Genetic mutations which cause a debilitating hereditary kidney disease affecting children and young adults have been fixed in patient-derived kidney cells using a potentially game-changing DNA repair-kit. The advance developed by University of Bristol scientists is published in Nucleic Acids Research.
 Your new post is loading... Your new post is loading...
 Your new post is loading... Your new post is loading...
We designed a unique nanocapsule for efficient single CRISPR-Cas9 capsuling, noninvasive brain delivery and tumor cell targeting, demonstrating an effective and safe strategy for glioblastoma gen
BigField GEG Tech's insight:
Many drugs cannot cross the blood-brain barrier (BBB), which also applies to most CRISPR-carrying vehicles. So Bingyang Shi made chemically functional nanocapsules to penetrate the BBB, according to a study recently published in Science Advances. He essentially packaged unique CRISPR-Cas9/sgRNA complexes in a coating consisting of a thin polymeric shell made of positively charged guanidine acrylate cross-linked with N,N-bis(acryloyl)cystamine that contains biodegradable disulfide bonds. The shell was finally decorated with the peptide angiopep-2. While coating rather than encapsulation was chosen to maintain the particle size at approximately 30 nm and thus facilitate BBB penetration, the angiopep-2 peptide served two purposes. It is a ligand that binds to low-density lipoprotein receptor-related protein-1, which is most highly expressed on BBB endothelial cells and glioblastoma (GBM) cells. This will result in the concentration of the nanocapsules around the BBB, increasing their chance of penetration into the brain, where they will then be preferentially taken up by GBMs. However, more work is needed before these nanocapsules can emerge as a therapy for GBM patients.
Estrogen and progesterone receptor (ER, PR) signaling control breast development and impinge on breast carcinogenesis. ER is an established driver of ER + disease but the role of the PR, itself an ER target gene, is debated. We assess the issue in clinically relevant settings by a genetic approach and inject ER + breast cancer cell lines and patient-derived tumor cells to the milk ducts of immunocompromised mice. Such ER + xenografts were exposed to physiologically relevant levels of 17-β-estradiol (E2) and progesterone (P4). We find that independently both premenopausal E2 and P4 levels increase tumor growth and combined treatment enhances metastatic spread. The proliferative responses are patient-specific with MYC and androgen receptor (AR) signatures determining P4 response. PR is required for tumor growth in patient samples and sufficient to drive tumor growth and metastasis in ER signaling ablated tumor cells. Our findings suggest that endocrine therapy may need to be personalized, and that abrogating PR expression can be a therapeutic option. The role of progesterone receptor (PR) and its interplay with estrogen receptor (ER) in breast cancer is controversial. Here, the authors demonstrate that PR can have an ER-independent role in breast cancer growth and metastasis and that its effects are dependent on MYC and androgen receptor signatures.
BigField GEG Tech's insight:
Proud to share one of our long-time partner's publication whom we have designed robust and efficient lentiviral vectors for its project in breast cancer.
BigField GEG Tech's insight:
In this study the scientists developed lentiviral vectors, which integrate in the host cell genome, that achieve efficient liver gene transfer in mice, dogs and non-human primates, by intravenous delivery.
Researchers utilized a CRISPR/Cas9 system to evaluate the usage of tRNA by deleting two tRNA genes from the genomes of hyper hepatocellular carcinoma and human near-haploid chronic myeloid leukemia cells.
BigField GEG Tech's insight:
In a recent study, a team of researchers used a CRISPR/Cas9 system to evaluate the use of tRNA by deleting two tRNA genes from the genomes of hyperhepatocellular carcinoma and human chronic myeloid quasi-haploid leukaemia cells. The authors discovered numerous unexpected genomic changes at the target region using an improved droplet-based target enrichment approach followed by Oxford Nanopore Technology long-read sequencing. The method used in this study demonstrates that CRISPR/Cas9 can lead to the integration of endogenous and exogenous DNA fragments and also produce local inversions, duplications and insertions of functional target-derived fragments. This research presents evidence that a combination of duplication and inversion, as well as integration of exogenous DNA fragments and clustered interchromosomal rearrangements, can occur simultaneously. Furthermore, it was shown for the first time that the target-derived fragments were nevertheless functional despite these modifications, which may complicate mechanistic explanations. These results reveal a new example of unintended CRISPR/Cas9 editing events that can go unnoticed and have a significant impact on the conclusions drawn from experimental reads.
