The CyaA vector is ideally adapted for the development of therapeutic HPV vaccines.
The CyaA vector was originally engineered at the Institut Pasteur. They modified the adenylate cyclase from Bordetella pertussis by inactivating the enzymatic activity and rendering it permissive to harbor a variety of polypeptide sequences. This was achieved without altering its high affinity to target Antigen Presenting Cells (APC) and its capability to translocate into the cytosol of the APC (Fayolle, Sebo et al. 1996). Vaccines that are built with the CyaA vector are chimeric recombinant proteins consisting of the CyaA protein and the antigen of choice.
The CyaA vector was used in Genticel’s lead investigational vaccine, ProCervix. This therapeutic vaccine is being developed for patient infected with the most common oncogenic genotypes: HPV16 and HPV18. A dose-escalating Phase I clinical trial with ProCervix demonstrated good safety, immunogenicity and a remarkable trend of efficacy to clear viral infection.
Via Krishan Maggon


 Your new post is loading...
Your new post is loading...

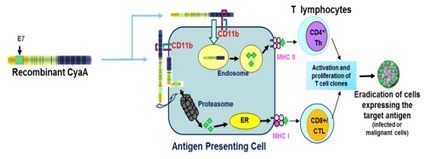






Vaxiclase has the advantage of effectively delivering large or acidic antigens to the cellular immune system. It is therefore a vector of choice for multivalent therapeutic vaccines or therapeutic vaccines carrying negatively charged antigens.
Vaxiclase has been shown to function with antigens exceeding 400 amino acids and very negatively charged antigens, not permissible in the classic CyaA Vector. In addition, Vaxiclase benefits from an improved E. coli gene-expression system allowing for further enhanced yield.
A schematic diagram of a Vaxiclase-based vaccine is given below: