Your new post is loading...
 Your new post is loading...

|
Scooped by
Gilbert C FAURE
April 4, 2024 3:46 AM
|
Analysis of the blood revealed various proteins involved in immunity, inflammation, coagulation, and stress response among other findings.

|
Suggested by
LIGHTING
October 4, 2023 7:08 AM
|
ABSTRACT Rapid antigen tests (RATs) have become an invaluable tool for combating the COVID-19 pandemic. However, concerns have been raised regarding the ability of existing RATs to effectively dete...

|
Scooped by
Gilbert C FAURE
July 19, 2023 3:53 AM
|
A new review of literature on #COVID19-sniffing canines shows #dogs outperformed or matched reverse-transcription polymerase chain reaction (#RTPCR) tests or…

|
Scooped by
Gilbert C FAURE
January 24, 2023 3:42 AM
|
BackgroundCOVID-19 is an infectious disease caused by the SARS-CoV-2 virus that spreads easily between people in a similar way to the common cold or ‘flu’. Most people with COVID-19 have a mild-to-moderate respiratory illness, and some may have no symptoms (asymptomatic infection). Others experience severe symptoms and need specialist treatment and intensive care.In response to COVID-19 infection, the immune system develops proteins called antibodies that can attack the virus as it circulates in their blood. People who have been vaccinated against COVID-19 also produce these antibodies against the virus. Tests are available to detect antibodies in peoples' blood, which may indicate that they currently have COVID-19 or have had it previously, or it may indicate that they have been vaccinated (although this group was not the focus of this review).Why are accurate tests important?Accurate testing allows identification of people who need to isolate themselves to prevent the spread of infection, or who might need treatment for their infection. Failure of diagnostic tests to detect infection with COVID-19 when it is present (a false negative result) may delay treatment and risk further spread of infection to others. Incorrect diagnosis of COVID-19 when it is not present (a false positive result) may lead to unnecessary further testing, treatment, and isolation of the person and close contacts. Accurate identification of people who have previously had COVID-19 is important in measuring disease spread and assessing the success of public health interventions.To determine the accuracy of an antibody test in identifying COVID-19, test results are compared in people known to have (or have had) COVID-19 and in people known not to have (or have had) COVID-19. The criteria used to determine whether people are known or not known to have COVID-19 is called the ‘reference standard’. Many studies use a test called reverse transcriptase polymerase chain reaction (RT-PCR) as the reference standard, with samples taken from the nose and throat. Additional tests that can be used include measuring symptoms, like coughing or high temperature, or ‘imaging’ tests like chest X-rays. People known not to have COVID-19 are sometimes identified from stored blood samples taken before COVID-19 existed, or from patients with symptoms confirmed to be caused by other diseases.What did the review study?We wanted to find out whether antibody tests:- are able to diagnose infection in people with or without symptoms of COVID-19, and- can be used to find out if someone has already had COVID-19.The studies we included in our review looked at three types of antibodies. Most commonly, antibody tests measure two types known as IgG and IgM, but some tests only measure a single type of antibody or different combinations of the three types of antibodies (IgA, IgG, IgM).What did we do?We looked for studies that measured the diagnostic accuracy of antibody tests to detect current or past COVID-19 infection and compared them with reference standard criteria. Since there are many antibody tests available, we included studies assessing any antibody test compared with any reference standard. People could be tested in hospital or in the community. The people tested may have been confirmed to have, or not to have, COVID-19 infection, or they may be suspected of having COVID-19.Study characteristicsWe found 178 relevant studies. Studies took place in Europe (94), Asia (45), North America (35), Australia (2), and South America (2).Seventy-eight studies included people who were in hospital with suspected or confirmed COVID-19 infection and 14 studies included people in community settings. Several studies included people from multiple settings (35) or did not report where the participants were recruited from (39).One hundred and forty-one studies included recent infection cases (mainly week 1 to week 3 after onset of symptoms), and many also included people tested later (from day 21 onwards after infection) (117).Main resultsIn participants that had COVID-19 and were tested one week after symptoms developed, antibody tests detected only 27% to 41% of infections. In week 2 after first symptoms, 64% to 79% of infections were detected, rising to 78% to 88% in week 3. Tests that specifically detected IgG or IgM antibodies were the most accurate and, when testing people from 21 days after first symptoms, they detected 93% of people with COVID-19. Tests gave false positive results for 1% of those without COVID-19.Below we illustrate results for two different scenarios.If 1000 people were tested for IgG or IgM antibodies during the third week after onset of symptoms and only 20 (2%) of them actually had COVID-19:- 26 people would test positive. Of these, 8 people (31%) would not have COVID-19 (false positive result).- 974 people would test negative. Of these, 2 people (0.2%) would actually have COVID-19 (false negative result).If 1000 people with no symptoms for COVID-19 were tested for IgG antibodies and 500 (50%) of them had previously had COVID-19 infection more than 21 days previously:- 455 people would test positive. Of these, 6 people (1%) would not have been infected (false positive result).- 545 people would test negative. Of these, 51 (9%) would actually have had a prior COVID-19 infection (false negative result).How reliable were the results of the studies of this review?We have limited confidence in the evidence for several reasons. The number of samples contributed by studies for each week post-symptom onset was often small, and there were sometimes problems with how studies were conducted. Participants included in the studies were often hospital patients who were more likely to have experienced severe symptoms of COVID-19. The accuracy of antibody tests for detecting COVID-19 in these patients may be different from the accuracy of the tests in people with mild or moderate symptoms. It is not possible to identify by how much the test results would differ in other populations.Who do the results of this review apply to?A high percentage of participants were in hospital with COVID-19, so were likely to have more severe disease than people with COVID-19 who were not hospitalised. Only a small number of studies assessed these tests in people with no symptoms. The results of the review may therefore be more applicable to those with severe disease than people with mild symptoms.Studies frequently did not report whether participants had symptoms at the time samples were taken for testing making it difficult to fully separate test results for early-phase infection as opposed to later-phase infections.The studies in our review assessed several test methods across a global population, therefore it is likely that test results would be similar in most areas of the world.What are the implications of this review?The review shows that antibody tests could have a useful role in detecting if someone has had COVID-19, but the timing of test use is important. Some antibody tests may help to confirm COVID-19 infection in people who have had symptoms for more than two weeks but who have been unable to confirm their infection using other methods. This is particularly useful if they are experiencing potentially serious symptoms that may be due to COVID-19 as they may require specific treatment. Antibody tests may also be useful to determine how many people have had a previous COVID-19 infection. We could not be certain about how well the tests work for people who have milder disease or no symptoms, or for detecting antibodies resulting from vaccination.How up-to-date is this review?This review updates our previous review. The evidence is up-to-date to September 2020. Authors' conclusions: Some antibody tests could be a useful diagnostic tool for those in whom molecular- or antigen-based tests have failed to detect the SARS-CoV-2 virus, including in those with ongoing symptoms of acute infection (from week three onwards) or those presenting with post-acute sequelae of COVID-19. However, antibody tests have an increasing likelihood of detecting an immune response to infection as time since onset of infection progresses and have demonstrated adequate performance for detection of prior infection for sero-epidemiological purposes. The applicability of results for detection of vaccination-induced antibodies is uncertain. Read the full abstract...

