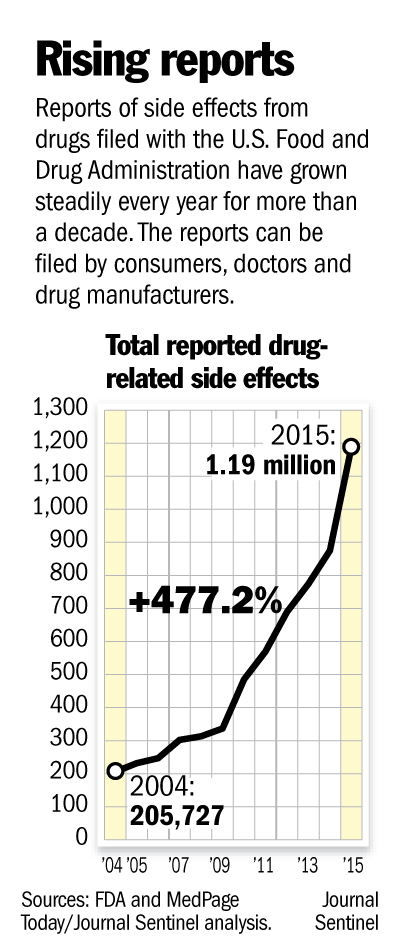
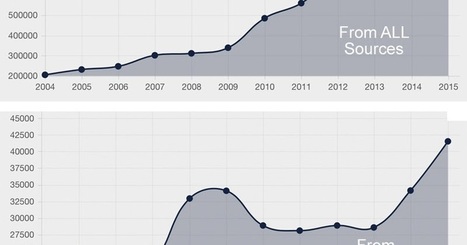
If you had a bad reaction after taking a medicine, there’s now an easy way to find out whether others experienced the same.
The U.S. Food and Drug Administration improved its online database of reports about side effects filed by patients and doctors, making it easily searchable by product, patient age, type of side effect or year it occurred.
It means that, for the first time, the public will have access to data it can make sense of. Previously, inquiring minds needed to know a little about coding to figure out the FDA’s adverse-reaction database. And even then, they had to file a Freedom of Information request with the government to get the actual reports and check the information’s accuracy.
The FDA said it hopes that the increased transparency will spur patients and doctors to submit more detailed and complete reports. Many patients don’t even know they can submit adverse-event reports to the agency.
Further Reading:
- "Institute for Safe Medicines Practices Calls for a Full-scale Modernization of FDA’s Adverse Event Reporting System"; http://sco.lt/7AC2N7
- “So Far, FDA’s Sentinel Drug Safety Monitoring Initiative Has Not Delivered as Hoped”; http://sco.lt/7P4T3Z
- “OpEd: FDA's Antiquated Drug Safety Program is ‘Obscene’"; http://sco.lt/7O6qVV
- “Another Aspect of Patient Power: Making It Easier to Report Adverse Events!”; http://sco.lt/6bgAZl
- “More Than 1 Million Adverse Event Reports Were Sent to FDA in 2015 – Only the Tip of the Iceberg”; http://sco.lt/6gfruL
- “Deregulation and Budget Cuts Likely to Lead to Increased Adverse Event Reporting”; http://sco.lt/5ZuET3
- “FDA's Dependence on User Fees & ‘Institutional Corruption’ Blamed for Dramatic Increase in Drug Adverse Events and Deaths”; http://sco.lt/8pXZD7
- “#FDA Blames Computer Glitch for Hundreds of Errors Found in Essure Adverse Event Reports”; http://sco.lt/6aCd0b
Via Pharma Guy



 Your new post is loading...
Your new post is loading...