluble or surface-exposed viral proteins that can interact with endothelial and/or epithelial barriers, as well as immune cells, to trigger signaling pathways, resulting in the transient breakdown of cellular structures maintaining barrier integrity. The disruption of these barriers induces vascular leak and facilitates virus dissemination, influencing viral tropism and pathogenesis. Importantly, blocking this process prevents leak, viral dissemination, and severe disease during infection, highlighting the value of therapeutic intervention against viral toxin activity. Here, we summarize our current understanding of recently discovered viral toxins from the Flaviviridae, Coronaviridae, Nairoviridae, and Filoviridae. Published March 13, 2026 in MBio:

|
Scooped by
Juan Lama
March 14, 12:26 PM
|
Evidence suggests reactivations of the varicella-zoster virus may accelerate aging and raise dementia risk. Now scientists want to know if vaccines and antivirals could help protect the brain. The lecturer—a 63-year-old viral immunologist whose identity has been kept anonymous—suffered alarming symptoms, including impaired memory, waning concentration, and difficulty reading. While giving lectures to students, he found he had difficulty focusing and was often unable to finish sentences without pausing. But medical tests, including a brain biopsy, failed to get to the source of the problem, and over the next four years, his symptoms continued to progress. His decline would have likely continued unabated had he not heard about a case of encephalitis—serious brain inflammation caused by a reactivation of the varicella-zoster virus, most commonly associated with childhood chickenpox and, later in life, shingles. Remembering that his own symptoms had been preceded by a brief case of shingles, subsequent tests confirmed the patient had indeed experienced a reactivation of varicella-zoster. And so he decided to treat the problem with a course of acyclovir, an antiviral drug commonly prescribed to shingles patients. To his colleagues’ amazement, the Colorado lecturer’s symptoms quickly faded away and his cognition returned to normal. This remarkable case study, published in 2016, has inspired neurovirologists to look deeper into the connection between shingles and brain aging. For decades, shingles has been predominantly associated with a form of nerve pain known as postherpetic neuralgia, which can be so severe that it was once cited as the leading cause of pain-related suicide in the elderly. Now, research is starting to reveal the devastating impact that shingles can have on brain health. According to Andrew Bubak, assistant professor of neurology at the University of Colorado Anschutz, the true burden of varicella-zoster “is totally underestimated. But it’s a very treatable virus.” In recent years, increasing numbers of studies have shown that the shingles vaccine appears to be capable of protecting the aging body and brain, and dementia specialists are taking note. In April 2025, a major study by researchers at Stanford University suggested vaccination against shingles could prevent one in five new cases of dementia. More recent studies have also linked getting a shingles vaccine to slower biological aging across a variety of measures. One explanation given for the findings is that the vaccine might be stimulating the immune system in a broadly beneficial manner. While there is likely some truth in this, additional research increasingly points to the value of avoiding shingles (or reactivations of the varicella-zoster virus) in the first place, with two separate studies finding associations between shingles and self-reported cognitive decline and dementia. Neurovirologists believe this emerging data underlines the importance of avoiding infection, through the childhood chickenpox vaccination—given to children in the US since 1995 and introduced in the UK in January 2026—and through the adult shingles vaccine and booster jabs in later life. Before the US started routinely vaccinating against chickenpox, more than 90 percent of children acquired the varicella-zoster virus in childhood. Following the infection, the virus takes up position in the peripheral nervous system—the neurons linking the brain and spinal cord to the limbs and organs—where it stays dormant, sometimes for decades. Varicella-zoster can reactivate in the body following various triggers, which range from acute stress to concussion, co-infections with Covid-19, immunosuppressive medications, and the general aging of the immune system. In many cases, such reactivations may be completely symptomless, with some studies suggesting many of us could unknowingly experience repeated “subclinical” reactivations—the virus reawakening from its dormant state without inducing visible symptoms—in mid- to later life. “We rely on specialized immune cells to continuously patrol the nervous system and keep the dormant virus suppressed,” says Tian-Shin Yeh, associate professor of medicine at Taipei Medical University and attending physician at Shuang Ho Hospital in Taiwan. “As we get older, these cells can become less effective, or exhausted.” Once varicella-zoster reactivates, several things can take place. The virus is part of the herpes family, members of which are particularly adept at infiltrating the brain and central nervous system due to their ability to exploit its internal transport mechanisms for their own use. “Our nerve cells contain molecular motors that shuttle cargo along the nerve fibers,” says Yeh. “Herpes viruses can commandeer these transport systems to move from peripheral tissues deep into the nervous system.”....