This clinical update looks at Graphite Bio's sickle cell disease candidate GPH101. GPH101 is an ex vivo CRISPR-edited cell therapy that is anticipated to provide a permanent cure by targeting the root cause of disease. Clinical trial enrolment is ongoing at multiple sites.
BigField GEG Tech's insight:
GPH101 is an ex vivo CRISPR-edited cell therapy designed to correct the HBB point mutation and should provide a permanent cure by targeting the root cause of the disease. Correction of the HBB gene results in a decrease in sickle cell hemoglobin production and restores normal expression of adult hemoglobin, thereby restoring normally functioning red blood cells. The candidate is engineered using patient-derived hematopoietic stem cells (HSCs) that are edited with Cas9-sgRNA gene editing machines and a DNA repair model, in a mechanism that essentially cuts the mutation from the genome and replaces it with the correct sequence. Cas9-sgRNA is delivered by ribonucleoprotein (RNP) complexes while delivery of a corrected DNA template is achieved by an adeno-associated virus 6 (AAV6) vector. The FDA approved an IND application in December 2020 for GPH101 and it will be evaluated in the Phase 1/2 CEDAR trial, an open-label, multicenter study to evaluate the safety, efficacy, and pharmacodynamics of the new therapeutic candidate in adults and adolescents with severe sickle cell disease. The trial will enroll approximately 15 adult and adolescent participants at up to five clinical trial sites in the United States.
Mutations that lead to muscle atrophy can be repaired with the gene editor CRISPR-Cas9. A team led by ECRC researcher Helena Escobar has now introduced the tool into human muscle stem cells for the first time using mRNA, thus discovering a method suitable for therapeutic applications.
BigField GEG Tech's insight:
Muscular dystrophies are currently incurable. However, the mutations that lead to muscle atrophy could be repaired with the CRISPR-Cas9 gene editor. A team led by the ECRC researcher introduced the tool into human muscle stem cells for the first time using mRNA, discovering a method suitable for therapeutic applications. First, to introduce the mRNA into the stem cells, the researchers used an electroporation process, which temporarily makes cell membranes more permeable to larger molecules. In the next step, the team used a deliberately modified molecule on the surface of human muscle stem cells to show that the method can be used to correct genetic defects in a targeted manner. Finally, the team tested a tool similar to the CRISPR-Cas9 gene editor that does not cut DNA, but only modifies it in one place with extreme precision. In particular, the researchers showed that the corrected muscle stem cells are just as capable as healthy cells of fusing together and forming young muscle fibers.
The mammalian brain contains many specialized cells that develop from a thin sheet of neuroepithelial progenitor cells. Single-cell transcriptomics revealed hundreds of molecularly diverse cell types in the nervous system, but the lineage relationships between mature cell types and progenitor cells are not well understood. Here we show in vivo barcoding of early progenitors to simultaneously profile cell phenotypes and clonal relations in the mouse brain using single-cell and spatial transcriptomics. By reconstructing thousands of clones, we discovered fate-restricted progenitor cells in the mouse hippocampal neuroepithelium and show that microglia are derived from few primitive myeloid precursors that massively expand to generate widely dispersed progeny. We combined spatial transcriptomics with clonal barcoding and disentangled migration patterns of clonally related cells in densely labeled tissue sections. Our approach enables high-throughput dense reconstruction of cell phenotypes and clonal relations at the single-cell and tissue level in individual animals and provides an integrated approach for understanding tissue architecture. Ratz et al. present an easy-to-use method to barcode progenitor cells, enabling profiling of cell phenotypes and clonal relations using single-cell and spatial transcriptomics, providing an integrated approach for understanding brain architecture.
