Your new post is loading...

|
Scooped by
mhryu@live.com
Today, 11:23 PM
|
Molecular docking is a pillar of structure-based drug design and shows advantages in structure prediction of small-molecule ligand-protein complexes over co-folding methods for novel ligands and novel binding pockets. Here, we describe substantial improvements of our physics-based docking algorithm Attracting Cavities, which is widely used through the SwissDock webserver. AC 3.0 includes enhanced sampling features, new functionalities, and technical improvements. These lead to better sampling at lower execution times and higher versatility. Comparison with AutoDock Vina demonstrates better docking results on multiple test sets. Availability: AC 3.0 will be made available free of charge through the SwissDock webserver (www.swissdock.ch).

|
Scooped by
mhryu@live.com
Today, 11:07 PM
|
Photobiocatalysis with photoautotrophic whole cells has demonstrated strong potential for producing chiral molecules and platform chemicals using sustainable inputs such as light, water and CO2 under mild reaction conditions. Coupling enzymatic transformations directly to natural photosynthesis enables higher atom efficiency compared with heterotrophic systems. However, large-scale application remains challenging, particularly due to light attenuation in photobioreactors. In this review, we summarize recent advances in whole-cell photobiotransformations with emphasis on process conditions. We also discuss strategies for intensifying photobiocatalysis through improved reactor design and new immobilization materials, along with developments in fast-growing photoautotrophic strains. Sustainability analyses indicate that organic electron donors represent only one factor influencing environmental performance, and simply replacing them with photosynthetic water splitting does not inherently yield a carbon-negative process. Nonetheless, our calculations show that when high substrate loadings are combined with wastewater use and optimized downstream processing, photosynthesis-driven biotechnology can offer substantial reductions in CO2 emissions.

|
Scooped by
mhryu@live.com
Today, 8:08 PM
|
RNA secondary structure plays a critical role in gene regulation, yet existing computational and experimental tools for structure analysis are often fragmented across prediction, ensemble modeling, and functional interpretation workflows. Here, we present ShapeRNA, a user-friendly web server for integrated RNA secondary structure prediction, ensemble inference, and structure-aware regulatory annotation. ShapeRNA supports three complementary analytical workflows, including sequence-based structure prediction, reactivity-guided modeling using SHAPE or DMS data, and sequencing–guided ensemble inference from high-throughput probing experiments. The platform integrates multiple established prediction algorithms and provides standardized data processing, ensemble clustering, and visualization. In addition, ShapeRNA enables mapping of RNA modification sites, microRNA target regions, and RNA-binding protein interaction motifs onto predicted RNA structures and representative ensemble conformations. We demonstrate the utility of ShapeRNA through applications including analysis of mutation-associated structural changes in MAPT exon 10, characterization of conformational heterogeneity in the HIV-1 Rev Response Element, and regulatory annotation of the oncogenic long non-coding RNA HULC. ShapeRNA provides an accessible and extensible platform for investigating RNA structural heterogeneity and regulatory mechanisms. This website is free and open to all users, and there is no login requirement. https://shaperna.com.

|
Scooped by
mhryu@live.com
Today, 7:20 PM
|
Microorganism and plant interactions are crucial for development and environmental adaptation. Plant growth promoting bacteria enhance agricultural productivity in a sustainable manner, while epigenetic modifications such as DNA methylation regulate gene expression and adaptive responses. The objective of this study is to determine how DNA hypomethylation influences early interactions between maize (Zea mays) and the endophytic diazotrophic bacterium Herbaspirillum seropedicae, particularly regarding plant growth, metabolism, and the root microbiome. Treatment with the hypomethylating agent 5-azacytidine (5-azaC) altered maize root morphology without affecting bacterial growth. Inoculation with H. seropedicae promoted plant growth and bacterial colonization in root mucilage, with higher accumulation in 5-azaC treated roots. Global methylation analysis showed that bacterial inoculation modulates cytosine methylation in a manner similar to 5-azaC, suggesting a role in epigenetic regulation. Gene expression analysis of DNA methylation machinery confirmed that hypomethylation drives plant-microbe interactions. Root microbiome profiling revealed that 5-azaC disrupted microbial composition, which was partially restored by bacterial inoculation. Proteomic analysis identified 1,818 proteins and highlighted significant changes in metabolic pathways, especially carbon metabolism and the citric acid cycle. These findings demonstrate that DNA hypomethylation combined with bacterial interaction profoundly affects cellular and metabolic processes and provide insights for sustainable agricultural practices through epigenetic and microbial modulation.