|
Scooped by
Juan Lama
February 23, 11:16 AM
|
An innovative approach supercharges the innate immune system to provide a first line of defence against respiratory infections. Imagine if a nasal spray could make you immune not only to the viruses that cause COVID-19 and influenza, but to all respiratory diseases. In a paper1 published in Science today, researchers describe a vaccine that has done just that. When given to mice, the vaccine protected them for at least three months against multiple disease-causing viruses and bacteria — including the SARS-CoV-2 virus that causes COVID-19 — and even quelling responses to respiratory allergens. If the research translates to humans safely and effectively, such a ‘universal vaccine’ could be offered to everyone at the start of each winter — and perhaps provide a first line of defence against future pandemics. Bali Pulendran, an immunologist at Stanford University in Palo Alto, California, and his group previously studied the Bacillus Calmette–Guérin (BCG) vaccine2, which provides temporary protection against numerous diseases and works by activating the innate immune system and keeping it active. This evolutionarily ancient system has a much broader reactivity than does the adaptive immune system — which is the one conventional vaccines utilize by teaching antibody-making B cells and T cells to recognize proteins found on specific pathogens. Activating the innate immune system can also induce the intrinsic capacity of the respiratory system’s epithelial cells to resist infection. These cells are the target of many pathogens. Double bulwark In the latest study, Pulendran’s team developed a universal vaccine that targets the innate immune system, with three components. The first two are drugs that stimulate specific receptor proteins that can activate innate immune cells, such as macrophages that reside in the lungs. The third component stimulates a population of T cells, which are part of the adaptive immune system. Their task is to keep sending signals to the innate immune system to maintain its active state. The vaccine contains an immunogenic protein from chicken eggs, and in experiments in which it was omitted, immunity quickly waned. Mice that were given four doses of the nasally delivered vaccine developed immunity to SARS-CoV-2 and other coronaviruses, plus to bacteria that cause certain respiratory infections. Another novel benefit was that the activated pathways also suppressed the mechanisms that mediate hypersensitivity to house dust mites, thereby preventing allergic asthma. Analyses of how the protection works revealed what Pulendran calls a two-bulwark system, in which an initial mucosal barrier limits pathogen entry into the lungs. “Then,” he says, “this mucosal vaccination has set up the lung immune system, so that it is extraordinarily rapid in eliciting the virus-specific immune response to send off those few viruses that slip through the initial bulwark.” Bridge vaccine “It’s really a fantastic paper, and it’s exciting. The data look very clear to me,” says Akiko Iwasaki, an immunobiologist at Yale University in New Haven, Connecticut. “If it works in humans, that would be really quite remarkable.” Zhou Xing, an immunologist at McMaster University in Hamilton, Canada, says that people who are familiar with the advances in mucosal vaccines over the last decade will not be surprised by the findings. “We call it a ‘bridge vaccine’,” he says — the idea of leveraging the innate immune system to generate non-selective pathogen protection. Both Xing and Iwasaki warn that translating the effectiveness seen in mice into clinical therapies for people isn’t straightforward. Xing fears that without advanced aerosol delivery methods, a nasally delivered vaccine in humans won’t reach the lungs to activate the immune pathways described here. (The small size of mice means that both nasal sprays and pathogens can easily access their lungs.) Xing also says that roll out of the vaccine on a large scale could prove impractical if people need four doses. Because this approach puts the immune system in a state of constant hypervigilance, it could also come with potential side effects. “The cost–benefit ratio is something that one has to assess very, very cautiously in humans,” Pulendran says. If no problems are encountered in an initial, dose-escalation safety trial, a second trial would then use controlled infections, he predicts. That is, sometime after vaccination, healthy volunteers would be deliberately exposed to the influenza virus, for example, to see if they are protected.If they are, a new form of preventive medicine might be in the offing. “Mice provide you with the conceptual framework and evidence of how it could work,” Pulendran says. “But really, the proof of the pudding is: does it really work in humans?” Study published in Science (feb. 19, 2026): https://www.science.org/doi/10.1126/science.aea1260

