 Your new post is loading...

|
Scooped by
Dr. Stefan Gruenwald
September 16, 2023 7:54 PM
|
A new type of vaccine developed by researchers at the University of Chicago’s Pritzker School of Molecular Engineering (PME) has shown in the lab setting that it can completely reverse autoimmune diseases like multiple sclerosis and type 1 diabetes— all without shutting down the rest of the immune system. A typical vaccine teaches the human immune system to recognize a virus or bacteria as an enemy that should be attacked. The new “inverse vaccine” does just the opposite: it removes the immune system’s memory of one molecule. While such immune memory erasure would be unwanted for infectious diseases, it can stop autoimmune reactions like those seen in multiple sclerosis, type I diabetes, or rheumatoid arthritis, in which the immune system attacks a person’s healthy tissues. The inverse vaccine, described in Nature Biomedical Engineering, takes advantage of how the liver naturally marks molecules from broken-down cells with “do not attack” flags to prevent autoimmune reactions to cells that die by natural processes. PME researchers coupled an antigen — a molecule being attacked by the immune system— with a molecule resembling a fragment of an aged cell that the liver would recognize as friend, rather than foe. The team showed how the vaccine could successfully stop the autoimmune reaction associated with a multiple-sclerosis-like disease. “In the past, we showed that we could use this approach to prevent autoimmunity,” said Jeffrey Hubbell, the Eugene Bell Professor in Tissue Engineering and lead author of the new paper. “But what is so exciting about this work is that we have shown that we can treat diseases like multiple sclerosis after there is already ongoing inflammation, which is more useful in a real-world context.”

|
Scooped by
Dr. Stefan Gruenwald
August 16, 2023 5:29 PM
|
A stroke survivor is learning to walk independently again thanks to high-tech trousers powered by AI. Julie Lloyd, 65, is part of the UK's first trial of the "smart garment" that she described as a breakthrough for fellow stroke patients.
The "NeuroSkin" trousers stimulate her paralysed leg using electrodes controlled by artificial intelligence. The Stroke Association said new technologies are giving hope to the UK's 1.3 million stroke survivors.
The developers of NeuroSkin said the invention is already revolutionizing stroke care in France, but Ms Lloyd is one of the first involved in the UK's own trial. "My leg is almost feeling as if it's being guided," she said. After first experiencing an uncomfortable "tingling feeling", she said that within a few minutes she was walking unaided for the first time in six months.
"My leg was suddenly propelled up from the floor and made me feel safe walking, and that's the part that I've honestly not felt at all with all the physiotherapy I've had," she said. Julie needs a cane to walk her dogs near her home. "I've never felt since my stroke as elated as I feel this moment."
The businesswoman, from Penarth in Vale of Glamorgan, had a minor stroke in January which resulted in her left arm and left leg being partially paralyzed. She currently relies on a cane to walk. "Life before was very energetic, very active," she said, explaining how she ran the Cardiff half marathon before her stroke. "It has taken away a lifestyle I had and that's been terribly tragic."
Her rehabilitation involves hours of repetitive exercises aimed at "teaching" her brain to work around the area damaged by the stroke and make new connections to control her left side. The progress has been slow and grueling. But when she got to about 3,000 steps a day with a cane, her physiotherapist recommended she enter the trial of the AI-powered tech.

|
Scooped by
Dr. Stefan Gruenwald
August 14, 2023 4:28 PM
|
The OncoNPC machine-learning model may help identify sites of origin for cancers whose origins are unknown, which could enable targeted tumor treatments. For a small percentage of cancer patients, doctors are unable to determine where their cancer originated. This makes it much more difficult to choose a treatment for those patients, because many cancer drugs are typically developed for specific cancer types. A new approach developed by researchers at MIT and Dana-Farber Cancer Institute may make it easier to identify the sites of origin for those enigmatic cancers. Using machine learning, the researchers created a computational model that can analyze the sequence of about 400 genes and use that information to predict where a given tumor originated in the body. Using this model, the researchers showed that they could accurately classify at least 40 percent of tumors of unknown origin with high confidence, in a dataset of about 900 patients. This approach enabled a 2.2-fold increase in the number of patients who could have been eligible for a genomically guided, targeted treatment, based on where their cancer originated. “That was the most important finding in our paper, that this model could be potentially used to aid treatment decisions, guiding doctors toward personalized treatments for patients with cancers of unknown primary origin,” says Intae Moon, an MIT graduate student in electrical engineering and computer science who is the lead author of the new study. Alexander Gusev, an associate professor of medicine at Harvard Medical School and Dana-Farber Cancer Institute, is the senior author of the paper, which appears today in Nature Medicine.

|
Scooped by
Dr. Stefan Gruenwald
July 13, 2023 5:14 PM
|
For the 4th time, Anumana has earned the FDA’s breakthrough device designation for another of its artificial intelligence algorithms designed to catch hard-to-spot heart conditions in their earliest stages. This time around, the FDA bestowed the label upon Anumana’s algorithm targeting cardiac amyloidosis, a condition in which abnormal tissues build up in and around the heart, making it more difficult for the heart to pump normally and potentially leading to heart failure or even death. Cardiac amyloidosis can be difficult to properly diagnose because many of its most prevalent symptoms—which include kidney inflammation, liver swelling, heart palpitations and fatigue—may be attributed to other, more common conditions. But Anumana claims to have cracked the code: Its AI tool is designed to pick up on signs of the disease that may otherwise go overlooked by doctors. Anumana developed the algorithm with support from Pfizer. The duo announced their partnership late last year, pledging to spend the next several years working together to bring the cardiac amyloidosis-spotting tech into clinics in the U.S., Europe and Japan. Like Anumana’s other AI tools, it works by scouring standard ECGs, digging into the peaks and valleys of the heart’s electrical signals to detect early indicators of the protein build-up that are typically invisible to human doctors. With the new breakthrough device label, which Anumana recently announced, the process will be somewhat fast-tracked, as the designation opens up a dedicated channel of communication between the startup and the regulator and also ensures that future applications are shuttled to the top of the agency’s pile for review. “The use of AI algorithms to identify subtle signals in ECGs that are imperceptible to humans stands to transform cardiovascular medicine by allowing us to detect disease early, in more easily treated stages, potentially avoiding serious consequences,” Paul Friedman, M.D., chair of Anumana’s Mayo Clinic board of advisors, said in the announcement. “It is encouraging to see an algorithm that can aid in the identification of cardiac amyloidosis receive breakthrough device designation, as it recognizes the importance of having new tools to detect rare cardiac diseases in ways never before possible.”

|
Scooped by
Dr. Stefan Gruenwald
July 10, 2023 3:09 PM
|
IgG3 antibodies are the most potent subclass of IgG, yet are the only subclass not used as therapeutics. That stems partly from the fact that nobody knew what they looked like and what complexes they formed and when. Now, Leoni Abendstein solved the structure of antigen-bound IgG3, both alone and activating complement, and discovered loads about how these unique antibodies work IgG3 is unique among the IgG subclasses due to its extended hinge, allotypic diversity and enhanced effector functions, including highly efficient pathogen neutralisation and complement activation. It is also underrepresented as an immunotherapeutic candidate, partly due to a lack of structural information. Here, we use cryoEM to solve structures of antigen-bound IgG3 alone and in complex with complement components. These structures reveal a propensity for IgG3-Fab clustering, which is possible due to the IgG3-specific flexible upper hinge region and may maximise pathogen neutralisation by forming high-density antibody arrays. IgG3 forms elevated hexameric Fc platforms that extend above the protein corona to maximise binding to receptors and the complement C1 complex, which here adopts a unique protease conformation that may precede C1 activation. Mass spectrometry reveals that C1 deposits C4b directly onto specific IgG3 residues proximal to the Fab domains. Structural analysis shows this to be caused by the height of the C1-IgG3 complex. Together, these data provide structural insights into the role of the unique IgG3 extended hinge, which will aid the development and design of upcoming immunotherapeutics based on IgG3.

|
Rescooped by
Dr. Stefan Gruenwald
from Virus World
July 7, 2023 1:18 PM
|
Human endogenous retroviruses (HERVs) are ancestral viral relics that constitute nearly 8% of the human genome. Although normally silenced, the most recently integrated provirus HERV-K (HML-2) can be reactivated in certain cancers. Here, we report pathological expression of HML-2 in malignant gliomas in both cerebrospinal fluid and tumor tissue that was associated with a cancer stem cell phenotype and poor outcomes. Using single-cell RNA-Seq, we identified glioblastoma cellular populations with elevated HML-2 transcripts in neural progenitor–like cells (NPC-like) that drive cellular plasticity. Using CRISPR interference, we demonstrate that HML-2 critically maintained glioblastoma stemness and tumorigenesis in both glioblastoma neurospheres and intracranial orthotopic murine models. Additionally, we demonstrate that HML-2 critically regulated embryonic stem cell programs in NPC-derived astroglia and altered their 3D cellular morphology by activating the nuclear transcription factor OCT4, which binds to an HML-2–specific long-terminal repeat (LTR5Hs). Moreover, we discovered that some glioblastoma cells formed immature retroviral virions, and inhibiting HML-2 expression with antiretroviral drugs reduced reverse transcriptase activity in the extracellular compartment, tumor viability, and pluripotency. Our results suggest that HML-2 fundamentally contributes to the glioblastoma stem cell niche. Because persistence of glioblastoma stem cells is considered responsible for treatment resistance and recurrence, HML-2 may serve as a unique therapeutic target. Published in JCI (July 3, 2023): https://doi.org/10.1172/JCI167929
Via Juan Lama

