Your new post is loading...
 Your new post is loading...

|
Scooped by
Gilbert C FAURE
March 5, 2023 3:33 AM
|
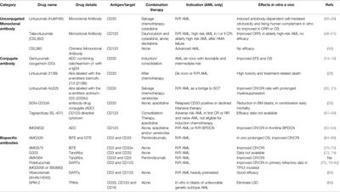
In the past few years, our improved knowledge of acute myeloid leukemia (AML) pathogenesis has led to the accelerated discovery of new drugs and the development of innovative therapeutic approaches. The role of the immune system in AML development, growth and recurrence has gained increasing interest. A better understanding of immunological escape and systemic tolerance induced by AML blasts has been achieved. The extraordinary successes of immune therapies that harness the power of T cells in solid tumors and certain hematological malignancies have provided new stimuli in this area of research. Accordingly, major efforts have been made to develop immune therapies for the treatment of AML patients. The persistence of leukemia stem cells, representing the most relevant cause of relapse, even after allogeneic stem cell transplant (allo-SCT), remains a major hurdle in the path to cure for AML patients. Several clinical trials with immune-based therapies are currently ongoing in the frontline, relapsed/refractory, post-allo-SCT and minimal residual disease/maintenance setting, with the aim to improve survival of AML patients. This review summarizes the available data with immune-based therapeutic modalities such as monoclonal antibodies (naked and conjugated), T cell engagers, adoptive T-cell therapy, adoptive-NK therapy, checkpoint blockade via PD-1/PD-L1, CTLA4, TIM3 and macrophage checkpoint blockade via the CD47/SIRPa axis, and leukemia vaccines. Combining clinica

|
Scooped by
Gilbert C FAURE
March 2, 2023 7:32 AM
|
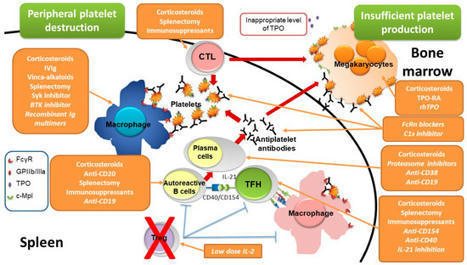
Immune thrombocytopenia (ITP) is a rare autoimmune disorder caused by peripheral platelet destruction and inappropriate bone marrow production. The management of ITP is based on the utilization of steroids, intravenous immunoglobulins, rituximab, thrombopoietin receptor agonists (TPO-RAs), immunosuppressants and splenectomy. Recent advances in the understanding of its pathogenesis have opened new fields of therapeutic interventions. The phagocytosis of platelets by splenic macrophages could be inhibited by spleen tyrosine kinase (Syk) or Bruton tyrosine kinase (BTK) inhibitors. The clearance of antiplatelet antibodies could be accelerated by blocking the neonatal Fc receptor (FcRn), while new strategies targeting B cells and/or plasma cells could improve the reduction of pathogenic autoantibodies. The inhibition of the classical complement pathway that participates in platelet destruction also represents a new target. Platelet desialylation has emerged as a new mechanism of platelet destruction in ITP, and the inhibition of neuraminidase could dampen this phenomenon. T cells that support the autoimmune B cell response also represent an interesting target. Beyond the inhibition of the autoimmune response, new TPO-RAs that stimulate platelet production have been developed. The upcoming challenges will be the determination of predictive factors of response to treatments at a patient scale to optimize their management.

|
Scooped by
Gilbert C FAURE
February 28, 2023 4:45 AM
|
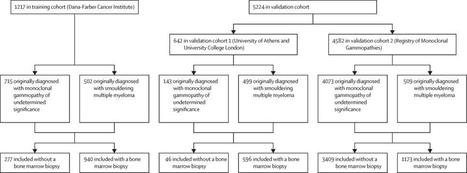
Use of the PANGEA models in clinical practice will allow patients with precursor disease
to receive more accurate measures of their risk of progression to multiple myeloma,
thus prompting for more appropriate treatment strategies.

|
Scooped by
Gilbert C FAURE
February 18, 2023 2:30 AM
|
HemaSphere, a premier hematology information resource, publishes results of highly relevant basic, translational and clinical research in hematology.

|
Scooped by
Gilbert C FAURE
February 11, 2023 3:01 AM
|
The National Health Service recently launched a world-first clinical trial that tests the safety of transfusing lab-grown blood cells into patients.