|
Scooped by
mhryu@live.com
Today, 6:20 PM
|
The ATP molecule is the universal energy currency across all living organisms. There are two fundamental pathways of ATP synthesis: substrate-level and oxidative phosphorylation. While substrate-level phosphorylation generates ATP directly, in oxidative phosphorylation, proton motive force (PMF) is required to power ATP synthesis via the F1Fo ATP synthase. Using E. coli, we show that due to simultaneous use of both pathways, the strength of coupling between ATP and PMF strongly depends on growth conditions: coupling is weak when requirements for independent generation of ATP and PMF are met, and becomes essential when not. We determine the conditions, under which PMF-ATP coupling becomes essential and show that PMF is required for bacterial growth irrespective of its ATP synthesis function. We propose that the main role of F1Fo in E. coli, contrary to the canonical view, is not to generate ATP but to provide an auxiliary pathway that allows both, ATP and PMF, to be produced.

|
Scooped by
mhryu@live.com
Today, 12:57 AM
|
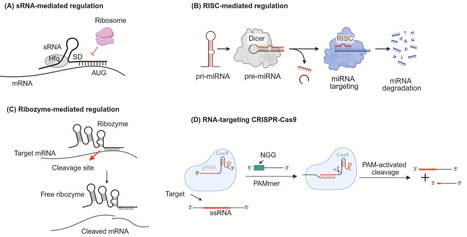
The world's increasing demand for petrochemical, pharmaceutical, and nutraceutical products necessitates the development of new strategies for producing these high-value chemicals. The depletion of the natural fossil reserves and environmental pollution associated with their procurement further compel us to find sustainable, greener, and cost-effective alternatives. In light of this, a shift has been witnessed in deriving these products from fossil-based sources or chemical synthesis to biomanufacturing (production using living systems). However, to fully utilize the potential of biomanufacturing, novel tools and strategies that can function in bacteria, archaea, and eukaryotes, regulate multi-enzyme pathways, offer precise and conditional gene regulation, and possess versatility are highly required. This review presents a comprehensive summary of the latest gene modulation tools and strategies used by metabolic engineers, along with a mechanistic overview and their applications. In addition, we presented the tools that have the potential to be used for pathway optimization but are still less explored. Within this context, we categorized these tools based on their molecular level of gene regulation, i.e., at and beyond the central dogma. We believe that a deeper understanding of the design, development, and application of these tools would be beneficial for metabolic engineers to reprogram biosynthetic pathways by adopting system-specific approaches, as a single strategy cannot be applied to all systems. Lastly, we discussed the challenges and future prospects of developing these gene regulatory tools to further advance the biomanufacturing field.

|
Scooped by
mhryu@live.com
Today, 12:38 AM
|
Precise, site-specific insertion of large gene sequences holds great promise for the treatment of diverse genetic disorders. Although prime editing using paired guide RNAs (pegRNAs) can mediate targeted integration, insertion efficiency drops sharply for payloads exceeding 300 base pairs. Here we present a rationally designed quadruple pegRNA strategy (QuadPE) for efficient and programmable insertion of large DNA fragments. Through screening different designs, we identified that combinations of two genome-targeting pegRNAs in a PAM-out or PAM-in orientation, when paired with two donor-targeting pegRNAs in linear or circular form, yield optimal efficiency. Using QuadPE, we achieved stable integration efficiency of DNA fragments ranging from 1.6 to 26 kb, with efficiencies of around 40% at multiple loci with minimal off-target insertion activity. QuadPE substantially outperformed recombinase-mediated (PASSIGE and PASTE) and transposase-mediated (CAST) insertion systems, particularly for larger payloads, showing a 11-fold, 61-fold and 12-fold improvement for a 9.5 kb insertion, respectively. Notably, QuadPE was effective in both dividing and non-dividing primary cells such as human primary T cells and post-mitotic neurons, establishing QuadPE as a powerful and precise platform for large-fragment gene insertion without the need for double-stranded breaks or recombinases. QuadPE—a quadruple paired prime editing guide RNA strategy—enables efficient and programmable insertion of large DNA fragments.