BigField GEG Tech's insight:
Proud to share one of our long-time partner publication whom we have designed very specific custom lentiviral vectors. @Micheal Ratz The mammalian brain contains many specialized cells that develop from a thin sheet of neuroepithelial progenitor cells. However, the lineage relationships between mature cell types and progenitor cells are not well understood. Therefore, researchers performed high-throughput clonal tracking and expression profiling of mouse forebrain cells using single-cell and spatial transcriptomics and found two populations of fate-restricted progenitor cells present as early as embryonic day (E) 9.5 in the murine hippocampus. They found that microglia are generated from a limited number of progenitor cells that undergo massive clonal expansion as well as widespread migration throughout the mouse telencephalon. The results thus demonstrate the utility of high-throughput clonal tracing in the mouse brain to provide molecular information about brain development at the single cell and tissue level. TREX is the technique that allowed them to profile TRacking and gene EXpression of clonally bound cells in the mouse brain by high-throughput single-cell RNA sequencing (scRNA-seq). TREX is based on a diverse library of lentiviruses containing barcodes downstream of a GFP protein.
More than 60% of patients with relapsed or refractory B-cell acute lymphoblastic leukemia achieved initial complete remission after receiving an investigational chimeric antigen receptor T-cell therapy, phase 1/phase 2 study results showed.By their initial 1-month evaluation, 73% of patients who received the highest dose levels had a complete response to therapy, according to findings presented
BigField GEG Tech's insight:
CARCIK-CD19, an investigational therapy developed by scientists in Italy, involves donor-derived CD19-targeted CAR T cells differentiated according to the cytokine-induced killer (CIK) cell protocol that uses non-viral Sleeping Beauty transposon gene transfer to prevent graft-versus-host disease. More than 60% of patients with relapsed or refractory B-cell acute lymphoblastic leukemia achieved initial complete remission after receiving experimental chimeric antigen receptor T-cell therapy, results from the phase 1/phase 2 study showed. At their initial one-month assessment, 73% of patients who received the highest doses had a complete response to treatment, according to results presented at the 4th European Society for Blood and Marrow Transplantation-European Hematology Association CAR-T Cell Meeting.
Existing approaches to therapeutic gene transfer are marred by the transient nature of gene expression following non-integrative gene delivery and by …
BigField GEG Tech's insight:
The efficacy, persistence and predictability of many future gene and cell therapies to treat diseases such as cancer, rare genetic diseases could be enhanced by what are called "genomic safe harbors" (GSHs). These are landing sites in the human genome that can safely accommodate new therapeutic genes without causing other unintended changes in a cell's genome that could pose a risk to patients. To date, only a few candidate GSHs have been explored, and they all come with some caveats. However, a collaboration of researchers from the Wyss Institute for Biologically Inspired Engineering at Harvard, Harvard Medical School (HMS) and ETH Zurich in Switzerland, has developed a computational approach to identify GSH sites with significantly higher potential for safe insertion of therapeutic genes and their sustained expression in multiple cell types. For two of the 2,000 predicted GSH sites, the team provided extensive validation with adoptive T cell therapies and in vivo gene therapies for skin diseases in mind. By modifying the identified GSH sites to carry a reporter gene in T cells and a therapeutic gene in skin cells, respectively, using CRISPR-Cas9, they demonstrated safe and sustained expression of the newly introduced genes. The study is published in Methods of Cell Reporting.
Researchers at Texas Biomedical Research Institute in San Antonio, Texas have developed a novel strategy to inhibit human immunodeficiency virus (HIV) replication, using guide RNAs that target highly conserved regions of HIV-1 in conjunction with the CRISPR-Cas13d nuclease system.
BigField GEG Tech's insight:
A recent publication in Viruses detailed a CRISPR-based strategy to effectively target and control HIV-1 infections in vitro with CasRx. Dr. Smita Kulkarni's laboratory at the Texas Biomedical Research Institute successfully combined a novel set of guide RNAs (gRNAs), known as polyHIV, with CasRx to target and inhibit HIV-1 replication. This work demonstrates for the first time the potential of the CRISPR-Cas13d nuclease system to target acute and latent HIV infections that could potentially be used to improve the effectiveness of current HIV treatment options. Kulkarni's team strategically selected four sites in the HIV genome that are more than 70% conserved from the HIV-1 transcript sequences deposited in the LANL database and specific gRNAs were designed against these sites. The four gRNAs were selected from 20 candidates to form a poly-gRNA assembly (polyHIV) and paired with CasRx, a Cas protein derived from the Cas13d family with high RNA cleavage activity. The selected target sites were non-overlapping and located in the gag, pol, protease (prot), integrase (int), cPPT and central termination sequence (CTS) coding regions in the HIV-1 genome. The study is the first to provide a single vector with four gRNAs targeting non-overlapping conserved regions of HIV-1.