|
Scooped by
Gilbert C FAURE
March 16, 2021 8:43 AM
|
medRxiv - The Preprint Server for Health Sciences

|
Scooped by
Gilbert C FAURE
March 9, 2021 1:57 PM
|
The new test, which has been authorized by the FDA, detects prior COVID-19 infection with T-cells and may be a game changer for some COVID-19 long haulers.

|
Scooped by
Gilbert C FAURE
March 2, 2021 4:38 AM
|
Saliva may be a reliable and cost-efficient way to diagnose COVID-19, according to a study published recently in the Journal of Evidence-Based Dental Practice. Saliva-based tests were found to be roughly as reliable as nasopharynx or throat swabs.

|
Scooped by
Gilbert C FAURE
February 13, 2021 10:54 AM
|
The FDA’s Experience with Covid-19 Antibody Tests Early in the pandemic, the FDA recognized that ensuring access to antibody tests could advance understanding of Covid-19 and help inform th

|
Rescooped by
Gilbert C FAURE
from Mucosal Immunity
January 19, 2021 11:05 AM
|
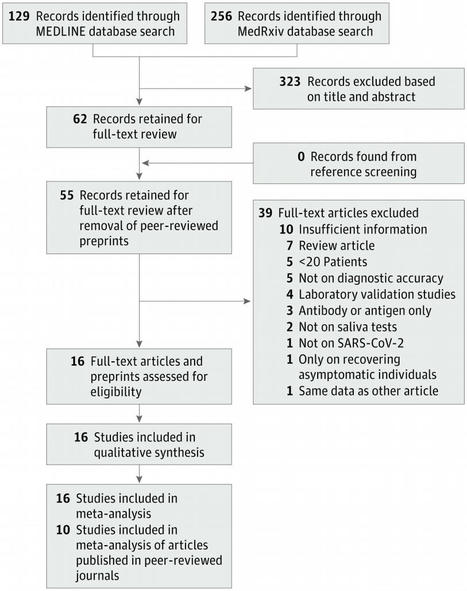
This systematic review and meta-analysis assess the diagnostic accuracy of saliva nucleic acid amplification tests for diagnosis of coronavirus disease 2019.

