Until recently, most immunotherapeutic approaches used to fight cancer were ineffective, counteracted by the tumour’s ability to evade immune attack. However, extensive research has improved our understanding of tumour immunology and enabled the development of novel treatments that can harness the patient’s immune system and prevent immune escape. Over the last few years, through numerous clinical trials and real-world experience, we have accumulated a large amount of evidence regarding the potential for long-term survival with immunotherapy agents in various types of malignancy. The results of these studies have also highlighted a number of recurring observations with immuno-oncology agents, including their potential for clinical application across a broad patient population and for both conventional and unconventional response patterns. Furthermore, given the numerous immune checkpoints that exist and the multiple mechanisms used by tumours to escape the immune system, targeting distinct checkpoint pathways using combination approaches is an attractive therapeutic strategy with the potential to further enhance the antitumour immune response.
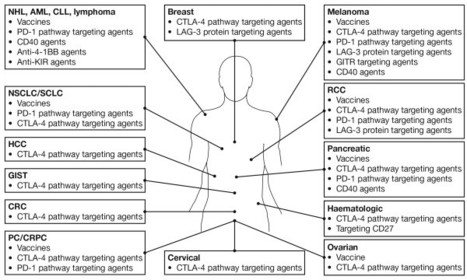
Fig 1. Immuno-oncology agentsa in clinical development across multiple tumour types.aSelected therapies and tumour types are shown: additional agents are, for example in phase 1 studies in patients with solid tumours [12]. AML, acute myeloid leukemia; CLL, chronic lymphocytic leukaemia; CRC, colorectal cancer; CRPC, castration-resistant prostate cancer; CTLA-4, cytotoxic T-lymphocyte antigen-4; GIST, gastrointestinal stromal tumour; HCC, hepatic cell carcinoma; LAG-3, lymphocyte activation gene 3; mAb, monoclonal antibody; NHL, non-Hodgkin lymphoma; NSCLC, non-small cell lung cancer; PC, prostate cancer; PD1, programmed death 1; RCC, renal cell carcinoma; SCLC, small cell lung cancer.
Ascierto and Marincola Journal of Translational Medicine 2014 12:141 doi:10.1186/1479-5876-12-141
Via Krishan Maggon


 Your new post is loading...
Your new post is loading...







This is an Open Access article distributed under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly credited. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated.
What have we learned from cancer immunotherapy in the last 3 years?Paolo A Ascierto1* and Francesco M Marincola2
*Corresponding author: Paolo A Ascierto paolo.ascierto@gmail.com
Author Affiliations
1Unit of Melanoma, Cancer Immunotherapy and Innovative Therapy Unit, Istituto Nazionale Tumori Fondazione “G. Pascale”, Via Mariano Semmola, 80131 Naples, Italy
2Sidra Medical and Research Centre, Doha, Qatar
For all author emails, please log on.
Journal of Translational Medicine 2014, 12:141 doi:10.1186/1479-5876-12-141
The electronic version of this article is the complete one and can be found online at:http://www.translational-medicine.com/content/12/1/141
Received:16 April 2014Accepted:13 May 2014Published:21 May 2014
© 2014 Ascierto and Marincola; licensee BioMed Central Ltd.