|
Scooped by
Gilbert C FAURE
January 21, 2023 11:20 AM
|
The detection of Minimal Residual Disease (MRD) by flow cytometry: lessons learned from the validation of Multiple Myeloma MRD assay for use in multi-centric clinical trials Flow Cytometry is a complex technology that allows multiparametric measurements on high numbers of single cells from complex mixtures. Given its high-throughput, flow cytometry is a highly sensitive technique that can be used for the detection of very rare cell populations. It is of no surprise that in the clinical field of cytometry, it has become a platform of choice for the detection of minimal residual disease (MRD) in various types of leukemia. The determination of MRD in cancer patients is of great clinical interest as it can be used as a good prognostic indicator of cancer and likelihood to relapse; thus MRD detection is increasingly being proposed as a potential surrogate endpoint for clinical trials. In the recent years, Labcorp Central Laboratory Services has developed and validated highly sensitive flow cytometry assays for MRD detection in Acute Myeloid Leukemia (AML), Chronic Lymphoid Leukemia (CLL) and Multiple Myeloma (MM) for primary/secondary endpoint use in clinical trials. In this presentation, Christèle will review the general use of flow cytometry in MRD detection in clinical trials and will take the audience through the CAP/CLIA validation of Labcorp MM MRD flow cytometry assay.

|
Scooped by
Gilbert C FAURE
December 24, 2022 7:10 AM
|
CC-98633-MM-001, Atalanta-1, KarMMa-2 Cohort 2a, CARTITUDE-2 Cohort B, CC‑95266‑MM‑001...

|
Scooped by
Gilbert C FAURE
December 20, 2022 1:49 PM
|
CAR T-cell Therapy Consults: Navigating a New Landscape is an interactive educational activity* that aims to assist clinicians in the identification and management of patients with various hematologic malignancies who may benefit from chimeric antigen receptor T-cell (CAR-T) therapy. CAR-T therapy has been rapidly integrated into clinical practice for various lymphomas, multiple myeloma, and B-cell acute lymphoblastic leukemia (B-ALL). CAR-T therapy is largely delivered at academic medical centers with stem cell transplant programs, but most patients in need of this therapy are often first evaluated and treated in community practices. In contrast to stem cell transplantation, which has been an established treatment for decades, practicing oncologists may not have direct experience with CAR-T therapy and the potential short- and long-term toxicities that their patients may experience. Additionally, the parallel development of other novel immune and small molecule therapies for hematologic malignancies has expanded the second-line treatment options for several hematologic malignancies, but there is a paucity of available data to guide the optimal sequencing of these novel treatments for relapsed disease. Thus, the current challenges that exist for the clinical management of CAR-T therapy include a lack of familiarity with both short- and long-term complications of treatment, optimal patient selection, and timing of CAR-T treatment. This educational activity focuses on CAR-T therapy for B-ALL, multiple myeloma, and B-cell lymphomas, and is delivered via both webinar and online modules. The webinar sessions are geographically based and led by experts from the Northeast, South, Midwest, and West regions of the United States (participants can attend any webinar that fits their schedule). The sessions focus on identification of patients for CAR-T therapy as well as details of treatment toxicities and best practices for long-term follow-up. Each session is 60 minutes, with the first 15 to 20 minutes reserved for an educational presentation and most of the session intended as an interactive, case-based discussion. Attendees are encouraged to bring real-world cases for additional discussion. Four additional sessions covering multiple myeloma and B-cell lymphoma will be presented beginning in January and ending in April 2023. Sessions are free of charge and participants are eligible to claim continuing medical education credits, as well as credits toward ABIM’s continuing certification program. All members of the multidisciplinary oncology team are welcome to attend. Additional details, including dates and registration, can be found on The France Foundation website (www.francefoundation. com/9155-car-t-cell-therapyconsults- navigating-a-newlandscape). Patrick M. Reagan, MD, is Associate Professor, Wilmot Cancer Institute, University of Rochester Medical Center, Rochester, NY. Kristen M. O’Dwyer, MD, is Associate Professor, Wilmot Cancer Institute, University of Rochester Medical Center, Rochester, NY. *This educational activity is the result of a collaboration between ASH, the American Society for Transplantation and Cellular Therapy, and The France Foundation, supported by educational grants from CRISPR Therapeutics, Janssen, Kite Pharma, and Novartis.

|
Scooped by
Gilbert C FAURE
December 20, 2022 8:30 AM
|
PURPOSE:Conventional hematology/oncology fellowship training is designed to foster careers in academic practice through intensive exposure to clinical and laboratory research. Even so, a notable pr...