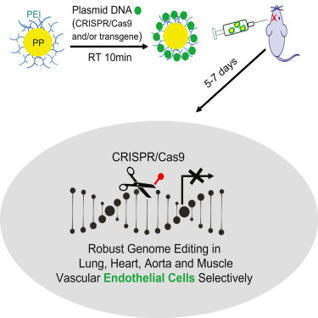
The ability to induce genome editing of the vascular endothelium has been challenging.
BigField GEG Tech's insight:
According to the journal Cell Reports, the lab of Youyang Zhao, PhD, of the Stanley Manne Children's Research Institute at Ann & Robert H. Lurie Children's Hospital in Chicago has developed a unique nanoparticle to deliver genome-editing technology such as CRISPR/Cas9 to endothelial cells. This is the first time that vascular endothelial cells can be reached for genome editing, as the usual way of delivering CRISPR/Cas9, via a virus, does not work for this cell type. With this nanoparticle, researchers can introduce genes to inhibit vascular damage and/or promote vascular repair, correct genetic mutations, and turn genes on or off to restore normal function. This technology also allows them to modify multiple genes at the same time. This is an important advance for the treatment of any disease caused by endothelial dysfunction.
Sonodynamic therapy uses ultrasound in combination with drugs to release harmful reactive oxygen species (ROS) at the site of a tumor.
BigField GEG Tech's insight:
Hepatocellular carcinoma, the most common form of liver cancer, and surgical treatment by removing part of the liver or transplanting a healthy liver is not suitable for patients with more advanced disease. That's why researchers have developed sonodynamic therapy (SDT), which uses ultrasound in combination with drugs to release harmful reactive oxygen species (ROS) at the site of a tumor. Because ultrasound can penetrate deep into tissue, SDT may be an effective, non-invasive way to treat hepatocellular carcinoma. However, the treatment is not very effective because cancer cells can quickly overcome the therapy by activating a gene called nuclear factor erythroid 2-related factor 2 (NFE2L2), which deploys cell detoxification and antioxidant enzymatic defenses.The researchers therefore used and encapsulated the CRISPR/Cas9 system and an ROS precursor molecule in lipid nanoparticles.Then, they treated hepatocellular carcinoma cells in a Petri dish with the nanoparticles. The lipid nanoparticles were taken up by the lysosomes of the cells. Ultrasound treatment caused the formation of ROS, which disrupted the lysosomes and allowed the CRISPR/Cas9 system to enter the nucleus and inhibit NFE2L2 gene expression. The ROS also damaged other cellular components. As a result, many more cancer cells died from SDT than without NFE2L2 gene editing. Next, the team injected the nanoparticle treatment into mice with implanted human hepatocellular carcinoma tumors. After 15 days of combined nanoparticle and ultrasound treatment, all of the mice's tumors disappeared and did not return. |
More than 70% of human breast cancers (BCs) are estrogen receptor α-positive (ER+). A clinical challenge of ER+ BC is that they can recur decades after initial treatments. Mechanisms governing latent disease remain elusive due to lack of adequate in vivo models. We compare intraductal xenografts of ER+ and triple-negative (TN) BC cells and demonstrate that disseminated TNBC cells proliferate similarly as TNBC cells at the primary site whereas disseminated ER+ BC cells proliferate slower, they decrease CDH1 and increase ZEB1,2 expressions, and exhibit characteristics of epithelial-mesenchymal plasticity (EMP) and dormancy. Forced E-cadherin expression overcomes ER+ BC dormancy. Cytokine signalings are enriched in more active versus inactive disseminated tumour cells, suggesting microenvironmental triggers for awakening. We conclude that intraductal xenografts model ER + BC dormancy and reveal that EMP is essential for the generation of a dormant cell state and that targeting exit from EMP has therapeutic potential. The study of tumour dormancy is limited by suitable in vivo models. Here the authors show that mammary intraductal breast cancer (BC) xenografts model estrogen receptor α-positive (ER+) BC dormancy and rapid metastatic progression characteristic of triple-negative (TN) BC. The dormant disseminated ER+ BC cells display characteristics of epithelial-mesenchymal plasticity and forced expression of E-cadherin allows them to overcome dormancy.