|
Rescooped by
Dr. Stefan Gruenwald
from Virus World
June 18, 2023 11:52 AM
|
Researchers recently discovered an unexpected link between the progression of ALS and PEG10, a protein traditionally known for its role in placental development. Overabundance of this protein in nerve tissue has been observed to alter cell behavior contributing to ALS. The team is now studying the molecular pathways involved with a view to inhibiting this rogue protein, potentially paving the way for new therapeutics. Key facts: - The ancient, virus-like protein PEG10, typically associated with placental development, has been linked to the progression of ALS when present in high levels in nerve tissue.
- The research marks the first time PEG10 has been connected to ALS, with its accumulation seen as a hallmark of the disease.
- Research findings suggest that inhibiting the rogue PEG10 protein could potentially lead to a new class of therapeutics for ALS.
More than 5,000 people are diagnosed annually with ALS (amyotrophic lateral sclerosis), a fatal, neurodegenerative disease that attacks nerve cells in the brain and spinal cord, gradually robbing people of the ability to speak, move, eat and breathe. To date, only a handful of drugs exist to moderately slow its progression. There is no cure. But CU Boulder researchers have identified a surprising new player in the disease—an ancient, virus-like protein that is best known, paradoxically, for its essential role in enabling placental development. The findings were recently published in the journal eLife. “Our work suggests that when this strange protein known as PEG10 is present at high levels in nerve tissue, it changes cell behavior in ways that contribute to ALS,” said senior author Alexandra Whiteley, assistant professor in the Department of Biochemistry. Whiteley’s lab is now working to understand the molecular pathways involved and to find a way of inhibiting the rogue protein. “It is early days still, but the hope is this could potentially lead to an entirely new class of potential therapeutics to get at the root cause of this disease.” Ancient viruses with modern-day impact Mounting research suggests about half the human genome is made up of bits of DNA left behind by viruses (known as retroviruses) and similar virus-like parasites, known as transposons, which infected our primate ancestors 30-50 million years ago. Some, like HIV, are well known for their ability to infect new cells and cause disease. Others, like wolves who have lost their fangs, have become domesticated over time, losing their ability to replicate while continuing to pass from generation to generation, shaping human evolution and health. PEG10, or Paternally Expressed Gene 10, is one such “domesticated retrotransposon.” Studies show it likely played a key role in enabling mammals to develop placentas—a critical step in human evolution. But like a viral Jekyll and Hyde, when it’s overly abundant in the wrong places, it may also fuel disease, including certain cancers and another rare neurological disorder called Angelman’s syndrome, studies suggest. Whiteley’s research is the first to link the virus-like protein to ALS, showing that PEG10 is present in high levels in the spinal cord tissue of ALS patients where it likely interferes with the machinery enabling brain and nerve cells to communicate. “It appears that PEG10 accumulation is a hallmark of ALS,” said Whiteley, who has already secured a patent for PEG10 as a biomarker, or way of diagnosing, the disease. Too much protein in the wrong places Whiteley did not set out to study ALS, or ancient viruses. Instead, she studies how cells get rid of extra protein, as too much of the typically good thing has been implicated in other neurodegenerative diseases, including Alzheimer’s and Parkinson’s. Her lab is one of a half-dozen in the world to study a class of genes called ubiquilins, which serve to keep problem proteins from accumulating in cells. In 2011, a study linked a mutation in the ubiquilin-2 gene (UBQLN2) to some cases of familial ALS, which makes up about 10% of ALS cases. The other 90% are sporadic, meaning they are not believed to be inherited. But it has remained unclear how the faulty gene might fuel the deadly disease. Using laboratory techniques and animal models, Whiteley and colleagues at Harvard Medical School first set out to determine which proteins pile up when the UBQLN2 misfires and fails to put the brakes on. Among thousands of possible proteins, PEG10 topped the list. Then Whiteley and her colleagues collected the spinal tissue of deceased ALS patients (provided by the medical research foundation Target ALS) and used protein analysis, or proteomics, to see which if any seemed overexpressed. Again, among more than 7,000 possible proteins, PEG10 was in the top five. In a separate experiment, the team found that with the ubiquilin brakes essentially broken, the PEG10 protein piles up and disrupts the development of axons—the cords which carry electrical signals from the brain to the body. PEG10 was over-expressed in the tissue of individuals with both sporadic and familial ALS, the study found, meaning the virus-like protein may be playing a key role in both. “The fact that PEG10 is likely contributing to this disease means we may have a new target for treating ALS,” she said. “For a terrible disease in which there are no effective therapeutics that lengthen lifespan more than a couple of months, that could be huge.” The research could also lead to a better understanding of other diseases that result from protein accumulation, as well as keener insight into how ancient viruses influence health. In this case, Whiteley said, the so-called “domesticated” virus could a be rearing its fangs again. “Domesticated is a relative term, as these virus-like activities may be a driver of neurodegenerative disease,” she said. “And in this case, what is good for the placenta may be bad for neural tissue.” Original research published in eLIfe(May 23, 2023): https://doi.org/10.7554/eLife.79452
Via Juan Lama

|
Rescooped by
Dr. Stefan Gruenwald
from Virus World
May 16, 2023 2:13 PM
|
A clinical trial of an experimental universal influenza vaccine developed by researchers at the National Institute of Allergy and Infectious Diseases’ (NIAID) Vaccine Research Center (VRC), part of the National Institutes of Health, has begun enrolling volunteers at Duke University in Durham, North Carolina. This Phase 1 trial will test the experimental vaccine, known as H1ssF-3928 mRNA-LNP, for safety and its ability to induce an immune response. The trial will enroll up to 50 healthy volunteers aged 18 through 49. Three groups of study participants (10 participants each) will be vaccinated with 10, 25 and 50 micrograms of the experimental vaccine, respectively. After evaluation of the data to determine an optimum dosage, an additional 10 participants will be enrolled to receive the optimum dosage. The study also will include a group of participants who will receive a current quadrivalent seasonal influenza vaccine. This will allow the researchers a point of direct comparison between the immunogenicity and safety of the candidate vaccine and available seasonal flu vaccines. Participants will be regularly evaluated to assess the vaccine’s safety (and, secondarily, its efficacy) and will receive follow-up appointments for up to one year after vaccination. Seasonal influenza, or flu, kills thousands of people in the United States each year. The Centers for Disease Control and Prevention estimates that between 2010 and 2020, between 12,000 and 52,000 people died of flu in the United States annually. Although annual seasonal flu vaccines are valuable tools in controlling the spread and severity of influenza, they do not provide immunity against every flu strain. Each year, before the flu season begins, scientific experts must predict which influenza strains are likely to be most common during the upcoming months and then select three or four of these strains to include in the next seasonal flu vaccine. Vaccine manufacturers then need time to produce and distribute the vaccine—during which the dominant strains of the virus can change in unexpected ways, potentially decreasing the efficacy of the vaccine. An effective universal flu vaccine could eliminate these problems by protecting its recipients against a wide variety of strains and ideally providing durable long-term immunity, so people would not need to be vaccinated every year. “A universal influenza vaccine would be a major public health achievement and could eliminate the need for both annual development of seasonal influenza vaccines, as well as the need for patients to get a flu shot each year,” said Acting NIAID Director Hugh Auchincloss, M.D. “Moreover, some strains of influenza virus have significant pandemic potential. A universal flu vaccine could serve as an important line of defense against the spread of a future flu pandemic.”
Via Juan Lama

