Your new post is loading...
 Your new post is loading...
A drug approved in Europe to treat osteoporosis has now been shown to stop the growth of breast cancer cells, even in cancers that have become resistant to current targeted therapies, according to a Duke Cancer Institute study. The findings, presented June 15, 2013, at the annual Endocrine Society meeting in San Francisco, indicate that the drug bazedoxifene packs a powerful one-two punch that not only prevents estrogen from fueling breast cancer cell growth, but also flags the estrogen receptor for destruction.
GE Healthcare recently announced that Clarient Diagnostic Services, Inc., a GE Healthcare company, will begin offering a next-generation sequencing assay focused on solid tumor targets for use in clinical trials. This assay will empower researchers to perform prospective and retrospective analysis to better understand which patients will respond to particular therapies, to help stratify patient populations for ongoing clinical trials, and to aid early research efforts.
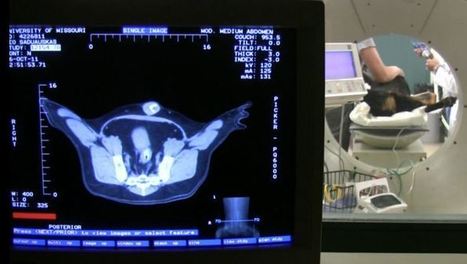
Shortly after the discovery of the neutron in 1932, some scientists recognized the potential of boron neutron capture therapy (BNCT) as a cancer treatment. But despite decades of research, the problem of finding a delivery agent that would more effectively target the tumor without harming surrounding tissue persisted. Researchers at the University of Missouri (MU) may finally have found a solution. BNCT traditionally involves injecting tumors with the non-radioactive boron-10 isotope capture agent that is then radiated with a beam of epithermal neutrons that interact with the capture agent to produce a biologically destructive nuclear caption reaction. This results in the formation of boron 11 with the release of lethal radiation in the form of alpha particles (helium-4) and lithium ions that kill the tumor. Although numerous clinical studies have demonstrated the safety of BNCT, the challenge has been finding more tumor-selective boron delivery agents.
ORLANDO—CT texture analysis of primary tumors may be a potential imaging biomarker in localized esophageal cancer following neoadjuvant chemotherapy, according to research being presented at the 2013 Cancer ...
Via Brian Shields
A degradable nanoscale shell to carry proteins to cancer cells and stunt the growth of tumors without damaging healthy cells has been developed by a team led by researchers from the UCLA Henry Samueli School of Engineering and Applied Science.
Tiny shells (about 100 nanometers in length, roughly half the size of the smallest bacterium) are composed of a water-soluble polymer that safely delivers a protein complex to the nucleus of cancer cells to induce their death. The shells degrade harmlessly in non-cancerous cells.
The process does not present the risk of genetic mutation posed by gene therapies for cancer, or the risk to healthy cells caused by chemotherapy, which does not effectively discriminate between healthy and cancerous cells, said Yi Tang, a professor of chemical and biomolecular engineering and a member of the California NanoSystems Institute at UCLA.
Biologists at Tufts University School of Arts and Sciences have discovered a bioelectric signal that can identify cells that are likely to develop into tumors. The researchers also found that they could lower the incidence of cancerous cells by manipulating the electrical charge across cells' membranes.
Seattle’s Presage Biosciences has developed a device which introduces small amounts of different chemotherapy drugs into a patient's tumor. The tumor is inspected after removal and the most effective of the drugs are used for post-surgical chemotherapy, resulting in more efficient, personalized cancer treatments. The new device is awaiting FDA approval, but is currently being used to facilitate development of new chemotherapy drugs.
One of the largest challenges faced by oncologists is finding an effective treatment for a particular patient that doesn’t cause the patient undue suffering.