BigField GEG Tech's insight:
Once again, proud to share a high-quality publication of the Brisken Lab at the EPFL, one of our long-standing partners. Two publications in Nature in two months, Wow ! As you said Cathrin, “tons more to do together” ! I take the opportunity to introduce the next workshop organized by Cathrin Brisken about the Preclinical and Personalized Breast Cancer Research.
https://www.epfl.ch/labs/brisken-lab/preclinicalmodelcourse/
On August 18, 2021, the American Society of Gene and Cell Therapy (ASGCT) hosted a virtual roundtable on adeno-associated virus (AAV) integration, fea…
BigField GEG Tech's insight:
The purpose of this white paper is to review the evidence of rAAV-related host genome integration in animal models and possible risks of insertional mutagenesis in patients. In addition, technical considerations, regulatory guidance and bioethics are discussed.
Unwanted immune responses threaten to derail some gene therapies. But researchers are seeking ways to combat harmful inflammation.
BigField GEG Tech's insight:
It is a pity that non integrating lentiviral vectors are unknown or underused. This is not THE solution but in several cases, I think their use would be more relevant than AAVs use in term of immune response. The advancements of gene therapy will go through the design and development of complementary vector platforms.
Wake Forest Institute for Regenerative Medicine (WFIRM) scientists working on CRISPR/Cas9-mediated gene editing technology have developed a method to increase efficiency of editing while minimizing DNA deletion sizes, a key step toward developing gene editing therapies to treat genetic diseases.
BigField GEG Tech's insight:
Although CRISPR/Cas9 mainly generates short insertions or deletions at the target site, it can also make large deletions of DNA around the specific target site. These large deletions pose safety concerns and can reduce the efficiency of functional editing. The WFIRM team is looking at ways to reduce the risk of this happening. The research described in their recent paper, published recently in Nucleic Acids Research, aimed to combat the generation of unpredictable long DNA deletions on target and find a way to prevent this, a key step towards the development of gene editing therapies to treat genetic diseases. The team evaluated a variety of human cells and genes of interest and found that fusing DNA polymerase I or the Klenow fragment to the Cas9 enzyme minimised large, unanticipated deletions of genomic DNA without sacrificing the efficiency of genome editing. On the contrary, it even increased editing efficiency in primary human cells.
Most pharmaceuticals don't reach the brain, leaving cognitive impairments due to diseases like mucopolysaccharidoses (MPS) hard to treat. Now, a Brazilian research team has developed a method for nasal delivery of CRISPR reagents to the brain that improves MPS cognitive symptoms in mice. Experiments will proceed with monkeys later this year.
BigField GEG Tech's insight:
Mucopolysaccharidosis I is caused by mutations in the gene for the enzyme alpha-L-iduronidase (IDUA). This enzyme is required for the breakdown of substances called glycosaminoglycans (GAGs) that are by-products of chemical reactions in the body's cells. In 2018, a team of researchers was able to demonstrate the feasibility of CRISPR gene editing in vivo in mice via intravenous injection. The treatment partially restored IDUA enzyme activity in organs such as the heart, lungs, liver and kidneys, but not in the brain because the blood-brain barrier protects it. However, this is a problem for patients with the severe form of the disease who may develop cognitive decline. The researchers therefore had the idea of going through the nose to reach the olfactory bulb just above. As a result of the experiments, it was finally shown that non-invasive administration of CRISPR reagents via nasal administration allows gene editing and partial restoration of IDUA activity in the brain and improved cognitive function, while biomarkers of the disease in other organs also improved.
Chimaeric antigen receptor (CAR) T cells can generate durable clinical responses in B-cell haematologic malignancies. The manufacturing of these T cells typically involves their activation, followed by viral transduction and expansion ex vivo for at least 6 days. However, the activation and expansion of CAR T cells leads to their progressive differentiation and the associated loss of anti-leukaemic activity. Here we show that functional CAR T cells can be generated within 24 hours from T cells derived from peripheral blood without the need for T-cell activation or ex vivo expansion, and that the efficiency of viral transduction in this process is substantially influenced by the formulation of the medium and the surface area-to-volume ratio of the culture vessel. In mouse xenograft models of human leukaemias, the rapidly generated non-activated CAR T cells exhibited higher anti-leukaemic in vivo activity per cell than the corresponding activated CAR T cells produced using the standard protocol. The rapid manufacturing of CAR T cells may reduce production costs and broaden their applicability. Potent chimaeric antigen receptor T cells can be generated within one day from T cells derived from peripheral blood without the need for T-cell activation.
BigField GEG Tech's insight:
In the process of engineering CAR T cells, if the patient's T cells are out of the body for too long, they can lose their ability to replicate, which is critical to their effectiveness as a live drug. Thus, a research team has developed a new approach that reduces the time needed to modify patients' immune cells so that they can be quickly reinjected into the body. The manufacturing process for CAR T cells typically takes between nine and 14 days, whereas this new process would reduce the manufacturing time to as little as 24 hours. Traditional manufacturing approaches require that T cells be stimulated in a way that encourages the cells to replicate and grow in number. The key to the Penn researchers' manufacturing approach is the lentiviral vector that delivers the CAR gene to T cells. Lentiviral vectors are able to transfer genes like the CAR gene to cells without needing this initial "activation" step. This approach has the dual advantage of speeding up the overall manufacturing process while maintaining T-cell potency. The team hopes that the reduced manufacturing time could make the therapy more cost-effective and accessible to more patients.
Intellia Therapeutics recently announced that the first patient had been dosed in its Phase 1/2a CRISPR trial for acute myleoid leukaemia (AML). The new therapeutic candidate, NTLA-5001, is a CRISPR-edited T cell receptor therapy designed to targeted Wilm's Tumour (WT1) antigen, which is found on AML and several other blood cancers as wel
BigField GEG Tech's insight:
Intellia Therapeutics recently announced that the first patient has received a dose in its Phase 1/2a CRISPR trial for acute myeloid leukemia (AML). The new therapeutic candidate, NTLA-5001, is a CRISPR-edited T-cell receptor therapy designed to target Wilm's tumor antigen (WT1), which is found on AML and several other blood cancers as well as certain types of solid cancer. To generate NTLA-5001, T cells are isolated from a patient with AML and the T cell receptor (TCR) is replaced with a naturally occurring high avidity TCR (i.e. from a healthy donor) with specificity for WT1. Complete elimination of the endogenous TCR is achieved by CRISPR-Cas9-mediated replacement of the TRAC locus with the WT1 TCR, and subsequent site-specific inactivation of the TRBC locus, which encodes the T cell receptor beta chains. For TCR locus disruption, SpCas9 and gRNAs are delivered to cells via lipid nanoparticles. The WT1 TCR is delivered to cells via adeno-associated virus (AAV) transduction. NTLA-5001 is therefore being evaluated in a multi-center, single-dose Phase 1/2a study that will assess its safety, tolerability, cellular kinetics (CK), activity and pharmacodynamics (PD) in AML participants.
Off-target genome editing in the liver can be reduced by using lipid nanoparticles to deliver oligonucleotides that disrupt the secondary structure of single-guide RNAs as well as short interfering RNAs targeting Cas9 mRNA.
BigField GEG Tech's insight:
In genome editing, it is crucial to ensure target-specific editing in diseased cells and to avoid altering the genome of spectator cells. Since a substantial fraction, i.e. 30-90%, of an intravenously injected dose of lipid nanoparticles (LPNs) accumulates in the liver, off-target editing in this organ must be minimized. According to a study published in Nature Biomedical Engineering , LNPs can be used to deliver two types of anti-CRISPR nucleic acid to the liver to disrupt genome editing in hepatocytes, thereby improving the specificity of CRISPR-Cas genome editing in the extrahepatic tissues and organs. To determine whether anti-CRISPR oligonucleotides could be used to reduce off-target editing in the liver, researchers first treated mice stably expressing Cas9 tagged with GFP with anti-CRISPR or scrambled oligonucleotides formulated in NLPs that preferentially target the liver. Two hours later, they treated the same mice with the same NPLs, but instead encapsulated gRNA targeting the GFP locus. Compared to scrambled oligonucleotides, the anti-CRISPR ones reduced the frequency of deletion mutations in hepatocytes by more than twofold. Therefore, Cas9-mediated on-target genome editing in extrahepatic tissues could be maintained while limiting off-target editing in the liver.