|
Scooped by
Dr. Stefan Gruenwald
May 14, 2023 2:02 PM
|
Goldenhar syndrome is a rare congenital disease, affecting early foetal development. This syndrome includes malformations of varying severity, affecting different parts of the face. Its causes and modes of transmission are still poorly understood. An international collaboration led by the University of Geneva (UNIGE) and Beihang University, in China, has discovered that pathogenic variants of the FOXI3 gene -- responsible for the development of the ear -- cause one form of this developmental disorder. The scientists were also able to identify the modes of transmission of the disease when this particular gene is involved. These results can be found in Nature Communications. First described in 1952 at the University of Geneva (UNIGE) by the ophthalmologist Maurice Goldenhar (1924-2001), Goldenhar syndrome or oculo-auriculo-vertebral dysplasia is a rare congenital disorder with facial asymmetry, malformations of the auditory and ocular systems, and spinal column anomalies, sometimes associated with mental retardation. It varies greatly in expression and severity depending on the individual and is thought to affect approximately one in 3,500 to 5,600 births. This syndrome is caused by mutations that appear during foetal development. Do these mutations occur spontaneously or is there an inherited form of the disease? The mode of transmission and the molecular mechanisms involved are still poorly understood. A recent international research effort, led by a joint team from the UNIGE and Beihang University, in China, is providing new elements for understanding the disease. The scientists have identified a gene -- FOXI3 -- whose pathogenic variants are involved in one form of the disease. A gene that builds the ear ''FOXI3 is critically involved in the development of the ear. It is an architect gene: it produces a protein that acts as a transcription factor. In other words, it controls the expression of other genes to start the construction of the ear,'' explains Stylianos Antonarakis, Emeritus Professor at the UNIGE Faculty of Medicine and co-last author of the study, which was funded by the ChildCare Foundation in Geneva. To determine the role of FOXI3, the scientists used the genetic profiles of a highly consanguineous Pakistani family involved in a larger research project on rare diseases. In this family, two brothers have the syndrome but not their parents, nor the other four siblings. Both patients have pathogenic mutations in both copies of the FOXI3 gene, while several other members of this family, who do not have Goldenhar syndrome, have a mutation in only one copy of the gene. 'This first analysis allowed us to confirm that pathogenic mutations in both copies of the FOXI3 gene, each inherited from a parent, are necessary for a form of the disease to develop,'' explains Stylianos Antonarakis. This is called autosomal recessive inheritance. Responsible for one form of the disease To find out whether pathogenic variants in FOXI3 and their autosomal recessive inheritance are systematically involved, the researchers also analyzed the genetic profiles of 670 unrelated patients in Europe and China. ''Eighteen pathogenic variants in the FOXI3 gene were identified in twenty-one patients. FOXI3 is therefore only one of the genes that can cause the disease, precisely the specific form observed in our Pakistani patients,'' explains Ke Mao, researcher at Beihang University and co-first author of the study with Christelle Borel, senior research associate in the Department of Genetic Medicine and Development at the UNIGE Faculty of Medicine, and Muhammad Ansar, senior research and teaching assistant in the same department and head of the ''Eye Genetics'' research group at the Jules-Gonin eye hospital. Another finding that baffled the researchers was that the mode of inheritance, in many other cases, is seemingly autosomal dominant. In this case, only one of the two copies of the gene needs to carry the mutation for the disease to occur. By carefully studying the genome around the FOXI3 gene, scientists have solved this mystery. There is a functional and frequent, but specific, variant of one copy of the FOXI3 gene that, in combination with a severe mutation in the other copy of the FOXI3 gene, also causes the disorder. 'We also created mouse models with mutations in the FOXI3 gene to validate the results obtained in human families. These mice showed facial deformities equivalent to human characteristics,'' adds Prof. Yong-Biao Zhang of Beihang University, the study's co-lead author. These results provide crucial elements for understanding this syndrome and would explain in part the extensive heterogeneity of the disease. They also pave the way for the study of other genes that may be involved in the disease, but also in the healthy development of certain parts of the face, the mechanisms of which are still poorly understood.

|
Scooped by
Dr. Stefan Gruenwald
April 4, 2023 12:33 PM
|
A new robotic arm could be a game changer in surgeries, with the capability to 3D-print biomaterial directly onto organs inside a patient’s body. The arm, designed by a team of engineers from the University of New South Wales in Australia, can place a tiny, flexible 3D bioprinter inside the body, using bio-ink to “print” tissue-like structures onto internal organs. The device, dubbed F3DB, is designed with a swivel head to offer full flexibility of movement and features an array of soft artificial muscles to allow movement in three separate directions. The entire structure can be controlled externally. “Existing 3D-bioprinting techniques require biomaterials to be made outside the body and implanting that into a person would usually require large open-field open surgery which increases infection risks,” said Thanh Do, study lead. “Our flexible 3D bioprinter means biomaterials can be directly delivered into the target tissue or organs with a minimally invasive approach. This system offers the potential for the precise reconstruction of three-dimensional wounds inside the body. Our approach also addresses significant limitations in existing 3D bioprinters such as surface mismatches between 3D-printed biomaterials and target tissues/organs as well as structural damage during manual handling, transferring, and transportation process.” Bioprinting in the medical industry is primarily used for research purposes, such as tissue engineering and drug development, and typically requires large-scale, external 3D printers to create the cellular structures. According to the team, in the next few years, the technology could be used to reach and operate on hard-to-reach areas in the body. In the project’s next stage, the F3DB will be used in animal test subjects, as well as further development of the arm to include an integrated camera and scanning system.

|
Rescooped by
Dr. Stefan Gruenwald
from Virus World
April 3, 2023 2:49 PM
|
Aging, or senescent cells, which stop dividing but don’t die, can accumulate in the body over the years and fuel chronic inflammation that contributes to conditions such as cancer and degenerative disorders. In mice, eliminating senescent cells from aging tissues can restore tissue balance and lead to an increased healthy lifespan. Now a team led by investigators at Massachusetts General Hospital (MGH) has found that the immune response to a virus that is ubiquitously present in human tissues can detect and eliminate senescent cells in the skin. For the study, which is published in Cell, the scientists analyzed young and old human skin samples to learn more about the clearance of senescent cells in human tissue. The researchers found more senescent cells in the old skin compared with young skin samples. However, in the samples from old individuals, the number of senescent cells did not increase as individuals got progressively older, suggesting that some type of mechanism kicks in to keep them in check. Experiments suggested that once a person becomes elderly, certain immune cells called killer CD4+ T cells are responsible for keeping senescent cells from increasing. Indeed, higher numbers of killer CD4+ T cells in tissue samples were associated with reduced numbers of senescent cells in old skin. When they assessed how killer CD4+ T cells keep senescent cells in check, the researchers found that aging skin cells express a protein, or antigen, produced by human cytomegalovirus, a pervasive herpesvirus that establishes lifelong latent infection in most humans without any symptoms. By expressing this protein, senescent cells become targets for attack by killer CD4+ T cells. “Our study has revealed that immune responses to human cytomegalovirus contribute to maintaining the balance of aging organs,” says senior author Shawn Demehri, MD, PhD, director of the High Risk Skin Cancer Clinic at MGH and an associate professor of Dermatology at Harvard Medical School. “Most of us are infected with human cytomegalovirus, and our immune system has evolved to eliminate cells, including senescent cells, that upregulate the expression of cytomegalovirus antigens.” These findings, which highlight a beneficial function of viruses living in our body, could have a variety of clinical applications. “Our research enables a new therapeutic approach to eliminate aging cells by boosting the anti-viral immune response,” says Demehri. “We are interested in utilizing the immune response to cytomegalovirus as a therapy to eliminate senescent cells in diseases like cancer, fibrosis and degenerative diseases.” Demehri notes that the work may also lead to advances in cosmetic dermatology, for example in the development of new treatments to make skin look younger.
Via Juan Lama

|
Scooped by
Dr. Stefan Gruenwald
March 31, 2023 12:45 PM
|
P. asymbiotica, a bacteria that normally grows inside C.elegans roundworms, uses these nematodes as Trojan horses to invade insect larvae. It works like this: a nematode invades the larva's body and regurgitates P. asymbiotica; the bacteria kills the insect's cells; and the nematode feasts on the dying larva's flesh. Thus, the nematodes and bacteria enjoy a beautiful symbiotic relationship. To kill the insect cells, P. asymbiotica secretes tiny, spring-loaded syringes, scientifically known as "extracellular contractile injection systems," that carry toxic proteins inside a hollow "needle" with a spike on one end. Small "tails" extend from the base of the syringe — imagine the landing gear of a space probe — and these tails bind to proteins on the surface of insect cells. Once bound, the syringe stabs its needle through the cell membrane to release its cargo. In previous studies, scientists isolated these syringes from Photorhabdus bacteria and also discovered that some could target mouse cells, not just insect cells. This raised the possibility that such syringes could be modified for use in humans. To test whether this idea might be feasible, the team first loaded the syringe's hollow tube with proteins of their choosing. Then, they used AlphaFold to better understand how the syringes hone in on insect cells, so they could be modified to target human cells instead. They used the AI system to predict the structure of the bottom of the syringe's landing gear — the part that first makes contact with the target cell surface. They then altered this structure so it would latch onto surface proteins found only on human cells. Without Deepmind's AlphaFold, the researchers would have had to conduct this analysis using advanced microscopy techniques and crystallography, meaning detailed studies of the landing gear's atomic structure, Joseph Kreitz, a doctoral student at the McGovern Institute for Brain Research at MIT and first author of the study, told Live Science in an email. "This could have taken many months," Kreitz said. "With AlphaFold, we were able to obtain predicted structures of candidate tail fiber designs almost in real-time, significantly accelerating our efforts to reprogram this protein." The researchers then used their modified syringes to tweak cells' genomes in lab dishes. Specifically, they delivered components of the powerful CRISPR-Cas9 gene editing tool into cells to cut and paste sections of DNA into their genomes. The team also used the syringes to insert tiny DNA-snipping scissors called zinc-finger deaminases into cells. They also used the system to deliver toxic proteins into cancer cells in lab dishes. And finally, they injected the syringes into live mice and found that their cargo could only be detected in the targeted areas and did not spark a harmful immune reaction. For this last experiment, the team used AlphaFold to design their syringes to specifically target mouse cells. These experiments demonstrate that the syringes can serve as "programmable protein delivery devices with possible applications in gene therapy, cancer therapy and biocontrol," the authors concluded. In contrast to therapies that deliver genetic instructions, like DNA or RNA, into cells, these protein-carrying syringes could provide "better control over the dose and half-life of a therapeutic inside cells," Kreitz and the study's senior author Feng Zhang told Live Science in an email. That's because genetic instructions prompt cells to build proteins for themselves, whereas the syringes would come with a premeasured dose of protein. This precise dosing would be useful for treatments involving transcription factors, which tweak a cell's gene activity, and chemotherapy, which has toxic effects at high doses, they said. The tiny syringes could also potentially be programmed to fight disease-causing bacteria in the body, Ericson and Pilhofer wrote. And in the future, it may be possible for scientists to connect multiple syringes to form multi-barreled complexes. "These might enable more cargo to be delivered per target cell than with a single injection system," they suggested. "However, we note that this system is still in its infancy; further efforts will be required to characterize the behavior of this system in vivo before it can be applied in clinical or commercial settings," Kreitz and Zhang told Live Science. The team is now studying how well the syringes diffuse through different tissues and organs, and continuing to examine how the immune system reacts to the new protein delivery system.

|
Rescooped by
Dr. Stefan Gruenwald
from TAL effector science
March 28, 2023 8:43 PM
|
Mitochondria are critical organelles that form networks within our cells, generate energy dynamically, contribute to diverse cell and organ function, and produce a variety of critical signaling molecules, such as cortisol. This intracellular microbiome can differ between cells, tissues, and organs. Mitochondria can change with disease, age, and in response to the environment. Single nucleotide variants in the circular genomes of human mitochondrial DNA are associated with many different life-threatening diseases. Mitochondrial DNA base editing tools have established novel disease models and represent a new possibility toward personalized gene therapies for the treatment of mtDNA-based disorders. Primary mtDNA-based disorders are currently incurable [ 2] and these diseases often cause significant illness and can lead to premature death [ 2]. Moreover, variations in mtDNA can occur in individuals who are otherwise healthy and have been implicated in the etiology of age-related multifactorial diseases, including neurodegenerative disorders such as Parkinson’s disease, metabolic conditions, heart failure, and several forms of cancer [ 10, 15]. The increasing prevalence of these conditions in our aging population highlights the need for the development of novel approaches for the investigation and prevention or treatment of these disorders [ 12, 16]. Gene editing techniques can introduce targeted DNA modifications in cells or tissues to correct a genetic defect, and have already been successfully used to correct pathogenic mutations in the nuclear genome [ 17, 18]. Mitochondrial gene therapy is a relatively new idea that, despite several challenges, has seen significant progress over the last few years [ 19]. Mitochondrial gene editing technologies can be designed to specifically act on variant mtDNA molecules, driving a heteroplasmic state toward a healthy, wild-type mtDNA population [ 20, 21]. Broadly, two distinct modalities are currently used for mtDNA manipulation: (i) nuclease-based and (ii) base editing approaches. Conceptually, nuclease-based methods can be utilized to decrease the amount of variant mtDNA in mitochondria by specifically targeting and cleaving the mutant mtDNA molecules [ 22, 23]. This technique relies on the premise that double-strand breaks (DSBs) in mtDNA induce the rapid degradation of the linearized molecule, instead of its repair [ 20, 23]. If mutant mtDNA is specifically eliminated, the residual mtDNA, mostly wild-type, replicates and repopulates the organelle, resulting in the restoration of normal mtDNA levels. In particular, nuclease-based approaches have been described to include mitochondrially targeted restriction endonucleases (mitoREs), zinc-finger nucleases (mtZFNs), and transcription activator-like effector nucleases (mitoTALENs) [ 20, 22, 23, 24, 25, 26]. These techniques have been extensively reviewed elsewhere [ 27, 28, 29, 30]. Additionally, mitochondrially targeted CRISPR (mitoCRISPR) systems have also been reported [ 31, 32, 33, 34]. However, these mitoCRISPR platforms have yet to be widely accepted within the scientific community due to the challenging nature of guide RNA (gRNA) import into the mitochondrial matrix, as well as a notable lack of follow-up studies [ 27, 35, 36]. Despite their potential usefulness for shifting heteroplasmy, mitochondrically-targeted nucleases are unable to correct variant genomes [ 20, 22]. Therefore, nuclease-based approaches cannot be utilized to rescue pathological conditions in homoplasmic states [ 20, 28]. As mentioned above, the main alternative to nuclease-based strategies for mtDNA manipulation is base editing [ 37, 38]. Consequently, in this work, the scientists focused on recent advancements in the field of mitochondrial base editing, which holds the potential to treat diseases caused by pathogenic mtDNA point mutations in both heteroplasmic and homoplasmic contexts without the risk for mtDNA depletion [ 39, 40]. This review also sought to provide insights into paths toward the development of therapeutic approaches for mtDNA-based disorders and the establishment of mitochondrial disease models to better understand the biology of these devastating diseases.
Via dromius
|

|
Rescooped by
Dr. Stefan Gruenwald
from Virus World
September 15, 2023 7:45 PM
|
Researchers discovered a potential link between "endogenous retroviruses" present in the human genome and the development of neurodegenerative diseases. Summary: Researchers discovered a potential link between “endogenous retroviruses” present in the human genome and the development of neurodegenerative diseases. Their study found that these ancient viral remnants might influence the spread of protein aggregates commonly associated with certain dementias. While these retroviruses don’t trigger neurodegeneration, they may exacerbate the disease process. This discovery offers new potential therapeutic avenues, such as suppressing gene expression or neutralizing viral proteins. Key Facts: - “Endogenous retroviruses”, ancient viral remnants in human DNA, might contribute to neurodegenerative disease progression.
- HERV-W and HERV-K, two such retroviruses, were found to aid in the transport of tau aggregates, protein clumps associated with diseases like Alzheimer’s.
- Potential therapeutic approaches include suppressing the retroviruses or neutralizing their proteins, possibly with antibodies.
Source: DZNE Genetic remnants of viruses that are naturally present in the human genome could affect the development of neurodegenerative diseases. Researchers at DZNE come to this conclusion on the basis of studies on cell cultures. They report on this in the journal Nature Communications. In their view, such “endogenous retroviruses” could contribute to the spread of aberrant protein aggregates – hallmarks of certain dementias – in the brain. Thus, these viral relicts would be potential targets for therapies. It has been suspected for some time that viral infections contribute to the genesis and development of neurodegenerative diseases. Laboratory studies by DZNE scientists now suggest a mechanism that, although related to viruses, does not require infection by external pathogens. According to this study, the culprits would be “endogenous retroviruses” that are naturally present in the human genome. “During evolution, genes from numerous viruses have accumulated in our DNA. Most of these gene sequences are mutated and normally muted,” explained Ina Vorberg, research group leader at DZNE and a professor at the University of Bonn. “However, there is evidence that endogenous retroviruses are activated under certain conditions and contribute to cancer and neurodegenerative diseases. Indeed, proteins or other gene products derived from such retroviruses are found in the blood or tissue of patients.” Experiments with Tau Aggregates Vorberg followed this trail together with colleagues from Bonn and Munich. Using cell cultures, the researchers simulated the situation in which human cells produce certain proteins from the envelope of endogenous retroviruses. Specifically, this involved HERV-W and HERV K – both viruses are present in the human genome but are usually dormant. However, studies indicate that HERV-W is activated in multiple sclerosis and HERV-K in the neurological disease “amyotrophic lateral sclerosis” (ALS) and in frontotemporal dementia (FTD). Now, Vorberg’s team found that the viral proteins facilitate the transport of so-called tau aggregates from cell to cell. “Tau aggregates” are tiny protein clumps that occur in the brains of people affected by certain neurodegenerative diseases – these include Alzheimer’s disease and FTD. “Certainly, conditions in the brain are much more complex than our cellular model system can replicate them. Nevertheless, our experiments show that endogenous retroviruses can influence the spread of tau aggregates between cells,” Vorberg said. “Endogenous retroviruses would thus not be triggers of neurodegeneration, but could fuel the disease process once it is already underway.” Viral Transport Mediators The current research and earlier studies by Vorberg’s team suggest that viral proteins serve as transport mediators for tau aggregates because they insert into the cell membrane and into the membrane of so-called extracellular vesicles: These are small fat bubbles that are naturally secreted by cells. “For the transport of tau aggregates from cell to cell, we see two pathways in particular. Transfer between cells that are in direct contact, and transport within vesicles that act as cargo capsules, so to speak, and pass from one cell to another to eventually merge with it,” Vorberg explained. “In both scenarios, membranes have to fuse. Proteins from the envelope of viruses can promote this process. That’s because many viruses are adapted to fuse with host cells. “This happens by means of special proteins that viruses carry on their surfaces. If precisely these proteins are incorporated into the cell membrane and the membrane of extracellular vesicles, it is understandable that the tau aggregates then spread more easily.” Starting Points for Therapy In the course of the natural aging process, the regulation of genes can change – originally “dormant” endogenous retroviruses could be “awakened” as a result. Indeed, the symptoms of most neurodegenerative diseases do not manifest until older age. This raises two conceivable approaches to therapy. “On the one hand, one could try to specifically suppress gene expression, that is, to inactivate the endogenous retroviruses again. That would get to the root of the problem,” Vorberg said. “But you could also start elsewhere and try to neutralize the viral proteins – for example, with antibodies.” Searching for Antibodies In the opinion of the researchers, it is likely that dementia patients with tau aggregates carry increased amounts of such antibodies. If it were possible to isolate these and reproduce them using biotechnological methods, it might be possible to develop a passive vaccine. Thus, in collaboration with DZNE colleagues in Berlin and Bonn, Vorberg’s team aims to specifically search for such antibodies in patients. In addition, the scientists are considering antiviral drugs. In cell culture, they have already found that such agents can actually stop the spread of protein aggregates. “This is another approach we intend to pursue,” said Vorberg. Research cited published in Nature (Aug. 18, 2023): https://doi.org/10.1038/s41467-023-40632-z
Via Juan Lama

|
Scooped by
Dr. Stefan Gruenwald
August 14, 2023 4:31 PM
|
Circulating proteins have important functions in inflammation and a broad range of diseases. To identify genetic influences on inflammation-related proteins, researchers conducted a genome-wide protein quantitative trait locus (pQTL) study of 91 plasma proteins measured using the Olink Target platform in 14,824 participants. We identified 180 pQTLs (59 cis, 121 trans).
Integration of pQTL data with eQTL and disease genome-wide association studies provided insight into pathogenesis, implicating lymphotoxin-α in multiple sclerosis. Using Mendelian randomization (MR) to assess causality in disease etiology, they identified both shared and distinct effects of specific proteins across immune-mediated diseases, including directionally discordant effects of CD40 on risk of rheumatoid arthritis versus multiple sclerosis and inflammatory bowel disease. MR implicated CXCL5 in the etiology of ulcerative colitis (UC) and they show elevated gut CXCL5 transcript expression in patients with UC.
These results identify targets of existing drugs and provide a powerful resource to facilitate future drug target prioritization.