|
Scooped by
Gilbert C FAURE
December 17, 2022 4:56 AM
|

|
Scooped by
Gilbert C FAURE
December 3, 2022 2:37 AM
|
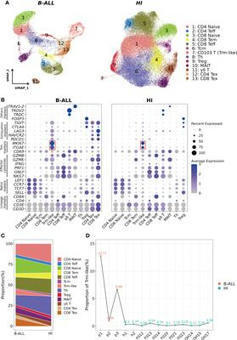
IntroductionThe character and composition of leukemia-related T cells are closely related to the treatment response and prognosis for patients. Though B cell-acute lymphoblastic leukemia (B-ALL) patients have benefited from immune-based approaches, such as chimeric antigen receptor T cells therapy, some of them still end with poor prognosis, especially for adult patients. Therefore, deep understanding of the developmental relationship between T cell subtypes in relation to B-ALL patient prognosis is urgently needed.MethodsWe analyzed the peripheral blood T cell single-cell RNA sequencing data of three B-ALL patients, using data from 11 healthy individuals as controls. In total, 16,143 and 53,701 T cells from B-ALL patients and healthy adults, respectively, were objectively analyzed for detailed delineation of 13 distinct T cell clusters. Cluster-specific genes were used as marker genes to annotate each T cell subtype.ResultsUnbiased analysis enabled the discovery of circulating CD103+ T cell (CD3+CD103+MKI67+), also defined as tissue-resident memory-like T (Trm-like) cell, populations were elevated in B-ALL patients, which expressed high level of cell proliferation and exhaustion related genes. In addition, cell fate trajectory analysis showed these Trm-like cells, which shared T-cell receptor (TCR) clonotypes with exhausted T (Tex) cells and effector T (Teff) cells, were supposed to transition into Teff cells; however, mainly transformed into Tex cells in leukemi

|
Scooped by
Gilbert C FAURE
November 27, 2022 6:34 AM
|
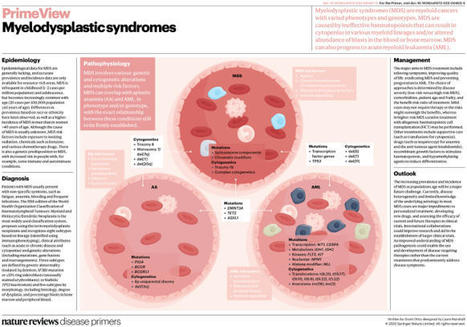
This PrimeView highlights the pathophysiology of myelodysplastic syndromes and summarizes their epidemiology, diagnosis and management. It accompanies the Primer on this topic by Liang and colleagues.