The up regulation of gut mucosal cytokines such as tumor necrosis factor (TNF)-α and oxidative stress have been related to inflammatory bowel diseases (IBD) such as ulcerative colitis (UC) and Crohn's disease (CD). This study investigated an immune-mediated model of colitis. TNF-α injected intraperitonally to mice induced a dose-dependent recruitment of neutrophils into abdominal mesentery. The leukocytes influx induced by TNF-α (10 μg kg(-1) body weight) increased by 3 fold liver and colon damage scores. TNF-α-colitis was characterized by hemorrhagic edemas and crypt abscesses massively infiltrated by inflammatory cells, namely neutrophils. Moreover, TNF-α-toxicity resulted in liver steatosis and foci of necrosis infiltrated by Kupffer cells and neutrophils in parenchyma and around the centrilobular veins. The involvement of oxidative stress was evaluated using aminoguanidine (AG) as selective inhibitor of inducible NO synthase (iNOS) and curcumin (Cur), the polyphenolic antioxidant of turmeric (Curcuma longa L.). TNF-α-toxicity led to significant increase in myeloperoxidase (MPO, an index of neutrophils infiltration), nitrites (stable nitric oxide metabolites) and malondialdehyde (MDA, a marker of lipid peroxides) levels and cell apoptosis in liver and colon. AG and Cur treatments significantly attenuated the hallmarks of oxidative stress, neutrophils influx and ROS-related cellular and histological damages, in TNF-α-treated mice. Taken together, our results provide insights into the role of phagocytes-derived oxidants in TNF-α-colitis in mice. Cur and AG, by inhibiting neutrophils priming and iNOsynthase could be effective against oxidative bowel damages induced in IBD by imbalanced gut immune response. REFERENCE: Int Immunopharmacol. 2012 Jan;12(1):302-11. Epub 2011 Oct 29. Aminoguanidine and curcumin attenuated tumor necrosis factor (TNF)-α-induced oxidative stress, colitis and hepatotoxicity in mice. Mouzaoui S, Rahim I, Djerdjouri B.
A common mutation in a tumor-suppressing gene could explain why blacks are more likely to get early breast and colon cancer, researchers at Georgia Health Sciences University said.The ability to more quickly and cheaply ...
Via Brian Shields
The Salk findings may help scientists develop small molecules — the basis for the vast majority of current drugs — capable of destroying tumors by binding and disrupting large and complex cellular components that allow cancer cells to grow and spread. Understanding how viruses overcome healthy cells may also help scientists engineer tumor-busting viruses, which offer a new and potentially self-perpetuating cancer therapy.
Animal studies have been conducted successfully using gold nanoparticles to target prostate cancer tumors. Next step-- human trials... Current treatments for prostate cancer are not effective in patients who have aggressive prostate cancer tumors. Most of the time, prostate cancers are slow-growing; the disease remains localized and it is easily managed. However, aggressive forms of the disease spread to other parts of the body, and is the second-leading cause of cancer deaths in U.S. men. The MU scientists believe their treatment will be able to shrink aggressive tumors or eliminate them completely. Axiak-Bechtel says this treatment can be safe and effective in dogs as well as humans because dogs are the only other mammal to naturally contract the aggressive form of prostate cancer.
A heavily publicized study came out last week indicating that the world’s best-selling weedkiller and genetically modified corn can cause tumors, multiple organ damage, and lead to premature death. "This research shows an extraordinary number of tumours developing earlier and more aggressively - particularly in female animals. I am shocked by the extreme negative health impacts,” said Dr Michael Antoniou, molecular biologist at King’s College London, and a member of CRIIGEN, the independent scientific council which supported the research. Further Reading: This 2009 study linked GM corn to liver and organ toxicity: http://www.biolsci.org/v05p0706.htm;
MIT researchers have developed RNA-delivering nanoparticles that allow for rapid screening of new drug targets in mice. By sequencing cancer-cell genomes, scientists have discovered vast numbers of genes that are mutated, deleted or copied in cancer cells. This treasure trove is a boon for researchers seeking new drug targets, but it is nearly impossible to test them all in a timely fashion.
|
Diagnosed with advanced lung cancer over a year ago, Gabe Tartaglia was loath to undergo the kind of harsh chemotherapy that had devastated his sister before her death three years earlier from pancreatic cancer. He decided to enter a clinical trial for a new drug designed to trigger the immune system to fight cancer. The results were better than anyone expected.