Hemonc Today | Although therapeutic advances during the past decade have dramatically improved cancer outcomes overall, the outlook for patients with malignant brain tumors has remained poor.The 5-year relative survival rate for these patients increased only from 23% between 1975 and 1977 to 36% between 2009 and 2015, with rates in the single digits for those with particularly lethal brain tumor types,
BigField GEG Tech's insight:
The challenge in treating brain tumors has been the inability of drugs to penetrate the blood-brain barrier. In addition, the use of treatments that can cross the barrier, such as chemotherapy and radiation therapy, is often limited due to their toxicity. Other standard treatments, such as surgery, are not even an option for some patients because of the location of the tumor. Among brain tumors, low-grade pediatric ones have specific mutations that can be effectively targeted with a single-pathway drug. However, this is not the case for high-grade pediatric brain tumors. Therefore, Vitanza and colleagues are conducting three trials at Seattle Children's, including pediatric patients with predominantly lethal disease that has progressed after initial standard treatment. BrainChild-01 is examining the use of locoregionally administered HER2-directed CAR T cells; BrainChild-02 is using EGFR-specific CAR T cells to treat children and young adults with relapsed or refractory EGFR-positive central nervous system tumors; and BrainChild-03 is using B7-H3-targeted CAR T cells for all patients with recurrent CNS tumors and those with the deadly brain tumor diffuse intrinsic glioma.
The adoptive transfer of T lymphocytes reprogrammed to target tumour cells has demonstrated potential for treatment of various cancers1–7. However, little is known about the long-term potential and clonal stability of the infused cells. Here we studied long-lasting CD19-redirected chimeric antigen receptor (CAR) T cells in two patients with chronic lymphocytic leukaemia1–4 who achieved a complete remission in 2010. CAR T cells remained detectable more than ten years after infusion, with sustained remission in both patients. Notably, a highly activated CD4+ population emerged in both patients, dominating the CAR T cell population at the later time points. This transition was reflected in the stabilization of the clonal make-up of CAR T cells with a repertoire dominated by a small number of clones. Single-cell profiling demonstrated that these long-persisting CD4+ CAR T cells exhibited cytotoxic characteristics along with ongoing functional activation and proliferation. In addition, longitudinal profiling revealed a population of gamma delta CAR T cells that prominently expanded in one patient concomitant with CD8+ CAR T cells during the initial response phase. Our identification and characterization of these unexpected CAR T cell populations provide novel insight into the CAR T cell characteristics associated with anti-cancer response and long-term remission in leukaemia. Infusion of CD19-directed chimeric antigen receptor T cells into two patients with chronic lymphocytic leukaemia resulted in complete tumour remission and persistence of the infused cells more than ten years later.
BigField GEG Tech's insight:
Good News ?
While multiple technologies for small allele genome editing exist, robust technologies for targeted integration of large DNA fragments in mammalian genomes are still missing. Here we develop a gene delivery tool (FiCAT) combining the precision of a CRISPR-Cas9 (find module), and the payload transfer efficiency of an engineered piggyBac transposase (cut-and-transfer module). FiCAT combines the functionality of Cas9 DNA scanning and targeting DNA, with piggyBac donor DNA processing and transfer capacity. PiggyBac functional domains are engineered providing increased on-target integration while reducing off-target events. We demonstrate efficient delivery and programmable insertion of small and large payloads in cellulo (human (Hek293T, K-562) and mouse (C2C12)) and in vivo in mouse liver. Finally, we evolve more efficient versions of FiCAT by generating a targeted diversity of 394,000 variants and undergoing 4 rounds of evolution. In this work, we develop a precise and efficient targeted insertion of multi kilobase DNA fragments in mammalian genomes. Mammalian genome engineering has advanced tremendously over the last decade, however there is still a need for robust gene writing with size scaling capacity. Here the authors present Find Cut-and-Transfer (FiCAT) technology to delivery large targeted payload insertion in cell lines and in vivo in mouse models.