|
Scooped by
mhryu@live.com
April 22, 11:42 PM
|
Pseudomonas aeruginosa, an opportunistic Gram-negative pathogen, uses a cell-cell signaling system called quorum sensing to coordinate group behaviors. Quorum sensing in P. aeruginosa is a model to study cooperative behaviors in populations. Several studies of cooperation have been conducted using strain PAO1. Wild-type PAO1 harbors a mutation in mexS, which encodes a negative regulator of the transcription factor MexT, which, in turn, activates many genes, including the efflux pump MexEF-OprN. We hypothesized that the PAO1 mexS mutation might affect cooperative behaviors. When P. aeruginosa is passaged daily on casein as a sole carbon source, quorum sensing is required to induce synthesis of the extracellular proteases needed to acquire carbon and energy. When PAO1 is grown on casein, individuals with inactivating mutations in the quorum-sensing regulator LasR are reproducibly enriched in the population. These LasR mutants are cheaters that benefit from cooperatively produced proteases and have a fitness advantage over cooperators. We passaged wild-type PAO1, PAO1 with a gene-corrected version of mexS, or PAO1 with a null mutant of mexT on casein as the sole carbon and energy source. We found that correcting the mexS mutation resulted in unstable cooperation: bacterial cultures failed to propagate after about 15 days, whereas the wild type propagated for the duration of our 30-day experiment. The MexS-corrected and MexT-deficient populations also reproducibly exhibited emergence of a particular quorum-sensing variant, LasR-V226I. This variant activated a subset of quorum-sensing regulated genes, suggesting an evolutionary pathway to alter P. aeruginosa quorum-sensing regulons.

|
Scooped by
mhryu@live.com
April 22, 11:27 PM
|
Ocean microbial communities are made up of thousands of diverse taxa whose metabolic demands set the rates of both biomass production and degradation. Thus, these microscopic organisms play a critical role in ecosystem dynamics, global carbon cycling, and climate. While we have frameworks for relating phytoplankton diversity to rates of carbon fixation, our knowledge of how variations in heterotrophic microbial populations drive changes in carbon cycling is in its infancy. Here, we leverage global metagenomic datasets and metabolic models to identify a set of metabolic niches with distinct growth strategies. These groupings provide a simplifying framework for describing microbial communities in different oceanographic regions and for understanding how heterotrophic microbial populations function. This framework, predicated directly on metabolic capability rather than taxonomy, will enable us to tractably link heterotrophic diversity directly to biogeochemical rates in large scale ecosystem models.

|
Scooped by
mhryu@live.com
April 22, 10:25 PM
|
Gut microbial community composition varies from one person to another. Potentially, this means the ecological interactions experienced by individual strains or species also vary among microbiomes of different people. However, testing this directly in human microbiomes and identifying ecological drivers involved are challenging. Here, we use replicated anaerobic microcosms to quantify variability of population growth for a key commensal species among microbiome samples from different individuals and to identify underlying intra- and interspecific interactions. In a reciprocal transplant experiment, both absolute and relative growth performance of different E. coli strains varied among gut microbiome samples from healthy individuals. This was partly explained by intraspecific competition: growth performance of individual E. coli strains was associated with displacement of resident conspecifics. However, the determinants of E. coli growth varied among samples. In one microbiome sample with a distinctive taxonomic composition, culture acidification by resident microbes impaired growth of all E. coli strains. We identified a strain of Clostridium butyricum contributing to this effect and showed that transferring it into other microbiomes predictably altered pH, fermentation product profiles (butyrate accumulation and acetate/lactate depletion), and population growth of other species including E. coli, thereby reshaping overall taxonomic composition. Our results suggest natural interindividual gut microbiome variation translates to variable ecological interactions with incoming bacteria, but these dynamics can be manipulated by a generalizable interspecies interaction.

|
Scooped by
mhryu@live.com
April 22, 10:16 PM
|
We present nanopore adapter-enriched Cas9-targeted sequencing (nAECATS), a method permitting inexpensive, ultra-deep, selective long-read sequencing of targeted regions in native, unamplified DNA. This method modifies previous Cas9-targeted sequencing approaches through the inclusion of a bead-based capture step that exploits the poly(T)8 stretch within the R10.4.1 ligation adapter for additional purification. Testing on a 10 kb Bacteroides fragilis genomic region achieved 90% on-target yield with 51,000× coverage from a single Flongle flow cell (353-fold increase versus whole-genome sequencing). Applied to a variable-length (>20 kb) Staphylococcus aureus genomic target containing dynamic gene amplifications conferring antibiotic resistance, nAECATS achieved 46,000× coverage and 74% on-target yield, revealing 2–4 tandem amplifications at single-cell resolution. While efficiency decreased with longer fragments (up to 41 kb), substantial enrichment improvements were demonstrated. We anticipate that nAECATS ultra-deep sequencing will find broad application for a wide range of biological questions in pro- and eukaryotic (epi)genomics and microbiology.