|
Scooped by
Gilbert C FAURE
November 14, 2022 4:57 AM
|
Among all controversies in hematology, the role of autologous stem cell transplant (ASCT) in first-line treatment for patients with peripheral T-cell lymphoma (PTCL) is one of the most long-lasting. Several hurdles have prevented a definitive solution being found to the problem. First, and compared to its B-cell lymphoma counterpart, no significant progress, except for brentuximab vedotin (BV) in ALK-positive or -negative anaplastic large cell lymphoma, has convincingly and significantly altered the course of PTCL during the last two decades.1 Such questions as to whether CHOP (cyclophosphamide, doxorubicin, vincristine and prednisone) alone or with etoposide (CHOEP) should be used frontline or whether stem cell transplant should be performed as first- or second-line treatment (if at all) are still hot topics in the field, whereas they might be considered completely outdated in other lymphoma subtypes. Second, despite tremendous efforts to better characterize the disease from molecular and pathological points of view, PTCL is still a highly heterogeneous disease. Combined with its rarity, this makes clinical research very difficult to conduct in order to conciliate the need for sufficient numbers of patients to be treated with homogeneous enough subtypes to be considered as one single disease. As a result and to date, more than five prospective trials and more than 20 retrospective studies have tried to address the benefit of ASCT in the first-line setting for PTCL.2-7 Let’s break the suspense: the study published in this issue of Haematologica by Garcia-Sancho and colleagues does not definitely answer the questions, but it does add a significant brick to the wall.8Compared to historical and more recent series showing a poor median progression-free survival of approximately 10 to 12 months in PTCL,9,10 the results from a prospective trial by d’Amore et al. published in 2012 convincingly demonstrated that six courses of CHOEP followed by ASCT in cases of partial or complete responses could yield progression-free survival of up to 44% at 5 years.3 Since then, numerous retrospective studies have produced conflicting results. For example, data from the Swedish registry were in favor of ASCT in multivariate analysis (for both progression-free survival and overall survival; number of patients in the analyses ~250) but were not adjusted for response status at the end of induction.11 A study by Cederleuf and colleagues based on Swedish and Danish patients (n=232), and limited to those reaching a complete response at the end of induction, did not find any survival advantage for ASCT in multivariate analysis.12 Our study from the Lymphoma Study Association (LYSA) also did not find any benefit associated with ASCT in patients (n=269) reaching a partial or complete response after six CHOP-like cycles of therapy when populations were matched based on a propensity-score.13 On the contrary, results based on patients in the prospective American COMPLETE registry (n=119) found a superiority of ASCT for patients in complete response.14 Similarly, Savage and colleagues recently reported on the outcome of patients with CD30+ PTCL in complete response following first-line treatment with BV-CHP (BV plus CHOP without vincristine) in the ECHELON-2 trial. Although ASCT consolidation was at the discretion of the treating investigator, post-hoc analysis showed a significantly longer progression-free survival for patients who received ASCT than for those who did not.15In fact, numerous irreducible statistical biases hamper proper retrospective comparisons of patients’ outcomes when it comes to stem cell transplant in general. Positive biases in favor of the procedure are that patients are usually younger, fitter, in better response at the end of induction, and have experienced lesser toxicity before ASCT; conversely, patients usually exhibit a more aggressive disease at diagnosis. As a result, positive and negative biases in favor and against ASCT make it very difficult to balance comparisons in retrospective studies. Usually, ways to control for those statistical biases are to perform matched-population comparisons, to conduct multivariate analyses, to use intent-to-treat groups (i.e., not comparing patients who actually receive ASCT or not; but comparing those for whom the physician decided before any treatment to go for stem cell transplant or not, information which is usually accessible through a review of medical charts), and to consider patients only in response after induction.The study by Garcia-Sancho et al. uses most of those approaches to try to avoid the usual pitfalls of retrospective comparisons when dealing with the procedure of stem cell transplantation. Imbalances in patients’ characteristics are “flattened” by using Cox multivariate analysis, only patients in complete response are considered for comparisons and, most importantly, the response must last at least 3 months to be considered. This circumvents another common problem of many studies since patients who can proceed to ASCT usually benefit from the so-called “guarantee-time bias”, i.e., that a patient needs to be in response until the transplant in the ASCT group, but not necessarily for so long in the non-ASCT group. However, the study is not performed based on an intent-to-treat decision by the local physician before any treatment, meaning that there might still be some uncontrolled biases between the two treatment groups. Finally, positron emission tomography/computed tomography is now frequently used for response assessment in PTCL, especially at the end of induction, but metabolic response was not considered in the study by Garcia-Sancho et al.Nevertheless, the authors report here on one of the largest retrospective cohort of patients (n=174) in first complete response from Spanish and Italian centers and show in multivariate analyses that ASCT is associated with better outcomes (both significantly prolonged progression-free and overall survival). Of note, a sensitivity analysis is performed to show that the benefit still exists when only ALK-negative anaplastic large cell lymphoma, angioimmunoblastic T-cell lymphoma and PTCL-not otherwise specified are taken into account, which are the usual histologies for which the role of ASCT has been extensively debated.In the next months, the LYSA academic group will enroll the first patients in the TRANSCRIPT (TRANSplantation after Complete Response In Patients with T-cell lymphoma) trial. This study will randomize 204 transplant-eligible patients (before any treatment) to six cycles of CHOP-like regimens (CHOP, CHOEP or BV-CHP) followed (n=102) or not (n=102) by ASCT for those in complete metabolic response. Only ALKnegative anaplastic large cell lymphoma, T follicular helper-phenotype PTCL and PTCL-not otherwise specified will be considered. Randomization will ensure theoretically similar baseline characteristics, ASCT allocation before induction will ensure intent-to-treat decision, and the positron emission tomography/computed tomography evaluation will ensure robust response assessment. The primary endpoint will be progression-free survival. Will the study finally put an end to an endless story in hematology? Will new therapeutic developments in first-line PTCL make the question obsolete by the time of the final analysis? Time will tell.FootnotesCorrespondenceemmanuel.bachy@chu-lyon.fr Disclosures EB has received honoraria from Kite, a Gilead Company, Bristol Myers Squibb, Novartis, Pfizer, Incyte; acted in a consultancy role for Takeda, Roche, and Gilead/Kite; received personal fees from Kite, a Gilead Company, Bristol Myers Squibb, Novartis, Pfizer; and received research funding from Amgen. References Horwitz S, O'Connor OA, Pro B. The ECHELON-2 trial: 5-year results of a randomized, phase III study of brentuximab vedotin with chemotherapy for CD30-positive peripheral T-cell lymphoma. Ann Oncol. 2022; 33(3):288-298. Google Scholar Corradini P, Tarella C, Zallio F. Long-term follow-up of patients with peripheral T-cell lymphomas treated up-front with high-dose chemotherapy followed by autologous stem cell transplantation. Leukemia. 2006; 20(9):1533-1538. Google Scholar d'Amore F, Relander T, Lauritzsen GF. Up-front autologous stem-cell transplantation in peripheral T-cell lymphoma: NLG-T-01. J Clin Oncol. 2012; 30(25):3093-3099. Google Scholar Mercadal S, Briones J, Xicoy B. Intensive chemotherapy (high-dose CHOP/ESHAP regimen) followed by autologous stem-cell transplantation in previously untreated patients with peripheral T-cell lymphoma. Ann Oncol. 2008; 19(5):958-963. Google Scholar Reimer P, Rudiger T, Geissinger E. Autologous stem-cell transplantation as first-line therapy in peripheral T-cell lymphomas: results of a prospective multicenter study. J Clin Oncol. 2009; 27(1):106-113. Google Scholar Rodriguez J, Conde E, Gutierrez A. Frontline autologous stem cell transplantation in high-risk peripheral T-cell lymphoma: a prospective study from the Gel-Tamo study group. Eur J Haematol. 2007; 79(1):32-38. Google Scholar Wilhelm M, Smetak M, Reimer P. First-line therapy of peripheral T-cell lymphoma: extension and long-term follow-up of a study investigating the role of autologous stem cell transplantation. Blood Cancer J. 2016; 6(7):e452. Google Scholar García-Sancho AM, Bellei M, López-Parra M. Autologous stem cell transplantation as consolidation of first-line chemotherapy in patients with peripheral T-cell lymphoma: a multicenter GELTAMO/FIL study. Haematologica. 2022; 107(11):2675-2684. Google Scholar Bachy E, Camus V, Thieblemont C. Romidepsin plus CHOP versus CHOP in patients with previously untreated peripheral T-cell lymphoma: results of the Ro-CHOP phase III study (conducted by LYSA). J Clin Oncol. 2022; 40(3):242-251. Google Scholar Simon A, Peoch M, Casassus P. Upfront VIP-reinforced-ABVD (VIP-rABVD) is not superior to CHOP/21 in newly diagnosed peripheral T cell lymphoma. Results of the randomized phase III trial GOELAMS-LTP95. Br J Haematol. 2010; 151(2):159-166. Google Scholar Ellin F, Landstrom J, Jerkeman M, Relander T.. Real-world data on prognostic factors and treatment in peripheral T-cell lymphomas: a study from the Swedish Lymphoma Registry. Blood. 2014; 124(10):1570-1577. Google Scholar Cederleuf H, Hjort Jakobsen L, Ellin F. Outcome of peripheral T-cell lymphoma in first complete remission: a Danish-Swedish population-based study. Leuk Lymphoma. 2017; 58(12):2815-2823. Google Scholar Fossard G, Broussais F, Coelho I. Role of up-front autologous stem-cell transplantation in peripheral T-cell lymphoma for patients in response after induction: an analysis of patients from LYSA centers. Ann Oncol. 2018; 29(3):715-723. Google Scholar Park SI, Horwitz SM, Foss FM. The role of autologous stem cell transplantation in patients with nodal peripheral T-cell lymphomas in first complete remission: report from COMPLETE, a prospective, multicenter cohort study. Cancer. 2019; 125(9):1507-1517. Google Scholar Savage KJ, Horwitz SM, Advani RH. An exploratory analysis of brentuximab vedotin plus CHP (A+CHP) in the frontline treatment of patients with CD30+ peripheral T-cell lymphomas (ECHELON-2): impact of consolidative stem cell transplant. Blood. 2019; 134(Suppl_1):464. Google Scholar
|