|
Scooped by
Juan Lama
February 18, 2:40 PM
|
The US Food and Drug Administration has reversed course and will review a new mRNA flu vaccine from Moderna, the pharmaceutical company said Wednesday. About two weeks ago, the FDA sent Moderna a letter in which it refused to accept the application to review its first mRNA seasonal flu vaccine — a rare move by the federal agency that raised concern about another setback for the technology that’s been a target of some Trump administration health officials. The FDA told Moderna that its application didn’t contain an “adequate and well-controlled” trial because the control arm didn’t reflect the “best-available standard of care in the United States at the time of the study,” according to the letter, that Moderna posted online. It didn’t identify any safety or efficacy concerns, the company said. But Moderna has since met with the FDA and “proposed a revised regulatory approach” with different pathways by age, according to a news release from the company. Moderna is now “seeking full approval for adults 50 to 64 years of age and accelerated approval for adults 65 and older, along with a post-marketing requirement to conduct an additional study in older adults,” the news release said. “Discussions with the company led to a revised regulatory approach and an amended application, which FDA accepted,” Andrew Nixon, a spokesperson for the US Department of Health and Human Services, said in a statement. “FDA will maintain its high standards during review and potential licensure stages as it does with all products.” If the FDA approves the vaccine, it could be available for people ages 50 and older for the upcoming 2026-2027 flu season. Last week, Moderna said that the initial refusal from the FDA was inconsistent with previous feedback from the agency. “We appreciate the FDA’s engagement in a constructive Type A meeting and its agreement to advance our application for review,” Moderna CEO Stéphane Bancel said in a statement. “Pending FDA approval, we look forward to making our flu vaccine available later this year so that America’s seniors have access to a new option to protect themselves against flu.”

|
Scooped by
Juan Lama
February 17, 11:53 AM
|
In Massachusetts, Moderna is pulling back on vaccine studies. In Texas, a small company canceled plans to build a factory that would have created new jobs manufacturing a technology used in vaccines. In San Diego, another manufacturing company laid off workers. When Robert F. Kennedy Jr. was picked in November 2024 to become the next health secretary, public health experts worried that the longtime vaccine skeptic would wreak havoc on the fragile business of vaccine development. Those fears are beginning to come true, according to executives and investors involved with companies that develop and sell vaccines and the technology that is best known for the Covid vaccines. At conferences and in interviews, they described the emerging consequences of the Trump administration’s dismantling of the longstanding federal support for vaccines. “There will be less invention, investment and innovation in vaccines generally, across all the companies,” Dr. Stephen Hoge, the president of Moderna, said in an interview. The Trump administration said it was not discouraging innovation. But investors have grown hesitant to bet on a field that has fallen out of favor in Washington. Major manufacturers are reporting declining sales of their shots. Smaller companies are taking the brunt of the impact, with some stocks whipsawing in response to the changes...

|
Scooped by
Juan Lama
February 12, 11:55 AM
|
Researchers at the Helmholtz Institute for One Health (HIOH), a site of the Helmholtz Centre for Infection Research (HZI), together with an interdisciplinary team of partners, have identified the fire-footed rope squirrel (Funisciurus pyrropus) as a likely natural reservoir of the monkeypox virus (MPXV). Their study was published today in the renowned scientific journal Nature. The discovery was based on the detailed investigation of an mpox outbreak among wild sooty mangabeys (Cercocebus atys) in Taï National Park, Côte d’Ivoire. Scientists combined ecological, behavioral and molecular evidence to document, for the first time, the interspecies transmission of MPXV in the wild, from fire-footed rope squirrels to sooty mangabeys. A deadly outbreak among mangabeys For decades, the researchers now at HIOH have worked closely with the Taï Chimpanzee Project to monitor the health of wild chimpanzees, sooty mangabeys and other wildlife in Taï National Park, Côte d'Ivoire — a long-term commitment that proved essential to detecting this transmission event. In early 2023, the team identified an outbreak of mpox in a well-studied group of sooty mangabeys: About one third of the group showed clinical signs of disease, and four infants died. Viral genome sequencing revealed that the virus detected in the infected monkeys was nearly identical to an MPXV strain identified in a fire-footed rope squirrel found dead 12 weeks earlier nearby. In an attempt to link both observations, the team analyzed fecal samples from the mangabeys, seeking evidence of pre-outbreak MPXV circulation and contact between the host species. One sample collected eight weeks before the outbreak onset contained DNA from both the virus and the rope squirrel, providing strong evidence of interspecies transmission at this moment. Behavioral data supported these findings. Sooty mangabeys from this group have already been observed catching and eating fire-footed rope squirrels, which provides a direct route for the transmission of viruses. Squirrels under suspicion: now confirmed Squirrels have long been suspected as potential reservoirs for MPXV. The first isolation of the virus from a wild animal was from a rope squirrel (Funisciurus anaerythrus) captured in the Democratic Republic of the Congo in 1985. In 2003, imported squirrels infected with MPXV were also among the rodents suspected to have been the source of an mpox outbreak in pet prairie dog owners in the US. Yet, these animals had never been shown to be directly responsible for an outbreak in another species in nature. The new study is a breakthrough that starts unveiling how the pathogen circulates in the wild. What this means for human health As hunting pressure has reduced populations of larger game species, rodents such as squirrels are increasingly hunted and consumed by humans, which likely heightens the risk of human exposure and zoonotic transmission of MPXV. Therefore, confirming the direct involvement of fire-footed rope squirrels in interspecies transmission carries important public health implications. “Identifying the animal sources of the virus and the exposure routes that lead to inter-species transmission are key steps towards understanding spillover mechanisms and developing effective prevention measures to mitigate the risk of transmission to humans,” says Livia V. Patrono, one of the senior authors at HIOH. The authors recommend increasing awareness among people who come into contact with squirrels and other wildlife, such as children. In addition, they call for a deeper understanding of MPXV ecology in reservoir species – especially squirrels – as well as in intermediate hosts, particularly non-human primates, in MPXV-endemic regions, to strengthen evidence-based prevention strategies. One Health approach more relevant than ever The findings underscore the importance of a One Health approach that recognizes the links between human, animal, and environmental health. “This discovery was only possible thanks to long-term ecological research, continuous health monitoring and systematic sample collection in the Taï National Park,” says Fabian Leendertz, senior author, director of HIOH and co-director of the Taï Chimpanzee Project. “We need to maintain and expand this kind of effort to better understand and hopefully reduce the risks posed by emerging infectious diseases, including mpox – we need to strengthen prevention.” Josef Penninger, Scientific Director of the Helmholtz Centre for Infection Research, adds: “This study also highlights the value of close cooperation with our African partners. Only through strong, trust-based collaborations with local authorities and research institutions can we effectively tackle zoonotic diseases and make an impact, not just regionally, but globally.” Study published in Nature (feb. 11, 2026): https://www.nature.com/articles/s41586-025-10086-y