|
Scooped by
mhryu@live.com
April 22, 9:48 PM
|
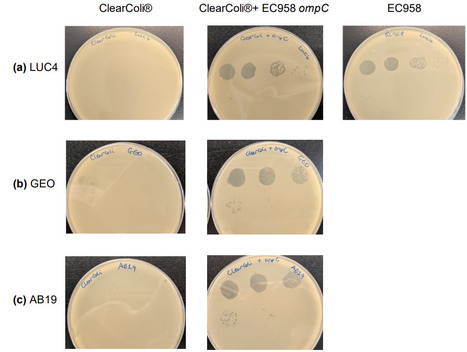
Phage therapeutics are re-emerging as adjuncts or alternatives to antibiotics and their clinical translation will be enhanced with production methods that minimise downstream processing. We evaluated whether an endotoxin-reduced E. coli strain developed for production of recombinant proteins, ClearColi®, can serve as a useful, safe phage production host without compromising yield and whether targeted receptor complementation can expand its utility. The parent strain BL21(DE3), and its lipid A modified derivative, ClearColi®, were compared with respect to infection and generation of phage. Across a panel of 31 phage, a similar host range was observed between BL21(DE3) and ClearColi. To expand host range ompC was genetically engineered into the chromosome of ClearColi, thereby adding OmpC-dependent phage to its production capacity. Production metrics were broadly comparable between the hosts; efficiency of plating and final titres for representative phage were not significantly different; burst size varied by phage but without consistent host bias. Endotoxin activity in ClearColi®-propagated lysates was reduced by over 1000-fold relative to BL21(DE3), reaching the low hundreds of endotoxin units (EU) versus hundreds of thousands for BL21(DE3). Intravesical administration of ClearColi- derived phage (LUC4) into pigs elicited no clinical abnormalities and no significant increases in circulating cytokines up to 48 hours after administration. ClearColi® allows efficient production of diverse phage with low endotoxin, reducing the requirement for downstream processing. Although its minimal LPS reduces its capacity for producing some LPS-dependent phage and its growth is slower than BL21(DE3), requiring optimisation for maximal phage titre, the safety and simplified manufacturing process support further development of endotoxin modified strains for phage production.

|
Scooped by
mhryu@live.com
April 22, 10:29 AM
|
CRISPR-Cas are widespread adaptive immune systems that protect bacteria and archaea from mobile genetic elements such as bacteriophages. Metagenomic sequencing identified CRISPR-Cas systems in phage genomes; however, their functions remain largely unknown. Here, we present Cas12r-CRISPR, a novel type V CRISPR-Cas system encoded by Lalka phages infecting thermophilic Thermus bacteria. We determined Cas12r-CRISPR PAM consensus sequence and crRNA structure and showed, that when provided with appropriate spacers and expressed in Thermus thermophilus, Cas12r-CRISPR efficiently interferes with plasmid transformation as well as infection by diverse Thermus phages. In the course of Lalka phage infection, the Cas12r-CRISPR locus is expressed with middle phage genes and its transcripts are among the most abundant phage RNAs. Notably, most Cas12r-CRISPR spacers target integrative mobile elements widespread in Thermus genomes. Both Lalka phages and targeted integrative mobile elements use host tRNA genes as attachment sites. We therefore propose that Cas12r-CRISPR participates in an inter-MGEs conflict.
|