|
Scooped by
Gilbert C FAURE
January 13, 2021 6:44 AM
|
(1) Background: The detection of SARS-CoV-2 RNA in nasopharyngeal samples through real-time reverse transcription-polymerase chain reaction (RT-PCR) is considered the standard gold method for the diagnosis of SARS-CoV-2 infection. Antigen detection (AD) tests are more rapid, less laborious, and less expensive alternatives but still require clinical validation. (2) Methods: This study compared the clinical performance of five AD tests, including four rapid AD (RAD) tests (biotical, Panbio, Healgen, and Roche) and one automated AD test (VITROS). For that purpose, 118 (62.8%) symptomatic patients and 70 (37.2%) asymptomatic subjects were tested, and results were compared to RT-PCR. (3) Results: The performance of the RAD tests was modest and allowed us to identify RT-PCR positive patients with higher viral loads. For Ct values ≤25, the sensitivity ranged from 93.1% (95% CI: 83.3–98.1%) to 96.6% (95% CI: 88.1–99.6%), meaning that some samples with high viral loads were missed. Considering the Ct value proposed by the CDC for contagiousness (i.e., Ct values ≤33) sensitivities ranged from 76.2% (95% CI: 65.4–85.1%) to 88.8% (95% CI: 79.7–94.7%) while the specificity ranged from 96.3% (95% CI: 90.8–99.0%) to 99.1% (95% CI: 95.0–100%). The VITROS automated assay showed a 100% (95% CI: 95.5–100%) sensitivity for Ct values ≤33, and had a specificity of 100% (95% CI: 96.6–100%); (4) Conclusions: Compared to RAD tests, the VITROS assay fully aligned with RT-PCR for Ct values up to 33, which might allow a faster, easier and cheaper identification of SARS-CoV-2 contagious patients.

|
Scooped by
Gilbert C FAURE
December 28, 2020 7:59 AM
|
Researchers from the Polytechnic University of Valencia, in collaboration with specialists from the University of Valencia (UV), have come up with a new system based on a high-sensitivity mass biosensor that continuously monitors signals for the direct detection of SARS-CoV-2 in environmental...

|
Scooped by
Gilbert C FAURE
December 16, 2020 1:39 PM
|
from The Conversation-- this post authored by Rodney E.Rohde, Texas State University...

|
Scooped by
Gilbert C FAURE
December 1, 2020 7:51 AM
|
BD Biosciences provides you with the tools and support required to enable your COVID-19 research. Learn more about our solutions and their applications here.
|

|
Scooped by
Gilbert C FAURE
March 24, 2024 6:13 AM
|
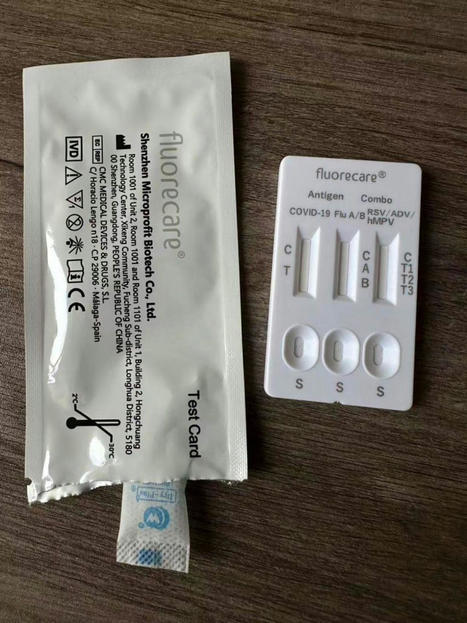
New product news!💡Ever heard of a 6 in 1 combo test?
Check this CE marked Fluorecare test!
🦠Detecting covid, influenza A&B, RSV, ADV and hMPV at the same…

|
Scooped by
Gilbert C FAURE
July 27, 2023 2:40 AM
|
Yeon Ju Seo, M.D., Inseong Oh, M.D., Minjeong Nam, M.D., Ph.D., Sue Shin, M.D., Ph.D., Eun Youn Roh, M.D., Ph.D., and Eun Young Song, M.D., Ph.D.. Ann Lab Med 2023;43:596-604.https://doi.org/10.3343/alm.2023.43.6.596...