|
Rescooped by
Dr. Stefan Gruenwald
from Virus World
August 9, 2023 4:41 PM
|
The development of cancer neoantigen vaccines that prime the anti-tumor immune responses has been hindered in part by challenges in delivery of neoantigens to the tumor. Here, using the model antigen ovalbumin (OVA) in a melanoma model, we demonstrate a chimeric antigenic peptide influenza virus (CAP-Flu) system for delivery of antigenic peptides bound to influenza A virus (IAV) to the lung. We conjugated attenuated IAVs with the innate immunostimulatory agent CpG and, after intranasal administration to the mouse lung, observed increased immune cell infiltration to the tumor. OVA was then covalently displayed on IAV-CPG using click chemistry. Vaccination with this construct yielded robust antigen uptake by dendritic cells, a specific immune cell response and a significant increase in tumor-infiltrating lymphocytes compared to peptides alone. Lastly, we engineered the IAV to express anti-PD1-L1 nanobodies that further enhanced regression of lung metastases and prolonged mouse survival after rechallenge. Engineered IAVs can be equipped with any tumor neoantigen of interest to generate lung cancer vaccines. A cancer vaccine is delivered to the lung by an engineered attenuated influenza virus. Published in Nature Biotechnology: https://doi.org/10.1038/s41587-023-01796-7
Via Juan Lama

|
Rescooped by
Dr. Stefan Gruenwald
from Virus World
July 13, 2023 4:07 PM
|
Gene therapies have made spectacular progress in delivering new cures for previously intractable disease, but they remain the world’s most expensive treatments by far. Now companies are replacing the virus in gene therapies with new delivery technologies that promise not only to overcome the limitations of viral vectors but to slash production costs too. Moderna and Generation Bio teamed up recently to develop non-viral gene therapies for liver and immune-related conditions, in a deal that builds on Generation Bio’s lipid nanoparticle (LNP) delivery platform. The collaboration is part of a growing trend to swap virus-driven gene therapeutics for innovative nucleic acid delivery platforms that escape the high costs and technical limitations of viral vectors. Engineered lipid or protein nanobodies, DNA-based nanocarriers, and novel physicochemical methods point the way toward re-dosable genetic medicines (Table 1). Virus-based vectors — particularly those based on adeno-associated virus (AAV) and lentiviruses — have dominated the first wave of gene therapies to gain regulatory approval. But their widespread deployment in dozens of clinical trials has exposed their limitations very clearly. “One of the issues is just the biomass needed to produce a sufficient amount, like AAV at the scale and in the doses needed for large indications,” says Akin Akinc, CEO of Aera Therapeutics, a company developing a protein-nanoparticle-based delivery system. AAV vectors are further limited by their 4.7-kilobase packaging capacity; poor tissue selectivity; risk of liver toxicity; and immunogenicity, which eliminates the possibility of redosing. Lentiviral vectors and, to a lesser extent, AAV vectors also elicit oncogenicity concerns arising from chromosomal integration and insertional mutagenesis. Moreover, the theoretical risk of replication-competent viruses emerging from recombination events during lentiviral production necessitates extensive testing in patients. To provide a viable alternative, non-viral technologies need to satisfy certain well-defined requisites. They must accommodate a large payload and deliver it to specific organs, lack immunogenicity to allow re-dosing, and have a high safety margin and low production costs. Although most are still preclinical, the field is gaining momentum. In recent months, Aera raised $193 million to progress a protein-nanoparticle system based on endogenous human proteins. These assemble spontaneously into capsid-like structures that can deliver a nucleic acid cargo. Intergalactic Therapeutics plans to start clinical trials next year of a gene therapy that employs in vivo electroporation to deliver covalently closed, circular DNA molecules to retinal cells. And ReCode Therapeutics has just entered the clinic with a nebulized LNP that delivers mRNA encoding a protein, DNAI, to the lungs of patients with primary ciliary dyskinesia arising from mutations in dynein axonemal intermediate chain 1 (DNAI1). The global rollout of inexpensive LNP-based mRNA COVID-19 vaccines has demonstrated that it is feasible to manufacture and deliver billions of doses, including repeat doses, of at least one form of nucleic acid. There is, however, a substantial difference between delivering mRNA to antigen-presenting cells locally in muscle and delivering LNP-encapsulated DNA or other cargoes to the cell nucleus within target tissues, be they in the liver, muscle, lungs, heart or other organs, while avoiding triggering innate immune responses. The cell targeting needs to be more sophisticated while the DNA payload may also require modification to ensure it is efficiently localized to the nucleus upon cell entry. For example, Generation Bio’s closed-end DNA (ceDNA) has a covalently closed structure to resist nuclease attack in the cytoplasm and two inverted terminal repeats flanking the transgenes and its regulatory elements to promote nuclear uptake. The liver is another obstacle for any systemically delivered gene therapy not intended for that organ, because it scavenges from the circulation viral and non-viral vectors alike, along with other exogenous and endogenous macromolecules and nanoparticles. The key culprit is the serum protein apolipoprotein E (ApoE), says David Lockhart, president and CSO of ReCode. It promiscuously binds circulating LNPs or other delivery vehicles, and most of them end up in the liver, as it is rich in ApoE-binding low-density lipoprotein receptors. “If you don’t avoid ApoE binding, the liver is a sink,” he says. To direct delivery to different organs, Recode adds a fifth, selective organ targeting (SORT) lipid to the classical four-lipid nanoparticle composition, an approach developed by company co-founder Daniel Siegwart of the University of Texas Southwestern Medical Center. “It’s not one magic lipid — this is a class effect,” Lockhart says. These lipids bind specific serum proteins whose receptors are strongly expressed by the target cells. Thus, adding a lipid that binds the adhesion protein vitronectin improves delivery to the lung, whereas one that binds β2-glycoprotein 1 boosts delivery to the spleen. Generation Bio also adds a fifth, targeting ligand to its LNPs to avoid default trafficking to resident macrophages in the liver and spleen, but the mechanism differs from that of ReCode’s. “It’s a direct receptor engagement,” says Matthew Stanton, CSO at Generation Bio. The company has not yet published details of its ‘stealth’ LNPs but claims it can avoid default trafficking to the liver and spleen, particularly to their resident macrophages. “That’s what got Moderna so excited,” he says. Anjarium Biosciences of Schlieren, Switzerland, achieves cell-specific delivery with antibodies or antibody fragments, which it attaches to its proprietary ‘hybridosme’ nanoparticles. The latter are based on LNPs and exosomes. “We have a composite particle with elements of each,” says CSO and co-founder Joël de Beer. Exosomes have proven to be difficult to load with genetic cargo, he says, but they can be readily modified externally. They are also immunologically silent, given their ubiquity in human circulation and tissues. “The issue with LNPs of course is biodistribution,” he says. But they are efficient carriers of nucleic acids. The company’s aim is to capture their combined benefits while avoiding their shortcomings. Delivery vehicles based on other materials can avoid the issue of liver trafficking from the outset. Code Biotherapeutics is developing a DNA-based nanocarrier that has no inherent liver tropism. Its technology comprises a multivalent structure assembled by hybridizing multiple single strands into double-stranded monomers, which are then cross-linked to form a stable particle. Up to 18 copies of a therapeutic payload and another 18 of a targeting moiety can be attached externally. “We have pretty precise control over how we can build that,” says Brian McVeigh, CEO and co-founder of Code. Yet another option is to build carriers from phage proteins. Gensaic is using three capsid proteins from phage M13, a resident of the human microbiome, in protein-based nanoparticles. “These particles don’t have a pre-existing tropism,” says CEO Lavi Erisson. The protein-based system is readily amenable both to protein engineering methods, for the addition of targeting moieties, and to high-throughput screening methods, for the identification of particles with the desired attributes. The company’s programs remain at an early stage, but efforts in Duchenne muscular dystrophy, cystic fibrosis and central nervous system disorders are underway. Aera’s approach also involves proteins that are present in the body. They are derived from retroelements, such as human endogenous retroviruses and retrotransposons, which constitute a sizeable percentage of the human genome. “By starting with human proteins, we’re starting at a lower immunogenic risk,” says CEO Akinc. Scientific founder Feng Zhang of the Broad Institute of MIT and Harvard has described one such particle, based on a protein called PEG10. Jason Shepherd of the University of Utah has supplied Aera with another protein nanoparticle, based on a protein called Arc, which is expressed in the brain. The company is actively seeking others. “I tend to think there’s going to be better ones out there,” says Akinc. For manufacturing, Aera is employing a cell-free process, unlike the cell-based process that Zhang and colleagues described in their original report. “We can precisely control what we’re putting into the mixture,” says Akinc. The company has not yet disclosed any development programs, but its initial focus will be on short interfering RNA (siRNA), mRNA and gene-editing cargoes. “We are interested in DNA delivery as well,” he says. “I tend to think there’s going to be better ones out there.” Other types of nanoparticles — polymer-based, for instance — do not traffic to the liver by default so developers do not have to deal with that problem. “Typically they’re designed or discovered on a per-tissue basis,” says Sean Kevlahan, CEO and co-founder of Nanite. The rules that determine why a particular particle traffics to a particular tissue are not fully understood, however, and the company is addressing this issue. “If you don’t understand why, then you can’t design them effectively,” he says. To explore the vast chemical space of polymer nanoparticles, the company has developed rapid synthesis and computational methods. It recently secured funding from the Cystic Fibrosis Foundation to identify polymer-based nanoparticles equipped to penetrate the thick mucus that clogs the airways of patients and deliver an mRNA payload. For working ex vivo or for tissues that are readily accessible, scientists may choose to apply physicochemical tools. Apart from one from SQZ Biotech, which is now struggling to stay afloat, most tools remain in preclinical development, although they are edging closer to the clinic. Intergalactic is employing a proprietary needle-like electrode to deliver its C3 double-stranded DNA molecules to target cells. These are assembled in vitro from libraries of transgenes and regulatory elements and then combined into the final product in engineered bacterial cells, which ensures ease of manufacture and low cost of goods. The final DNA product is fully human and does not contain any viral or bacterial sequences. In the company’s lead program, aimed at ABCA4 retinopathy, a solution containing the DNA is administered by subretinal injection and the same area is then exposed to a low-energy electric field. The method is broadly applicable. “Any tissue that can be biopsied is a potential candidate for local [gene] delivery,” says José Lora, Intergalactic’s CSO. Maynooth, Ireland-based Avectas is developing a novel method of ex vivo gene transfer, in which it transiently exposes cells to a solubilizing spray, which contains ethanol and other constituents. This temporarily permeabilizes the cell membrane, allowing macromolecules, such as nucleic acids or gene editing components, to cross into the cytoplasm. Lab tests on T-cells suggest it is on a par with electroporation in terms of gene transfer efficiency, but the transfected cells exhibit less perturbation of gene expression. “Ultimately, that results in a cell that has the potential to be more highly functional,” says Justin McCue, chief technology officer at Avectas. Ultimately, gene therapy’s claims of ‘one and done’ may need revising as transgene expression wanes over time, and if the therapy is virus-based, potential immunogenicity means patients will be unable to receive a second dose. Non-viral gene delivery particles that can reliably demonstrate a lack of immunogenicity could eliminate that issue. What’s more, they could also change the safety equation during clinical development. “It’s not about getting to the maximum-tolerated dose and seeing if you’re efficacious,” says Stanton. Instead, developers can stack multiple “well-tolerated doses” over time, he says, which allows patient-specific dose titration. And if those nanoparticles are also able to evade the liver and reach their target organ, the whole experience of gene therapy will become similar to taking a biologic drug. It will take time and effort to deliver this vision — but it is now starting to take shape. Published in Nat. Biotechnology (June 14, 2023):
Via Juan Lama