|
Scooped by
mhryu@live.com
Today, 11:20 PM
|
Highly repetitive DNA sequences remain difficult to synthesize, assemble, and verify, limiting their use in synthetic biology. Here, we report a modular plasmid framework for the scalable, sequence-defined assembly of repetitive DNA. As a model system, plasmids encoding repeats of the pentapeptide Gly–Val–Gly–Val–Pro (GVGVP)n were constructed to produce elastin-like polypeptides (ELPs) with temperature-dependent solubility. A synthetic DNA fragment encoding GVGVP17 was incorporated into a plasmid architecture that enables iterative repeat amplification through a Gibson-based digest-and-assemble workflow. Sequential HindIII and BamHI digestion followed by Gibson Assembly increased repeat number (2n–1 per cycle) while preserving plasmid architecture, yielding constructs up to GVGVP1025, as verified by whole-plasmid sequencing. A superfolder GFP was added to the GVGVP library with expression of up to 513 repeats and functional characterization up to 257 repeats in E. coli NEB 5-alpha cells. These results establish a generalizable strategy for constructing large repetitive DNA sequences and encoding programmable protein polymers.

|
Scooped by
mhryu@live.com
Today, 11:00 PM
|
Metagenomic sequencing has transformed virus discovery; however, downstream bioinformatic analyses for viral identification, classification, and host prediction remain fragmented across multiple tools. Here, we present PhaBOX2, a major upgrade that extends the platform from a specialized bacteriophage identification tool to a comprehensive and integrated suite for viral sequence analysis. PhaBOX2 broadens its detection, taxonomic, and host prediction scope beyond phages to enable the characterization of archaeal and eukaryotic viruses. The updated workflow incorporates rigorous quality control and quantitative analyses, automatically removes host contamination, clusters sequences into viral operational taxonomic units, and performs phylogenetic analysis based on marker genes. In contrast to traditional “black–box” deep learning approaches, PhaBOX2 combines alignment–based strategies with machine–learning models under a “glass–box” design philosophy, providing interpretable intermediate evidence alongside final predictions to improve transparency and biological interpretability. Powered by a dedicated high–performance computing infrastructure, the server delivers a fully automated, end–to–end workflow, while achieving an ~80% reduction in processing time. PhaBOX2 thus provides a robust and user–friendly ecosystem for viral metagenomic analysis and is freely available at https://phage.ee.cityu.edu.hk/.

|
Scooped by
mhryu@live.com
Today, 7:25 PM
|
Antimicrobial resistance (AMR) has evolved into a severe global public health crisis, with plasmid-mediated horizontal gene transfer (HGT) serving as a core driver for the rapid dissemination of multidrug resistance (MDR). Traditional “bactericidal” antibiotic strategies impose strong selective pressure, failing to eradicate the root cause of resistance while accelerating the enrichment of resistant clones. “Plasmid curing”—a strategy that specifically eliminates resistance plasmids to restore antibiotic susceptibility—has emerged as a promising paradigm shift. While early synthetic curing agents suffered from severe cytotoxicity, natural products (e.g., alkaloids, quinones, terpenoids) exhibit unique potential owing to their structural diversity and multi-target profiles. This review systematically elucidates the molecular mechanisms by which natural products achieve plasmid eradication, including the disruption of Rep-ori replication initiation, interference with ParA-ParB partitioning dynamics, and the blockade of conjugation via type IV secretion system (T4SS) and quorum sensing (QS) inhibition. Crucially, we critically evaluate the methodological workflows—from high-throughput screening to absolute quantitative PCR—necessary to strictly differentiate true in vivo plasmid curing from mere selective bactericidal artifacts. Furthermore, we address current translational bottlenecks, particularly the “therapeutic window paradox,” and highlight how integrating advanced nanotechnology, artificial intelligence (AI)-guided drug discovery, and CRISPR-Cas9 synergies will propel the field forward. By shifting the therapeutic paradigm from violent “bacterial killing” to ecologically intelligent “genetic disarmament,” natural plasmid-curing agents offer a vital, adjunctive solution for safeguarding the lifespan of legacy antibiotics.

|
Scooped by
mhryu@live.com
Today, 6:24 PM
|
The fission yeast Schizosaccharomyces pombe is a prominent model organism widely used to investigate fundamental cellular mechanisms. In addition to S. pombe, the genus Schizosaccharomyces includes six other species—S. octosporus, S. japonicus, S. cryophilus, S. osmophilus, S. lindneri, and S. versatilis. These fission yeast species share a common ancestor from which the genus diversified over more than 200 million years. This extensive evolutionary divergence provides opportunities for comparative genomics. Here, we present the Schizosaccharomyces orthogroup (SOG) resource, a web platform developed from our high-quality genome assemblies, gene annotations, and orthology assignments. Most fission yeast genes are assigned to one of over 5,000 orthogroups. The platform enables users to visualize orthogroup sequence alignments and phylogenetic trees, retrieve coding and flanking sequences, and explore the conservation of local synteny. This resource will benefit researchers focusing on individual genes as well as those investigating gene evolution at broader scales. It is freely accessible at https://www.sogweb.org.