What causes a cell to metastasize into a cancerous tumor? To find out, Corey Neu, an assistant professor in Purdue University‘s Weldon School of Biomedical Engineering, and colleagues have combined an atomic force microscope (AFM) and a nuclear magnetic resonance system. An AFM uses a tiny vibrating probe called a cantilever with a tip that travels over the surface of a cell to yield information about materials and surfaces at the scale of nanometers, or billionths of a meter.
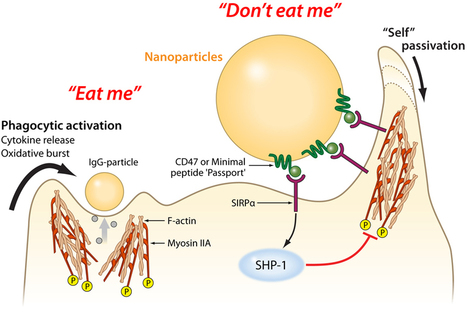
Macrophages — literally, "big eaters" — are a main part of the body's innate immune system . These cells find and engulf invaders, like bacteria, viruses, splinters and dirt. Unfortunately, macrophages also eat helpful foreigners, including nanoparticles that deliver drugs or help image tumors.
Along with members of his lab, Dennis Discher, professor of chemical and biomolecular engineering in the School of Engineering and Applied Science, has developed a "passport" that could be attached to therapeutic particles and devices, tricking macrophages into leaving them alone.
Taking a cue from a membrane protein that the body's own cells use to tell macrophages not to eat them, the researchers engineered a the simplest functional version of that protein and attached it to plastic nanoparticles. These passport-carrying nanoparticles remained in circulation significantly longer than ones without the peptide, when tested in a mouse model.
In 2008, Discher’s group showed that the human protein CD47, found on almost all mammalian cell membranes, binds to a macrophage receptor known as SIRPa in humans. Like a patrolling border guard inspecting a passport, if a macrophage’s SIRPa binds to a cell’s CD47, it tells the macrophage that the cell isn’t an invader and should be allowed to proceed on. “There may be other molecules that help quell the macrophage response,” Discher said. “But human CD47 is clearly one that says, ‘Don’t eat me’.” Since the publication of that study, other researchers determined the combined structure of CD47 and SIRPa together. Using this information, Discher’s group was able to computationally design the smallest sequence of amino acids that would act like CD47. This “minimal peptide” would have to fold and fit well enough into the receptor of SIRPa to serve as a valid passport. After chemically synthesizing this minimal peptide, Discher’s team attached it to conventional nanoparticles that could be used in a variety of experiments. “Now, anyone can make the peptide and put it on whatever they want,” Rodriguez said. The research team’s experiments used a mouse model to demonstrate better imaging of tumors and as well as improved efficacy of an anti-cancer drug-delivery particle. As this minimal peptide might one day be attached to a wide range of drug-delivery vehicles, the researchers also attached antibodies of the type that could be used in targeting cancer cells or other kinds of diseased tissue. Beyond a proof of concept for therapeutics, these antibodies also served to attract the macrophages’ attention and ensure the minimal peptide’s passport was being checked and approved.
Video is here: http://tinyurl.com/b6dthgb
Via Dr. Stefan Gruenwald
Matching Tumors With The Optimal Cancer Drugs
Science 2.0
The main driver of pancreatic cancer, a mutation in a gene called KRAS, has proven difficult to target with treatments.