|
Scooped by
Dr. Stefan Gruenwald
July 7, 2023 6:52 PM
|
Creating new drug candidates is a heroic endeavor, often taking over 10 years to bring a drug to market. New supercomputing-scale large language models (LLMs) that understand biology and chemistry text are helping scientists understand proteins, small molecules, DNA, and biomedical text. These state-of-the-art AI models help generate de novo proteins and molecules and predict the 3D structures of proteins. They can predict the binding structure of a small molecule to a protein and are offering scientists easier ways to engineer new candidate drugs and ultimately bring hope for patients. After Exscientia brought an AI-designed drug candidate to a clinical trial in 2021, several other companies have announced that their candidates are in trials. Within drug companies focused on AI-based discovery, there is publicly available information on about 160 discovery programs, of which 15 products are reportedly in clinical development. At the forefront of AI-based drug discovery are generative AI models for applications such as generating high-quality proteins. These large, powerful models learn from unlabeled data (such as sequencing data) on multi-GPU, multi-node, high-performance computing (HPC) infrastructures. With the novel NVIDIA BioNeMo Service, workflows for generative AI for biology are optimized and turnkey. You can focus on adapting AI models to the right drug candidates instead of dealing with configuration files and setting up supercomputing infrastructure. BioNeMo Service BioNeMo Service is a cloud service for generative AI in early drug discovery, featuring nine state-of-the-art large language and diffusion models in one place. The models in BioNeMo are accessible through a web interface or fully managed APIs and can be further trained and optimized on NVIDIA DGX Cloud. With BioNeMo Service, you can perform any of the following tasks: - Generate large libraries of proteins.
- Build property predictors using embeddings to refine protein libraries.
- Generate small molecules with specific properties.
- Rapidly and accurately predict and visualize the 3D structure for billions of proteins.
- Run large campaigns of ligand-to-small-molecule pose estimations.
- Download proteins, molecules, and predicted 3D structures.
Generative AI models in BioNeMo Service BioNeMo Service features nine AI generative models covering a wide spectrum of applications for developing AI drug discovery pipelines: - AlphaFold 2, ESMFold, and OpenFold for 3D protein structure prediction from a primary amino acid sequence
- ESM-1nv and ESM-2 for protein property predictions
- ProtGPT2 for protein generation
- MegaMolBART and MoFlow for small molecule generation
- DiffDock for predicting the binding structure of a small molecule to a protein

|
Scooped by
Dr. Stefan Gruenwald
July 1, 2023 4:07 PM
|
While generative AI is a relatively new household term, drug discovery company Insilico Medicine has been using it for years to develop new therapies for debilitating diseases. The company’s early bet on deep learning is bearing fruit — a drug candidate discovered using its AI platform is now entering Phase 2 clinical trials to treat idiopathic pulmonary fibrosis, a relatively rare respiratory disease that causes progressive decline in lung function. Insilico used generative AI for each step of the preclinical drug discovery process: to identify a molecule that a drug compound could target, generate novel drug candidates, gauge how well these candidates would bind with the target, and even predict the outcome of clinical trials. Doing this using traditional methods would have cost more than $400 million and taken up to six years. But with generative AI, Insilico accomplished them for one-tenth of the cost and one-third of the time — reaching the first phase of clinical trials just two and a half years after beginning the project. “This first drug candidate that’s going to Phase 2 is a true highlight of our end-to-end approach to bridge biology and chemistry with deep learning,” said Alex Zhavoronkov, CEO of Insilico Medicine. “This is a significant milestone not only for us, but for everyone in the field of AI-accelerated drug discovery.” Insilico is a premier member of NVIDIA Inception, a free program that provides cutting-edge startups with technical training, go-to-market support and AI platform guidance. The company uses NVIDIA Tensor Core GPUs in its generative AI drug design engine, Chemistry42, to generate novel molecular structures — and was one of the first adopters of an early precursor to NVIDIA DGX systems in 2015. AI Enables End-to-End Preclinical Drug Discovery Insilico’s Pharma.AI platform includes multiple AI models trained on millions of data samples for a range of tasks. One AI tool, PandaOmics, rapidly identifies and prioritizes targets that play a significant role in a disease’s effectiveness — like the infamous spike protein on the virus that causes COVID-19. The Chemistry42 engine can design within days new potential drug compounds that target the protein identified using PandaOmics. The generative chemistry tool uses deep learning to come up with drug-like molecular structures from scratch. “Typically, AI companies in drug discovery focus either on biology or on chemistry,” said Petrina Kamya, head of AI platforms at Insilico. “From the start, Insilico has been applying the same deep learning approach to both fields, using AI both to discover drug targets and generate chemical structures of small molecules.” Over the years, the Insilico team has adopted different kinds of deep neural networks for drug discovery, including generative adversarial networks and transformer models. They’re now using NVIDIA BioNeMo to accelerate the early drug discovery process with generative AI.

|
Scooped by
Dr. Stefan Gruenwald
May 27, 2023 10:51 AM
|
A team of engineers at the University of Colorado Boulder has designed a new class of tiny, self-propelled robots that can zip through liquid at incredible speeds -- and may one day even deliver prescription drugs to hard-to-reach places inside the human body. The researchers describe their mini healthcare providers in a paper published last month in the journal Small. "Imagine if microrobots could perform certain tasks in the body, such as non-invasive surgeries," said Jin Lee, lead author of the study and a postdoctoral researcher in the Department of Chemical and Biological Engineering. "Instead of cutting into the patient, we can simply introduce the robots to the body through a pill or an injection, and they would perform the procedure themselves." Lee and his colleagues aren't there yet, but the new research is big step forward for tiny robots. The group's microrobots are really small. Each one measures only 20 micrometers wide, several times smaller than the width of a human hair. They're also really fast, capable of traveling at speeds of about 3 millimeters per second, or roughly 9,000 times their own length per minute. That's many times faster than a cheetah in relative terms. They have a lot of potential, too. In the new study, the group deployed fleets of these machines to transport doses of dexamethasone, a common steroid medication, to the bladders of lab mice. The results suggest that microrobots may be a useful tool for treating bladder diseases and other illnesses in people. "Microscale robots have garnered a lot of excitement in scientific circles, but what makes them interesting to us is that we can design them to perform useful tasks in the body," said C. Wyatt Shields, a co-author of the new study and assistant professor of chemical and biological engineering. Fantastic Voyage If that sounds like something ripped from science fiction, that's because it is. In the classic film Fantastic Voyage, a group of adventurers travels via a shrunken-down submarine into the body of a man in a coma. "The movie was released in 1966. Today, we are living in an era of micrometer- and nanometer-scale robots," Lee said. He imagines that, just like in the movie, microrobots could swirl through a person's blood stream, seeking out targeted areas to treat for various ailments. The team makes its microrobots out of materials called biocompatible polymers using a technology similar to 3D printing. The machines look a bit like small rockets and come complete with three tiny fins. They also include a little something extra: Each of the robots carries a small bubble of trapped air, similar to what happens when you dunk a glass upside-down in water. If you expose the machines to an acoustic field, like the kind used in ultrasound, the bubbles will begin to vibrate wildly, pushing water away and shooting the robots forward.