|
Scooped by
mhryu@live.com
Today, 9:24 AM
|
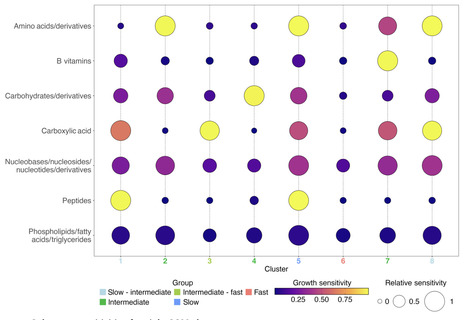
Metabolic phenotypes vary within microbial species, yet how such variation is organized remains unclear. Diversification in carbon‑source utilization, in particular, often appears idiosyncratic, showing weak correspondence to phylogeny or simple gene content. Here, we combine quantitative growth phenotyping of natural E. coli isolates across 32 carbon sources with diversification observed during de novo laboratory evolution, together with a reaction-level description of metabolic similarity. Despite deep phylogenetic divergence, growth‑rate profiles varied independently of lineage. Instead, growth rates across carbon sources covaried in recurrent modular patterns aligned with similarities in required metabolic reactions. Closely analogous modular relationships re-emerged during de novo evolution, indicating parallel diversification across evolutionary contexts. Growth-rate variation in natural and experimentally evolved datasets collapsed onto a shared low-dimensional variance structure. Together, our results indicate that quantitative metabolic phenotypes vary along a limited set of recurring, module-linked axes, providing an organizational perspective on intraspecific metabolic diversity despite weak phylogenetic signal.

|
Scooped by
mhryu@live.com
Today, 12:48 AM
|
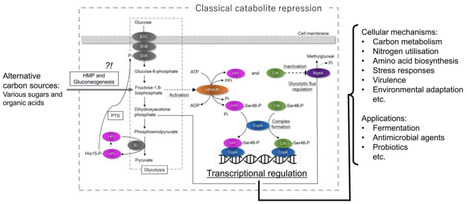
Carbon catabolite repression (CCR) in Gram‑positive bacteria is a sophisticated, evolutionarily tuned regulatory network that integrates carbon availability with global cellular physiology. This review traces the development from initial observations of CCR to the molecular elucidation of catabolite control protein A (CcpA)-mediated control, describing the roles of HPr, Crh, and HPrK/P in sensing glycolytic flux and transcriptional regulation. CCR ensures the preferential utilization of the most efficient carbon source while simultaneously suppressing unnecessary catabolic pathways, thereby regulating a wide range of biological processes. Species‑specific variations in CCR underscore the ecological and evolutionary diversity of this system. Besides metabolism, CCR influences pathogenicity in some pathogenic bacteria, linking nutrient sensing to host interaction strategies. Its multidimensional impacts extend to biotechnology, fermentation optimisation, probiotic performance, and potential antimicrobial strategies targeting nutrient prioritisation. As omics‑based approaches reveal CCR as a global integrator of carbon, nitrogen, and stress pathways, future research must unravel species‑specific architectures, regulatory nuances, and evolutionary implications to understand and harness the complex regulatory system.