|
Scooped by
Juan Lama
February 6, 11:55 AM
|
The establishment of human intestinal enteroids (HIEs) as a model for human norovirus (HuNoV) replication has been transformative for studying this leading cause of gastroenteritis. However, indefinite passaging of HuNoVs in HIEs remained a challenge, necessitating the use of patient stool samples as viral inocula. Using RNA-seq, we identified CXCL10, CXCL11, and CCL5 as up-regulated chemokines, suggesting their potential as host restriction factors. TAK-779, a CXCR3/CCR5/CCR2 antagonist, enhanced GII.3 HuNoV replication and viral spread in a dose- and time-dependent manner, enabling successful passaging of GII.3 HuNoV in two different HIE lines and generation of viral stocks. Sequencing passaged virus revealed one consensus change in the major capsid protein and several dynamic adaptations, suggesting emergence of variants. TAK-779 also enhanced replication of GI.1 and GII.17 strains, but not GII.4, suggesting strain-specific host interactions. This breakthrough in passaging provides insight into HuNoV-host interactions, establishes a scalable in vitro system for virus propagation, and opens avenues for structural, biochemical, and therapeutic studies. Published in Science Advances (Feb. 4, 2026): https://www.science.org/doi/10.1126/sciadv.aeb0455

|
Scooped by
Juan Lama
February 2, 11:41 AM
|
Key to the success of influenza A virus as a pathogen are its numerous tactics of immune evasion. To suppress anti-viral cellular and organismal responses, influenza A virus encodes several immunomodulatory proteins, including the endoribonuclease PA-X. PA-X decreases inflammation and immune responses in in vivo infections by limiting host gene expression. PA-X is conserved in 99% of all influenza A viral strains, pointing to its importance as a crucial immunomodulator. However, it is not yet known how PA-X activity alters the antiviral response in the human airway or how it benefits the virus. To define how influenza A virus uses this protein to evade immune responses, we characterized the impacts of PA-X on the host response to infection in the infected and bystander cells of the airway epithelium using a 3D ex vivo model. We discovered that PA-X exerts a dual action on immune responses, dampening aspects of both the innate and adaptive immune systems. Consistent with reports in model organisms, PA-X significantly decreases secretion of multiple cytokines from airway epithelium, including IFN-λ, which likely plays a role in its ability to reduce inflammation and lung damage. In addition, we revealed that PA-X decreases and delays MHC I antigen presentation from infected cells. This reduction likely aids influenza A virus in hiding from antigen-specific T cells and allows the virus to successfully replicate prior to immune detection. This new function for PA-X highlights how influenza A virus employs active mechanisms to block immune detection, in addition to tolerating high levels of antigen mutations to escape it. Moreover, as evasion of recognition by the adaptive immune system is particularly important during infections of animals and humans with pre-existing immunity, this immunomodulatory activity may be key to the longevity of influenza A viruses and their continued circulation. Preprint in bioRxiv (Jan. 31, 2026): https://doi.org/10.64898/2026.01.30.702929
|


 Your new post is loading...
Your new post is loading...