|
Scooped by
Dr. Stefan Gruenwald
May 15, 2023 6:04 PM
|
AI algorithms can screen for pancreatic cancer and predict whether patients will develop the disease up to three years before a human doctor can make the same diagnosis, according to research published in Nature on Monday.
Pancreatic cancer is deadly; the five-year survival rate averages 12 percent. Academics working in Denmark and the US believe AI could help clinicians by detecting pancreatic cancer at earlier stages, if the software can reliably predict which patients are at higher risk of developing the disease.
The researchers trained AI algorithms on millions of medical records obtained in the Danish National Patient Registry and the US Veterans Affairs Corporate Data Warehouse. The models were trained to correlate diagnosis codes – labels used by hospitals describing different medical conditions – to pancreatic cancer.
Some diagnosis codes for jaundice, abdominal and pelvic pain, weight loss, for example, are more closely related to the disease – especially if they are found in patients about six months before diagnosis – while others like Type 2 diabetes, anemia, or inflammation of the pancreas are usually found earlier.
"Cancer gradually develops in the human body, often over many years and fairly slowly, until the disease takes hold," Chris Sander, the study's co-senior investigator and leader of a lab working at the Department of Systems Biology at Harvard Medical School, told The Register.
"The AI system attempts to learn from signs in the human body that may relate to such gradual changes. But it is early days for this, and while the AI system can make reasonably accurate predictions, it cannot, or not currently, identify mechanisms or causative events. Like often in science, correlation is useful for prediction, but causation is much harder to establish," he said.
Researchers teach AI to pinpoint mutations linked to cancer
The most effective model, based on a transformer-based architecture, showed that out of the top 1,000 highest-risk patients over 50, about 320 would go on to develop pancreatic cancer. The model is less accurate when trying to predict pancreatic cancer over longer time intervals compared to shorter ones, and for patients younger than 50.
"AI on real-world clinical records has the potential to produce a scalable workflow for early detection of cancer in the community, to shift focus from treatment of late-stage to early-stage cancer, to improve the quality of life of patients and to increase the benefit/cost ratio of cancer care," the paper reads.
Effective prediction in real-world settings will rely on the quality of patients' medical histories. Future AI-based screening tools for pancreatic cancer will have to be trained on specific local population data, the study found. A model trained on data from Danish patients, for example, was not as accurate when applied to US patients.
"Given the experience in Denmark and one or two US health systems, this means that in each country with different conditions and different systems, it is best to re-train the model locally. AI needs a lot of data to train. Access in different locations is not straightforward, as medical records are and should be confidential. So local approval and data security is essential," Sander said.
The study is still in its early stages, and the software cannot yet be used to run screening programs. Improvements are needed before even a trial can be conducted. "Once a surveillance program is implemented, the actual computing costs for applying the software are moderate. The training is what consumes considerable computing resources. The actual clinical tests to see early signs of cancer or to detect cancer when it is still very small are costly, much more expensive than for example mammograms," Sander added. Still, the team believes that as the technology improves and operating costs decrease, AI could become a valuable screening tool in the future.

|
Rescooped by
Dr. Stefan Gruenwald
from Virus World
April 18, 2023 4:24 PM
|
Researchers reviewed data on the incidence of autoimmune diseases in the post-acute coronavirus disease 2019 period. Background COVID-19 has caused unprecedented morbidity and mortality across the globe. The immune protection conferred by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) vaccines and prior COVID-19 history, coupled with developing effective therapeutic agents, has reduced COVID-19-associated mortality. However, post-COVID-19 conditions continue to increase, particularly new-onset autoimmune diseases in COVID-19 convalescents. Autoimmune diseases have been reported following COVID-19 among adults; however, the prevalence and magnitude of the diseases, and the incidence risks among individuals infected with SARS-CoV-2, compared to uninfected individuals, are not well-characterized. Improving understanding of the impact of COVID-19 on the risk of developing post-acute COVID-19 complications, such as autoimmune disease, could aid in implementing preventive measures and initiating prompt treatment to prevent COVID-19-associated morbidities. The findings would also be highly relevant for future pandemics and to analyze the long-term protective effects of COVID-19 vaccines. About the review In the review, researchers presented findings of two large-scale cohort studies, evaluating the incidence of autoimmune diseases following acute SARS-CoV-2 infections using participants' electronic medical records. Evidence on post-COVID autoimmune disease obtained before the studies Studies comprising pediatric COVID-19 patients suffering from multisystem inflammatory syndrome among children (MIS-C) have indicated that COVID-19 results in dysregulated immunological responses. The clinical manifestations of MIS-C overlap with hyperinflammatory syndromes, including macrophage activation syndrome, Kawasaki disease, and toxic shock syndrome. The pathophysiology of COVID-19-associated immunological dysfunction includes molecular mimicry by SARS-CoV-2 proteins, multiorgan involvement due to the presence of angiotensin-converting enzyme 2 (ACE2) receptors, essential for SARS-CoV-2 entry into host cells, bystander immune cell activation, autoantigen release following SARS-CoV-2-induced tissue damage, superantigen-regulated lymphocyte activation, and epitope spread. Additionally, factors such as genetic susceptibility, age, and comorbidities may contribute to COVID-19 pathogenesis. A previous study comparing the immune responses in SARS-CoV-2 infection and autoimmune disease reported that tissue damage in both conditions is mainly immune-regulated, demonstrated by the presence of anti-nuclear antibodies, anti-Ro/SSA antibodies, and lupus anticoagulant cold agglutinins in both conditions. A retrospective analysis was performed from January 31, 2020, to June 30, 2021, using the Clinical Practice Research Datalink Aurum database, comprising data from 458,147 and 1,818,929 SARS-CoV-2-infected and uninfected adults, respectively, residing in England. A study preprint reported that the incidence of inflammatory bowel disease, psoriasis, and type 1 diabetes was significantly related to COVID-19. Findings of the two cohort studies assessed post-COVID autoimmune disease Chang et al. utilized the TriNetX health research network, comprising six million adults across 48 health organizations globally. The propensity score-matched SARS-CoV-2-infected and uninfected groups comprised 887,455 COVID-19 vaccinees each. Post-COVID-19 autoimmune disease incidence was evaluated between January 1, 2020, and December 31, 2021. At six months of follow-up, autoimmune disease incidence was significantly greater in SARS-CoV-2-infected individuals, including systemic lupus erythematosus, rheumatoid arthritis, vasculitis, type 1 diabetes, and inflammatory bowel disease, with adjusted hazards ratios (aHR) of 2.99,2.98, 1.96, 2.68, and 1.78, respectively. Post-COVID autoimmune disease risk was consistent across ages. Tesch et al. conducted a study, yet to be peer-reviewed, to evaluate autoimmune disease risks among 640,701 unvaccinated individuals having polymerase chain reaction (PCR)-confirmed SARS-CoV-2 infections in 2020. The findings showed that COVID-19 patients were 43.0% more likely to acquire autoimmune conditions, within three to 15.0 months of infection, compared to over one million age- and sex-matched uninfected individuals. Among autoimmune diseases, the incidence rate ratio (IRR) values were the highest for vasculitis. Further, among those with prior history of autoimmune diseases, COVID-19 resulted in a 23.0% higher risk of another autoimmune disease. Generally, most autoimmune diseases were not COVID-19-specific; however, an essential aspect of SARS-CoV-2 infection was a considerable rise in the incidence and spectrum of autoimmune diseases following acute COVID-19. Conclusions Based on the review findings, COVID-19 elevates autoimmune disease risks. Due to the retrospective study designs, the two cohort studies could not provide a causal association between COVID-19 and autoimmune disease development. However, the temporal relationship with COVID-19 history provides reliable and compelling evidence that COVID-19 is associated with elevated autoimmune disease development risks. Health governments and authorities must conduct future studies on the topic to obtain national data and increase the generalizability of the findings. The definitive mechanisms, including genetic and epigenetic predilection, underlying the association and related pathophysiology are not completely understood. However, further research must be performed using particular gene-deficient experimental animals, bioinformatic analysis, and biological approaches, e.g., analyzing transcriptomic pandemic data to obtain genomic signatures, and host responses to viral triggers must be evaluated. Research cited published (April 12, 2023) In Nature Reviews Rheumatology: https://www.nature.com/articles/s41584-023-00964-y
Via Juan Lama