|
Scooped by
mhryu@live.com
Today, 12:20 AM
|
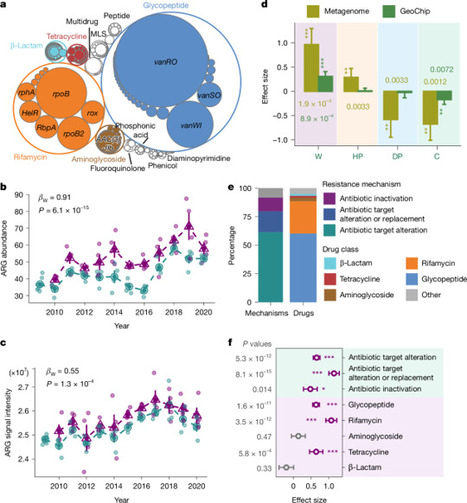
Soils are critical reservoirs of antibiotic-resistance genes (ARGs) which are strongly shaped by microbial interactions and environmental conditions and are therefore highly sensitive to disturbance. Although climate warming is recognized as one of the most significant disturbances to microbial communities and their functions, its impacts on soil resistomes remain poorly understood. Here we investigated the effects of decade-long experimental warming on ARGs in grassland soils using integrated experimental and computational approaches. Our results revealed that ARG abundance substantially increased (23.9%) under warming—particularly glycopeptide- and rifamycin-resistance genes. Warming specifically enriched Actinomycetota hosts, including various potential plant pathogens, and enhanced ARG mobility. Large-scale unprecedented isolates-based phenotypic analyses also validated that warming increased bacterial resistance to multiple antibiotics. Further mechanistic analyses revealed that warming increased ARG abundance primarily through co-selection of resistance genes physically linked to adaptive traits (for example, thermal tolerance and nitrogen assimilation) and positive selection for thermal tolerance genes, which could be further amplified via horizontal gene transfer. Together, these findings convincingly demonstrate that climate warming substantially accelerates soil antibiotic resistance at genomic, ecological and evolutionary levels, with broad implications for public health and environmental sustainability in a warming world. Decade-long warming increased soil antibiotic-resistance genes by about 24%, enriching resistant microbes and gene transfer, driven by selection for thermal tolerance and linked traits, with implications for public health.

|
Scooped by
mhryu@live.com
April 22, 11:35 PM
|
The prediction of biological sequence properties has traditionally relied on alignment-based methods that assume evolutionary homology and depend on curated reference databases. This, in turn, limits scalability and sensitivity for large or heterogeneous datasets, remote homologs, short sequences, and rapidly evolving genomic regions. Although Machine-Learning (ML) approaches offer alignment-free alternatives, their broader adoption is limited by: (i) the lack of standardized, externally validated benchmark models across diverse datasets, and (ii) the technical expertise required for feature engineering, model selection, and evaluation. Automated machine learning (AutoML) alleviates these challenges by systematically optimizing representations and models with minimal user intervention. However, most existing frameworks prioritize task-specific model construction and lack mechanisms for preserving trained models as persistent, comparable benchmarks. We introduce BioAutoML-FAST, an end-to-end web platform for automated ML analysis of nucleotide and amino acid sequences. It supports both classification and regression tasks and automates feature extraction, model training, and evaluation without requiring prior user expertise. Uniquely, it serves as a community benchmarking resource, hosting a continuously expanding repository of reusable, standardized models (currently 60) for genomic, transcriptomic, and proteomic applications. Extensive validation on independent datasets demonstrates performance comparable to or exceeding that of state-of-the-art methods, including protein language models such as ESM-2. BioAutoML-FAST is available at https://bioautoml.icmc.usp.br/. This website is free and open to all users, and there is no login requirement.

|
Scooped by
mhryu@live.com
April 22, 10:37 PM
|
Bacterial pathogens adapt rapidly to clinical and within-host selective pressures. Insertion sequences (IS) are transposable elements that can contribute to pathogenic adaptation, but their activity and consequences in contemporary clinical populations are not well characterized. Here, combining large-scale genomic surveys with long-read sequencing of clinical isolates and longitudinal gut metagenomes, we quantify pathogen IS dynamics from global patterns to within-host evolution. Across 19,485 publicly available high-contiguity ESKAPEE pathogen genomes, Enterococcus faecium genomes are the most IS dense, dominated by replicative ISL3 family elements, which have proliferated in clinical lineages over the past 30 years. We find extensive chromosomal structural variation, largely involving ISL3, within a new single-hospital collection of bloodstream isolates. Long-read metagenomic sequencing of 28 longitudinal stool samples from 12 haematopoietic cell transplantation (HCT) recipients demonstrates within-host IS dynamics and their regulatory consequences. In one patient, an ISL3 insertion upstream of a folate transporter formed a strong promoter, increasing transcription and improving relative fitness under folate limitation. Enhanced folate scavenging may enable E. faecium to thrive in the setting of microbiome collapse, which is common in HCT and other critically ill patients. Together, these results show that a recent ISL3 expansion is driving rapid evolution in healthcare-associated E. faecium, with consequences for its metabolic fitness that may help explain its increasing clinical burden. Several other pathogens also show elevated IS loads in our survey, which suggests that IS expansion-mediated evolution might be more broadly relevant. Over three decades, rapid expansion of the transposable element ISL3 has reshaped Enterococcus faecium, which helps to explain this pathogen’s growing clinical threat.