|
Scooped by
Gilbert C FAURE
March 2, 2023 8:24 AM
|
Feline leukemia virus (FeLV) and feline immunodeficiency virus (FIV) are retroviruses affecting cats worldwide, and the prevalence of infection varies considerably according to the geographic area.We retrospectively described FIV- and FeLV-associated diseases in a population of 1470 necropsied cats...

|
Scooped by
Gilbert C FAURE
February 28, 2023 4:49 AM
|
Our study demonstrated the distinct peripheral immune cell profile of DHL patients by contrast to DLBCL patients and healthy people, as well as the heterogeneity within the DHL group, which could provide valuable guidance for the diagnosis and treatment of DHL.

|
Scooped by
Gilbert C FAURE
February 18, 2023 6:52 AM
|
A Member Board of the American Board of Medical Specialties

|
Scooped by
Gilbert C FAURE
February 17, 2023 3:41 AM
|
The ability to eliminate specific pathologic cells is creating more options for multiple myeloma patients. Several targeting approaches are in development.

|
Scooped by
Gilbert C FAURE
February 11, 2023 3:00 AM
|
While the genetic landscape of chronic lymphocytic leukemia (CLL) has been broadly profiled by large-scale sequencing studies performed over the past decade, the molecular basis of the transformation of CLL to aggressive lymphoma, or Richter syndrome (RS), has remained incompletely characterized.