BigField GEG Tech's insight:
Gene editing developed for clinical applications has limited potential. It does not provide the large DNA fragments needed to restore gene function in diseases like Duchenne muscular dystrophy. According to Nature Communications, a research team led by Professor Marc Güell has revealed a new gene-writing tool called "FiCAT," to find, cut and transfer, that has been developed to repair large defects in genomic DNA. "FiCAT" is composed of CRISPR-Cas and the piggyBac transposase. The team's challenge was to make them work together. The team reported that "FiCAT" resulted in twice the delivery efficiency compared to methods involving HDR or enhanced HITI to create targeted DNA insertions, achieving an insertion of up to 8 kb with high precision. After some adjustments and optimizations, the on-target insertion efficiency of "FiCAT" could be pushed up to 22% of cultured cells, with early experiments suggesting that off-target risks are manageable. One advantage of “FiCAT” is that circular DNA templates can be used, unlike the linear DNA fragments required by HDR or HITI, which are more likely to be associated with increased genome toxicity and uncontrolled insertion risks. In addition, studies show that "FiCAT" can also be used in vivo, which opens the way for future clinical trials.
Adeno-associated virus (AAV) vectors are important delivery platforms for therapeutic genome editing but are severely constrained by cargo limits. Simultaneous delivery of multiple vectors can limit dose and efficacy and increase safety risks. Here, we describe single-vector, ~4.8-kb AAV platforms that express Nme2Cas9 and either two sgRNAs for segmental deletions, or a single sgRNA with a homology-directed repair (HDR) template. We also use anti-CRISPR proteins to enable production of vectors that self-inactivate via Nme2Cas9 cleavage. We further introduce a nanopore-based sequencing platform that is designed to profile rAAV genomes and serves as a quality control measure for vector homogeneity. We demonstrate that these platforms can effectively treat two disease models [type I hereditary tyrosinemia (HT-I) and mucopolysaccharidosis type I (MPS-I)] in mice by HDR-based correction of the disease allele. These results will enable the engineering of single-vector AAVs that can achieve diverse therapeutic genome editing outcomes. Long-term expression of Cas9 following precision genome editing in vivo may lead to undesirable consequences. Here we show that a single-vector, self-inactivating AAV system containing Cas9 nuclease, guide, and DNA donor can use homology-directed repair to correct disease mutations in vivo.
BigField GEG Tech's insight:
Most studies using AAVs to deliver gene therapy attempt to insert a Cas9 nuclease and a single guide RNA (sgRNA), along with their respective promoters, into the vector to create a single double-stranded break in the DNA. Given the 4.7 kb packaging limit of most AAVs, this is typically a significant challenge, especially for studies aimed at correcting genetic mutations by providing a donor DNA template for HDR. In this study, members of Professor Erik Sontheimer's lab at the University of Massachusetts Chan Medical School demonstrated the use of a cleverly designed, self-inactivating AAV vector with a compact Cas9 variant for therapeutic CRISPR genome editing in vivo. The study takes this packaging problem a step further, with the goal of creating a vector with enough cargo space to hold two sgRNAs rather than just one. In doing so, CRISPR could be used to excise pathogenic trinucleotide repeat expansions, such as that which causes Friedreich's ataxia. With the compact size of Staphylococcus aureus Cas9 (Nme1Cas9), this makes it easier to package into AAVs. It has a dinucleotide PAM sequence, which means it has a wider range of genomic targets and it displays high editing efficiencies in mammalian cells with low off-target activity, making it ideal for therapeutic applications. |


















Podocin is a protein normally located on the surface of specialized kidney cells and is essential for kidney function. The defective podocin, however, gets stuck inside the cell and never reaches the surface, permanently damaging the podocytes, a common genetic cause of inherited steroid-resistant nephrotic syndrome (SRNS). Gene therapy that repairs the genetic mutations that cause defective podocin offers hope to patients. However, current delivery systems that include lentiviruses, adenoviruses and adeno-associated viruses all share the same limitation in that they are limited in the space inside their viral shells and this in turn limits the amount of cargo they can deliver. Applying synthetic biology techniques, the researchers therefore redesigned the baculovirus, a human-friendly insect virus that is no longer limited by cargo capacity. By creating a DNA repair kit, including the CRISPR/Cas 9 system and DNA sequences to replace the defective gene. The team delivered a single modified baculovirus with a healthy copy of the podocin gene together with the CRISPR/Cas machines to insert it with base-pair precision into the genome and this was able to reverse the pathogenic phenotype and restore podocin to the cell surface.