|
Scooped by
mhryu@live.com
April 22, 10:22 PM
|
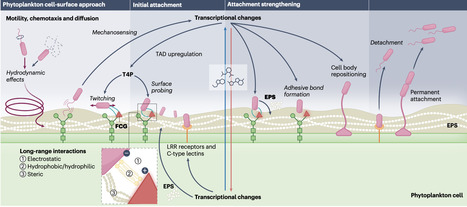
Phytoplankton are responsible for approximately half of Earth’s net primary production and, together with heterotrophic bacteria—the main consumers of organic matter—play a pivotal role in biogeochemical cycles. Their key ecological importance has led to growing interest in the interactions between these two groups. Yet, our understanding of the microscale mechanisms driving these interactions remains limited. Recent work highlighted the contribution of bacterial motility and chemotaxis to promoting encounters and nutrient exchange between bacteria and phytoplankton. In contrast, the ecological role of bacterial attachment—an important adaptation enabling bacteria to establish the closest contact with their phytoplankton host and retain it over extended periods of time—remains less explored. Here we describe the current evidence and understanding of bacterial attachment to phytoplankton and highlight recent insights from single-cell studies. Motivated by the implications for large-scale ecosystem processes, we discuss promising research avenues to further unveil the ecological relevance of bacterial attachment to phytoplankton. Here the authors review current understanding of the mechanistic marine microbial interactions that underpin large-scale ecosystem processes and biogeochemical cycling.

|
Scooped by
mhryu@live.com
April 22, 10:10 PM
|
Mutualism in the symbiosis between arbuscular mycorrhizal fungi and plants is based upon the exchange of carbon for soil minerals, with phosphate being of central importance. The exchange of nutrients occurs when the fungus transiently colonizes root cells, producing hyphal structures called arbuscules. The movement of phosphate from fungus to plant is well established, however its coordination and regulation at the ephemeral arbuscules remains elusive. Here, non-invasive imaging captures the complete growth and collapse of the arbuscules in unprecedented resolution, revealing heterogeneity in arbuscule development. Tracking the dynamics of rice PHosphate Transporter 1;11 (OsPHT1;11/ PT11) as a proxy for symbiotic phosphate transport shows consistent localization across diverse arbuscules. However, we uncover phosphate-responsive variability in PT11 abundance, representing an essential, cellular-level layer of nutrient regulation. Such plasticity in arbuscule phosphate uptake capacity evidences uncoupling of arbuscule presence and arbuscule function, thereby demonstrating that arbuscules are not identical units of nutrient exchange. This study uncovers the previously-hidden dynamics of nutrient exchange structures central to the symbiosis between plants and arbuscular mycorrhizal fungi, revealing highly variable development, lifespans and phosphate transport capacities.

|
Scooped by
mhryu@live.com
April 22, 5:40 PM
|
Microbial diversity is often assumed to be limited by the number of available resources, yet many communities persist well beyond that expectation. Understanding the mechanisms that enable such coexistence remains a central question in microbial ecology. Here, using a four-species bacterial consortium, we asked whether coexistence can emerge from interactions between species rather than from the external environment alone. Across 31 simple nutrient conditions, including 16 single-resource environments, all four species persisted and repeatedly reached stable coexistence. We then chose 27 additional conditions to further probe the boundaries of coexistence by varying resource concentrations, temporal dynamics, nutrient complexity and relief of auxotrophy-associated dependencies, and only observed the extinction of one species in one of these conditions. Although the community composition in each environment was largely shaped by species' fitness on the supplied resources, experimental assays and consumer-resource modeling showed that the coexistence was not explained by resource supply, but rather by cross-feeding and niche partitioning of metabolic byproducts. These metabolic interactions were strong enough to sustain coexistence even for species unable to use the supplied resources directly. Furthermore, robust coexistence across environments appears to be an emergent property of microbial communities, ingrained in members' metabolic byproduct profiles and niche differences. Our findings demonstrate how microbes can increase the chemical complexity of their environment sufficiently to maintain coexistence well beyond what is expected from external resource supply.
|
 Your new post is loading...
Your new post is loading...