|
Scooped by
Gilbert C FAURE
January 10, 2023 10:47 AM
|
Correspondances en Onco-Hématologie / N° 6 décembre 2022 Correspondances en Onco-Hématologie N° 6 | décembre 2022 Acheter ce numéro → S'abonner → Déjà abonné ? Connectez-vous 1 / 2 >> suivant Tous les articles de cette édition Editorial Immunothérapie en hématologie, une vieille idée toute neuve...

|
Rescooped by
Gilbert C FAURE
from Virus World
December 20, 2022 1:57 PM
|
Babies develop risk of cancer in womb, but will not go on to develop it without second ‘hit’ from an infection, scientists believe. Childhood leukaemia is driven by common childhood infections meeting pre-cancerous cells in the blood, scientists believe. Experts at the Institute of Cancer Research (ICR) in London have found babies develop the risk for leukaemia in the womb, but will not go on to develop the disease without a second ‘hit’ from a viral or bacterial infection, such as flu. The research highlights the importance of allowing infants to socialise with other children early in their lives, to prime their immune systems against infections. The discovery came by studying pairs of twins, where only one initially developed acute lymphoblastic leukaemia (ALL) - the most common type of the cancer in children. Identical twins are around 15-25 per cent more likely to go on to develop ALL if their sibling already has the disease, while less than one per cent of non-identical twins or other siblings go on to develop the disease. Researchers followed the twins for up to 15 years and found the high risk only applies if the identical twins shared a single placenta before birth - which only happens in around 60 per cent of identical twin pairs. Findings 'confirm disease can be traced back to womb' It confirms that the conditions needed to trigger leukaemia first arise in the womb, and even the healthy twin will carry "pre-leukaemia" cells in the blood, which have occurred through a spontaneous developmental error, and passed between the two. But clinically silent cells will not develop into cancer without a post-birth "hit", probably from common childhood infections. Prof Sir Mel Greaves, the founding director of the Centre for Evolution and Cancer and Professor of Cell Biology at The Institute of Cancer Research, London, said: “Our study provides new insights into the origins of childhood acute lymphoblastic leukaemia. “These new findings confirm that the disease can be traced back to the womb when pre-leukaemia cells spread via the twins’ shared blood supply. "What remained a mystery until now was why sometimes only one twin is diagnosed with leukaemia. “We still do not know for certain what leads to the first ‘hit’ of genetic changes in the womb, but we think that the second ‘hit’ of genetic changes is probably triggered by common childhood infections – opening up the possibility of ‘priming’ the immune system in infancy to avoid the development of the disease later on in life.” Acute lymphoblastic leukaemia is the most common type of childhood cancer, accounting for 80 per cent of leukaemia cases in children. The team are now focussed on finding the the second infection-driven hit after birth. Boosting the gut could protect children against disease They believe that the gut microbiome may be playing a key role in protecting children against developing leukaemia even if they have pre-cancerous cells. Although vaccines have little impact on preventing ALL, boosting the gut in early life may help. Prof Greaves added: “Risk of ALL is elevated by C-section birth, lack of breast feeding and paucity of social contacts in infancy. “Conversely, attendance at play groups in infancy is protective. So, to some extent , risk can be modified without medical intervention.” The findings will also allow doctors to assess the risk of ALL for twins, firstly by determining whether the twins are identical and sharing a placenta and then by regularly tracking the levels of pre-leukaemia cells in their blood. Sarah McDonald, the deputy director of research at Blood Cancer UK, who funded the work said: “Understanding the mechanism as to how the cancer develops in identical twins, and why often only one develops leukaemia is an important question to answer. “It helps us understand both the risk of the other sibling developing leukaemia and provides insight into how leukaemia develops in all children. “This research shows that in cases where one twin develops leukaemia, and both twins both share a placenta during pregnancy, two events are needed to determine whether the other sibling develops the disease - one before birth and the other after.” Research published (Dec. 20, 2022) in the journal Leukaemia: https://doi.org/10.1038/s41375-022-01756-1
Via Juan Lama

|
Scooped by
Gilbert C FAURE
December 20, 2022 8:31 AM
|
Next Generation Flow (NGF) represents a gold standard for the evaluation of Minimal Residual Disease (MRD) in Multiple Myeloma (MM) patients at any stage of treatment.Although the assessment of MRD is still not universally employed in clinical practice, numerous studies have demonstrated the streng...