|
Scooped by
Gilbert C FAURE
February 11, 2023 3:01 AM
|
While molecular testing with real-time polymerase chain reaction (RT-PCR) remains the gold-standard test for COVID-19 diagnosis and screening, more rapid or affordable molecular and antigen testing options have been developed.

|
Scooped by
Gilbert C FAURE
December 22, 2021 2:15 AM
|
Diagnostics have proven to be crucial to the COVID-19 pandemic response. There are
three major methods for the detection of SARS-CoV-2 infection and their role has evolved
during the course of the pandemic. Molecular tests such as PCR are highly sensitive
and specific at detecting viral RNA, and are recommended by WHO for confirming diagnosis
in individuals who are symptomatic and for activating public health measures. Antigen
rapid detection tests detect viral proteins and, although they are less sensitive
than molecular tests, have the advantages of being easier to do, giving a faster time
to result, of being lower cost, and able to detect infection in those who are most
likely to be at risk of transmitting the virus to others.

|
Scooped by
Gilbert C FAURE
March 16, 2021 5:17 AM
|
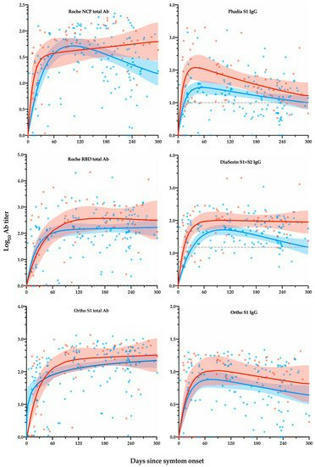
Several studies have described the long-term kinetics of anti-SARS-CoV-2 antibodies but long-term follow-up data, i.e., >6 months, are still sparse. Additionally, the literature is inconsistent regarding the waning effect of the serological response. The aim of this study was to explore the temporal dynamic changes of the immune response after SARS-CoV-2 infection in hospitalized and non-hospitalized symptomatic patients over a period of 10 months. Six different analytical kits for SARS-CoV-2 antibody detection were used. Positivity rates, inter-assay agreement and kinetic models were determined. A high inter-individual and an inter-methodology variability was observed. Assays targeting total antibodies presented higher positivity rates and reached the highest positivity rates sooner compared with assays directed against IgG. The inter-assay agreement was also higher between these assays. The stratification by disease severity showed a much-elevated serological response in hospitalized versus non-hospitalized patients in all assays. In this 10-month follow-up study, serological assays showed a clinically significant difference to detect past SARS-CoV-2 infection with total antibody assays presenting the highest positivity rates. The waning effect reported in several studies should be interpreted with caution because it could depend on the assay considered.

|
Scooped by
Gilbert C FAURE
March 3, 2021 2:53 AM
|
We describe robust correlations across six SARS‐CoV‐2 serology assays detecting distinct antibody features. Our comprehensive analyses provide important insights into SARS‐CoV‐2 humoral immunit

|
Scooped by
Gilbert C FAURE
March 1, 2021 8:04 AM
|
webinars and international
webinars of the Portuguese Society of Clinical Pathology.

|
Scooped by
Gilbert C FAURE
January 28, 2021 6:46 AM
|
Among the key findings, which have not yet been peer-reviewed, are: Individuals who have asymptomatic or mild disease show a robust immune response early on

|
Scooped by
Gilbert C FAURE
January 18, 2021 6:23 AM
|

|
Scooped by
Gilbert C FAURE
January 5, 2021 2:41 PM
|
COVID-19 is a unique disease characterized by extensive lung thrombosis, long-term
persistence of viral RNA in pneumocytes and endothelial cells, along with the presence
of infected cell syncytia. Several of COVID-19 features might be consequent to the persistence of virus-infected cells for the...

|
Scooped by
Gilbert C FAURE
December 17, 2020 5:12 AM
|

|
Scooped by
Gilbert C FAURE
December 12, 2020 2:32 PM
|
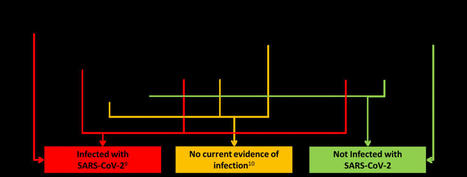
Proper interpretation of antigen test results is important for accurate clinical management of patients with suspected COVID-19. This guidance can help healthcare providers make the most effective use of antigen tests in different situations.
|

 Your new post is loading...
Your new post is loading...
 Your new post is loading...
Your new post is loading...