Via Brian Shields
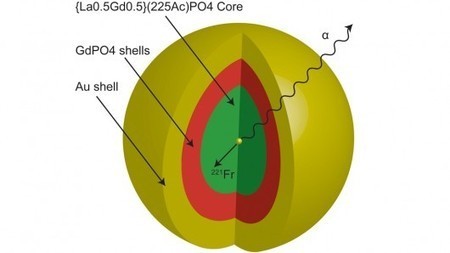
Gold nanoparticles have already shown promise in precisely highlighting brain tumors, “blowing up” individual diseased cells, and developing a lung cancer breath test. Now researchers have created gold nanoparticles that allow an alpha particle-emitting element to be directed to small cancer tumors. The researchers say the gold coating keeps the powerful radioactive particles in place at the cancer site so they do negligible damage to healthy organs and tissue.
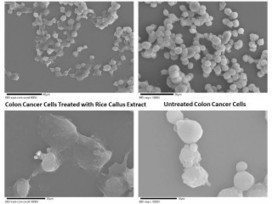
Juice from rice cells can knock out two kinds of human cancer cells as well or better than the potent anti-cancer drug Taxol, a Michigan Technological University scientist has discovered in laboratory tests. Plus, it does something Taxol can’t do: it plays nice with normal cells.
Gene therapy successfully destroyed cancer tumors in patients with leukemia. This development took over 20 years to achieve. Researchers at the University of Pennsylvania engineered patients' own pathogen-fighting T-cells to target a molecule found on the surface of leukemia cells. The altered T-cells were grown outside of the body and infused back into patients suffering from late-stage chronic lymphocytic leukemia (CLL), which affects the blood and bone marrow and is the most common form of leukemia.
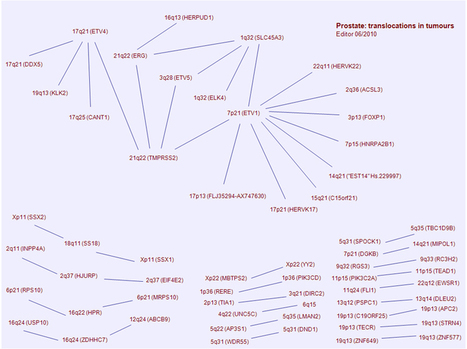
Solid Tumors, Chromosome Abnormalities and Genes involved in Cancer reviewed and published in the Atlas of Genetics and Cytogenetics in Oncology and Haematology...
Via Brian Shields
A drug made from a plant known as “thunder god vine,” or lei gong teng, that has been used in traditional Chinese medicine, wiped out pancreatic tumors in mice, researchers said, and may soon be tested in humans. Mice treated with the compound showed no signs of tumors after 40 days or after discontinuing the treatment, according to researchers at the University of Minnesota’s Masonic Cancer Center. The research, funded by the university and the National Institutes of Health. was published today in the journal Science Translational Medicine. “This drug is just unbelievably potent in killing tumor cells,” said Ashok Saluja, vice chairman of research at the center and the study’s leader, said in a telephone interview. “You could see that every day you looked at those mice, the tumor was decreasing and decreasing, and then just gone.” The plant, also known as Tripterygium wilfordii, contains triptolide, which earlier studies have shown can cause cancer cells to die. In traditional Chinese medicine, the plant is used as a treatment for rheumatoid arthritis. While the researchers hope to start human trials in six months, Saluja said it’s still a long leap from mice to people.
Via Dr. Stefan Gruenwald
A new study from MIT cancer researchers reveals some of the cellular adhesion molecules that are critical to this how cancer cells break free from tumors, spread, and reattach to a new site. Although tumor metastasis (spreading) causes about 90 percent of cancer deaths, the exact mechanism that allows cancer cells to spread from one part of the body to another is not well understood.
"Consumption of spinach can partially offset the damaging effects of many food-induced carcinogens. In tests with laboratory animals, the leafy greens cut the incidence of colon tumors almost in half, from 58 percent to 32 percent. The researchers concluded 'The good news about epigenetics and microRNA alterations is that we may be able to restore normal cell function, via diet and healthy life style choices.' "
|



 Your new post is loading...
Your new post is loading...