|
Scooped by
Gilbert C FAURE
December 20, 2022 8:21 AM
|
Burkitt lymphoma (BL) is an aggressive form of B cell lymphoma that can affect children and adults. The study of BL led to the identification of the first recurrent chromosomal aberration in lymphoma, t(8;14)(q24;q32), and subsequent discovery of the central role of MYC and Epstein–Barr virus (EBV) in tumorigenesis. Most patients with BL are cured with chemotherapy but those with relapsed or refractory disease usually die of lymphoma. Historically, endemic BL, non-endemic sporadic BL and the immunodeficiency-associated BL have been recognized, but differentiation of these epidemiological variants is confounded by the frequency of EBV positivity. Subtyping into EBV+ and EBV− BL might better describe the biological heterogeneity of the disease. Phenotypically resembling germinal centre B cells, all types of BL are characterized by dysregulation of MYC due to enhancer activation via juxtaposition with one of the three immunoglobulin loci. Additional molecular changes commonly affect B cell receptor and sphingosine-1-phosphate signalling, proliferation, survival and SWI–SNF chromatin remodelling. BL is diagnosed on the basis of morphology and high expression of MYC. BL can be effectively treated in children and adolescents with short durations of high dose-intensity multiagent chemotherapy regimens. Adults are more susceptible to toxic effects but are effectively treated with chemotherapy, including modified versions of paediatric regimens. The outcomes in patients with BL are good in high-income countries with low mortality and few late effects, but in low-income and middle-income countries, BL is diagnosed late and is usually treated with less-effective regimens affecting the overall good outcomes in patients with this lymphoma. Burkitt lymphoma is an aggressive form of B cell lymphoma that can affect children and adults. Its research has been central in advancing haematopathology and cancer research. This Primer by Siebert and colleagues summarizes the epidemiology, mechanisms, diagnosis and treatment of this disorder, patient quality of life and open research questions.