|
Rescooped by
Dr. Stefan Gruenwald
from Virus World
April 3, 2023 2:51 PM
|
A research team led by gene editing pioneer David R. Liu, PhD, reports the application of base editing technology to develop a one-time treatment for spinal muscular atrophy (SMA), showing promising effectiveness in cell and mouse models. Liu, a professor at Harvard University, the Broad Institute, and an investigator with the Howard Hughes Medical Institute, together with 16 co-authors, published their findings today in Science, in a report entitled, “Base editing rescue of spinal muscular atrophy in cells and in mice.” The paper is the first one published in a peer-reviewed journal that details a base editing approach to treating SMA—the leading genetic cause of infant mortality. Two months ago, Benjamin P. Kleinstiver, PhD, assistant professor in the Center for Genomic Medicine at Massachusetts General Hospital (MGH) and Harvard Medical School, and 12 co-authors posted a related preprint on bioRxiv detailing successful results from their study to optimize and implement a base editing treatment for SMA. In SMA, patients have either a loss of, or mutations in, the SMN1 gene. The result is a lack of its corresponding protein SMN, which is critical to cellular functions that include those required for embryo development. Liu and co-authors detailed how they restored SMN protein abundance to normal levels and rescued disease phenotypes in cell and mouse models of SMA by developing one-time genome editing approaches targeting another gene, called SMN2. SMN2 differs from SMN1 at just one position—a single C-to-T mutation in exon 7. The result is a defective truncated SMN protein that fails to fully compensate for the loss of SMN1, but which provides just enough SMN protein activity to allow babies to be born with SMA. In the most common SMA cases (type 1), those babies typically live less than two years without treatment. Liu and colleagues developed a customized base editor (out of more than 40 tested) that directly converts SMN2 to the same sequence as SMN1 by reversing the C-to-T mutation that occurred during the duplication of SMN1 to SMN2. The investigators reported that their strategy converted SMN2 to SMN1 with near-ideal editing efficiency of 99% in cells, fully restoring SMN protein levels ~40-fold to normal levels, while generating minimal off-target edits. “Because all SMA patients have SMN2—otherwise loss of SMN1 would not allow them to be born—this base editing approach to treating SMA also should be applicable to all SMA patients, regardless of the specific mutation that caused their SMN1 loss,” Liu said.
Via Juan Lama

|
Scooped by
Dr. Stefan Gruenwald
April 1, 2023 1:53 AM
|
Original article is here Artificial intelligence has developed a treatment for cancer in just 30 days and can predict a patient’s survival rate. In a new study published in the journal Chemical Science, researchers at the University of Toronto along with Insilico Medicine developed a potential treatment for hepatocellular carcinoma (HCC) with an AI drug discovery platform called Pharma.AI. HCC is the most common type of liver cancer and occurs when a tumor grows on the liver, according to Cleveland Clinic. Researchers applied AlphaFold, an AI-powered protein structure database, to Pharma.AI to uncover a novel target — a previously unknown treatment pathway — for cancer and developed a “novel hit molecule” that could bind to that target without aid. The creation of the potential drug was accomplished in just 30 days from the selection of the target and after synthesizing just seven compounds. After a second round of generating compounds, they discovered a more potent hit molecule — but any potential drug would need to go through clinical trials before widespread use.

|
Scooped by
Dr. Stefan Gruenwald
March 30, 2023 3:42 PM
|
“Whole genome doubling (WGD)”, a process where an entire set of chromosomes within a cell is duplicated, occurs in approximately 30% of all human cancers. This event causes genomic instability within the cell, potentially leading to chromosomal changes and other mutations that contribute to cancer development. Now, a team of researchers led by Elisa Oricchio at EPFL and Giovanni Ciriello at UNIL, has uncovered a new clue as to how WGD drives cancer. In a study published in Nature, the scientists show that WGD can affect the 3D organization of the chromatin inside the cell through a phenomenon called “loss of chromatin segregation”. The researchers looked at cells that lack the tumor suppressor gene p53, making them prone to WGD. They found that WGD leads to a reduction in the segregation of chromatin’s structural elements, such as loops, domains, and compartments, upending its careful organization in the cell. The result is a mixing of genetic material that is normally kept separate, changing the position of genomic regions in the 3D space, known as “sub-compartment repositioning.” This sets the stage for the activation of oncogenes, which are genes that contribute to the development of cancer. The researchers also found that the effects of WGD on chromatin organization are largely independent of chromosomal alterations, meaning that loss of chromatin segregation and chromosomal instability are complementary mechanisms that work together to promote cancer development. The work provides a new way of looking at the role of WGD and chromatin organization in the development of cancer. In the future, highly multiplexed single-cell molecular profiles, combined with barcoding technologies and new computational approaches, could help to further uncover what role disorganization of chromatin’s 3D structure plays in transforming a cell into a cancerous one. Reference: “Whole-genome doubling drives oncogenic loss of chromatin segregation” by Ruxandra A. Lambuta, Luca Nanni, Yuanlong Liu, Juan Diaz-Miyar, Arvind Iyer, Daniele Tavernari, Natalya Katanayeva, Giovanni Ciriello and Elisa Oricchio, 15 March 2023, Nature. DOI: 10.1038/s41586-023-05794-2
|



 Your new post is loading...
Your new post is loading...


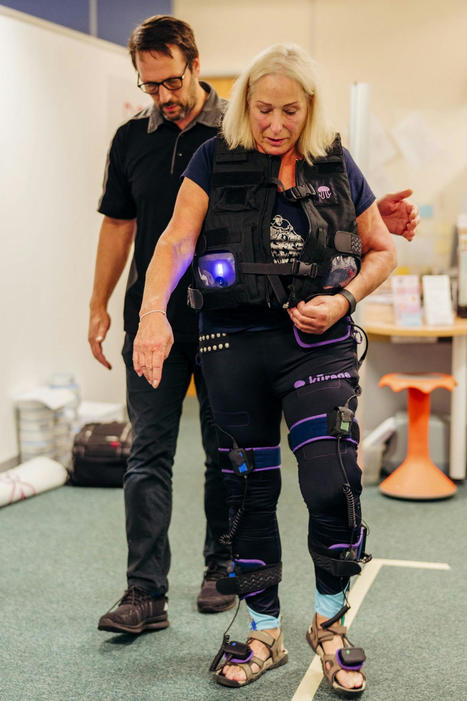
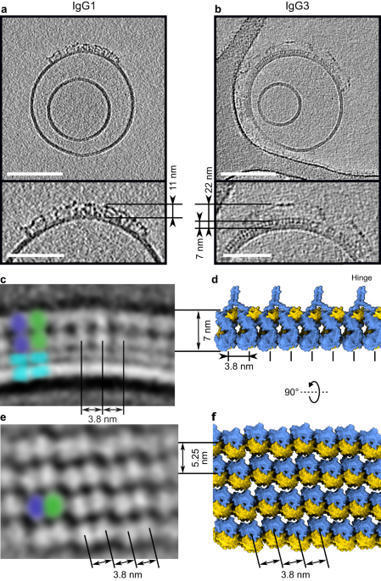


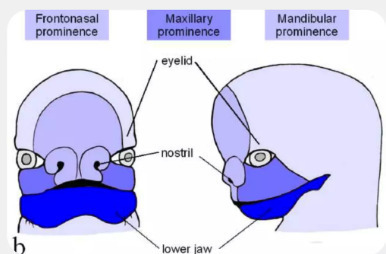

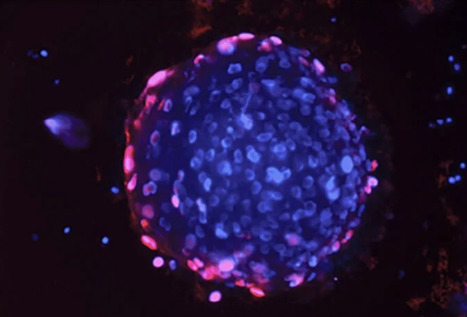

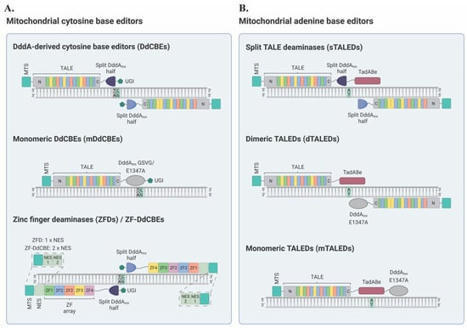

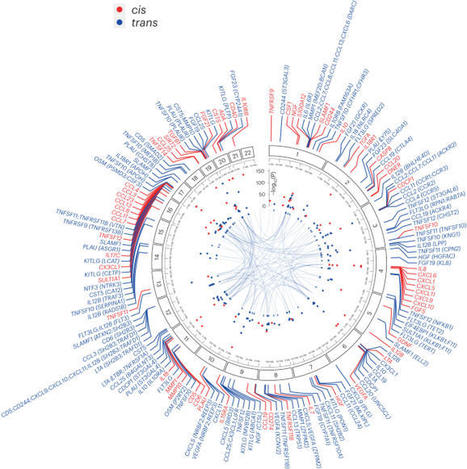
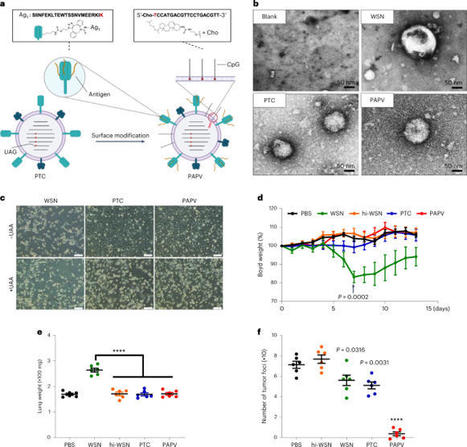






Oxycodone without a prescription
Phentermine 37.5 mg for sale
Phentremin weight loss
purchase Adderall online
Where Fentanyl Patches online
Where to buy Acxion Fentermina