|
Scooped by
Gilbert C FAURE
December 11, 2022 12:44 PM
|
Symposia: Emerging Tools, Techniques and Artificial Intelligence in Hematology Program: Oral and Poster Abstracts Type: Oral Hematology Disease Topics & Pathways:Research, Lymphoid Leukemias, ALL, Acute Myeloid Malignancies, AML, artificial intelligence (AI), Translational Research, Lymphomas, bioinformatics, Diseases, aggressive lymphoma, Lymphoid Malignancies, computational biology, Myeloid Malignancies, emerging technologies, Technology and Procedures, profiling, imaging, machine learning, molecular testing Monday, December 12, 2022: 10:30 AM-12:00 PM 391-392 (Ernest N. Morial Convention Center) Moderators: Roni Shouval, MD, PhD, Memorial Sloan Kettering Cancer Center and Taiga Nishihori, MD, Moffitt Cancer Center Disclosures: Shouval: MyBiotics: Consultancy; Medexus: Consultancy, Ended employment in the past 24 months. 10:30 AM 787 Machine Learning Algorithm Correctly Identifies 95% of Cells in Differential Count of Blood Smears: A Prospective Study on >29,000 Cases and >17 Million Single Cells Torsten Haferlach, MD, Niroshan Nadarajah*, Claudia Haferlach, MD, Wolfgang Kern, MD and Christian Pohlkamp, MD*MLL Munich Leukemia Laboratory, Munich, Germany 10:45 AM 788 Prediction of Initial Risk Group, B/T Subtype, and ETV6-RUNX1 Translocation in Pediatric Acute Lymphoblastic Leukemia By Deep Convolutional Neural Network Analysis of Giemsa-Stained Whole Slide Images Gil Shamai1*, Ran Schley1*, Yoav Binenbaum2*, Ron Kimmel1* and Ronit Elhasid, MD31Technion - Israel Institute of Technology, Haifa, Israel2Dana farber cancer institute, Boston3Pediatric Hemato-Oncology Department, Sourasky Medical Center, Tel Aviv, Israel 11:00 AM 789 Regularized Mixture Cure Models Identify a Gene Signature That Improves Risk Stratification within the Favorable-Risk Group in 2017 European Leukemianet (ELN) Classification of Acute Myeloid Leukemia (Alliance 152010) Kellie J. Archer, PhD1*, Han Fu, PhD1*, Krzysztof Mrózek, MD, PhD2, Deedra Nicolet, MS3,4, Jessica Kohlschmidt, PhD4,5, Alice S. Mims, MD6,7, Geoffrey L. Uy, MD8, Wendy Stock, MD9,10, John C. Byrd, MD11 and Ann-Kathrin Eisfeld, MD31Division of Biostatistics, College of Public Health, The Ohio State University, Columbus, OH2Clara D. Bloomfield Center for Leukemia Outcomes Research, The Ohio State University Comprehensive Cancer Center, Columbus, OH3Clara D. Bloomfield Center for Leukemia Outcomes Research, The Ohio State University, Columbus, OH4Alliance Statistics and Data Management Center, The Ohio State University Comprehensive Cancer Center, Columbus, OH5Clara D. Bloomfield Center for Leukemia Outcomes Research, The Ohio State University Comprehensive Cancer Center, Columbus6Division of Hematology, Department of Internal Medicine, The Ohio State University, Columbus, OH7Arthur G. James Cancer Center Hospital, The Ohio State University Comprehensive Cancer Center, Columbus, OH8Division of Oncology, Department of Medicine, Washington University School of Medicine, Saint Louis, MO9University of Chicago, Chicago, IL10Department of Medicine, Section of Hematology/Oncology, University of Chicago, Chicago, IL11University of Cincinnati College of Medicine, Cincinnati, OH 11:15 AM 790 Evaluation of a Transparent Artificial Intelligence (AI) Disease Classification System with Whole Genome Sequencing (WGS) and Whole Transcriptome Sequencing (WTS) Data in a Prospective Study with 325 Cases Niroshan Nadarajah*, Sven Maschek*, Stephan Hutter, PhD*, Manja Meggendorfer, PhD, Wolfgang Kern, MD, Claudia Haferlach, MD and Torsten Haferlach, MDMLL Munich Leukemia Laboratory, Munich, Germany 11:30 AM 791 Multi-Omics Data Integration of Genomics and RNA-Splicing in Myeloid Neoplasia Arda Durmaz1*, Carmelo Gurnari, MD1, Courtney E. Hershberger, PhD2, Simona Pagliuca, MD, PhD3, Hassan Awada, MD4*, Hussein Awada1*, Minako Mori, MD, PhD5*, Yasuo Kubota, MD, PhD1*, Tariq Kewan, MD1*, Waled Bahaj, MD1, John Barnard, PhD2*, Jacob Scott, MD1*, Richard A Padgett, PhD6*, Torsten Haferlach, MD7, Jaroslaw P. Maciejewski, MD, PhD, FACP1 and Valeria Visconte, PhD11Department of Translational Hematology and Oncology Research, Taussig Cancer Institute, Cleveland Clinic Foundation, Cleveland, OH2Department of Quantitative Health Sciences, Cleveland Clinic, Cleveland, OH3Service d'Hématologie Clinique, Hôpital Brabois, CHRU de Nancy, Nancy, France4Roswell Park Comprehensive Cancer Center, Buffalo, NY5Department of Hematology and Oncology, Kyoto University, Kyoto, Japan6Department of Cardiovascular and Metabolic Sciences, Cleveland Clinic Foundation, Cleveland, OH7MLL Munich Leukemia Laboratory, Munich, Germany 11:45 AM 792 Prediction of Poor Outcome after Tisagenlecleucel in Patients with Relapsed or Refractory Diffuse Large B Cell Lymphoma (DLBCL) Using Artificial Intelligence Analysis of Pre-Infusion PET/CT Margot Jak1*, Bas H.M. van der Velden, PhD2*, Bart de Keizer, MD PhD3*, Sjoerd G Elias, MD PhD4*, Monique C. Minnema, MD, PhD5 and Kenneth G.A. Gilhuijs, PhD2*1Department of Hematology, University Medical Center Utrecht, Utrecht, Netherlands2Image Sciences Institute, The University Medical Center Utrecht, Utrecht, Netherlands3Radiology, The University Medical Center Utrecht, Utrecht, Netherlands4Julius Center for Health Sciences and Primary Care, The University Medical Center Utrecht, Utrecht, Netherlands5Department of Hematology, University Medical Center, Utrecht, Netherlands, Utrecht, Netherlands See more of: Oral and Poster Abstracts << Previous Session | Next Session >> *signifies non-member of ASH

|
Scooped by
Gilbert C FAURE
November 30, 2022 10:49 AM
|
Somatic HLA loss in patients with AA. We used a combination of targeted NGS of HLA class I genes and single nucleotide polymorphism array (SNP-A) genotyping in 505 AA patients from 2 multi-institutional consortia, NAPAAC (n = 156 patients) and CIBMTR-discovery (n = 349 patients) (Figure 1 and...

|
Scooped by
Gilbert C FAURE
November 15, 2022 4:56 AM
|
Waldenström macroglobulinemia (WM) is a rare, incurable, low-grade, B cell lymphoma. Symptomatic disease commonly results from marrow or organ infiltration and hyperviscosity secondary to immunoglobulin M paraprotein, manifesting as anemia, bleeding ...
|

 Your new post is loading...
Your new post is loading...
 Your new post is loading...
Your new post is loading...